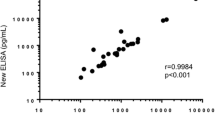
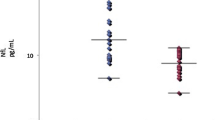
Abstract
Antibodies have different avidities that can be evaluated using modified enzyme-linked immunosorbent assay (ELISA) techniques. We determined levels and avidities of antibodies to light (NFL) and medium (NFM) subunits of neurofilaments and tau protein in serum and cerebrospinal fluid (CSF) from 26 patients and anti-tau antibody levels and their avidities in 20 multiple sclerosis (MS) patients and 20 age- and sex-matched controls. Each sample was analyzed using both standard ELISA and also using a similar ELISA protocol with the addition of urea. The avidities of anti-neurocytoskeletal antibodies were higher in the CSF than those in serum (anti-NFL, p < 0.0001; anti-tau, p < 0.01; anti-NFM, n.s.). There was no relationship between avidities in serum and CSF for individual anti-neurocytoskeletal antibodies. We did not observe the relationship among the avidities of various anti-neurocytoskeletal antibodies. The avidities of anti-tau antibodies in the CSF were significantly higher in the MS patients than those in the controls (p < 0.0001). The study demonstrates the differences in avidities of CSF or serum neurocytoskeletal antibodies measured as the urea resistance by ELISA method. Avidity determination of anti-neurocytoskeletal antibodies could contribute to the evaluation of the immunological status of patients.


Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- CSF:
-
Cerebrospinal fluid
- ELISA:
-
Enzyme-linked immunosorbent assay
- Ig:
-
Immunoglobulin
- MS:
-
Multiple sclerosis
- NFL:
-
The light subunit of neurofilament
- NFM:
-
Medium subunit of neurofilament
- n.s.:
-
Not significant
- PBS:
-
Phosphate-buffered saline
- r :
-
Spearman’s correlation coefficient
References
Avrameas S, Ternynck T (1993) The natural autoantibodies system: between hypotheses and facts. Mol Immunol 30:1133–1142
Bartos A, Fialova L, Soukupova J, Kukal J, Malbohan I, Pit'ha J (2007a) Elevated intrathecal antibodies against the medium neurofilament subunit in multiple sclerosis. J Neurol 254:20–25
Bartos A, Fialova L, Soukupova J, Kukal J, Malbohan I, Pitha J (2007b) Antibodies against light neurofilaments in multiple sclerosis patients. Acta Neurol Scand 116:100–107
Berger T, Rubner P, Schautzer F, Egg R, Ulmer H, Mayringer I, Dilitz E, Deisenhammer F, Reindl M (2003) Antimyelin antibodies as a predictor of clinically definite multiple sclerosis after a first demyelinating event. N Engl J Med 349:139–145
Cui Z, Zhao MH (2005) Avidity of anti-glomerular basement membrane autoantibodies was associated with disease severity. Clin Immunol 116:77–82
de Souza VA, Pannuti CS, Sumita LM, de Andrade Junior HF (1997) Enzyme-linked immunosorbent assay-IgG antibody avidity test for single sample serologic evaluation of measles vaccines. J Med Virol 52:275–279
Fialova L, Bartos A, Soukupova J, Svarcova J, Ridzon P, Malbohan I (2009) Synergy of serum and cerebrospinal fluid antibodies against axonal cytoskeletal proteins in patients with different neurological diseases. Folia Biol (Praha) 55:23–26
Flori P, Tardy L, Patural H, Bellete B, Varlet MN, Hafid J, Raberin H, Sung RT (2004) Reliability of immunoglobulin G antitoxoplasma avidity test and effects of treatment on avidity indexes of infants and pregnant women. Clin Diagn Lab Immunol 11:669–674
Giovannoni G, Chapman MD, Thompson EJ (2006) The role of antibody affinity for specific antigens in the differential diagnosis of inflammatory nervous system disorders. J Neuroimmunol 180:29–32
Huizinga R, Heijmans N, Schubert P, Gschmeissner S, t Hart BA, Herrmann H, Amor S (2007) Immunization with neurofilament light protein induces spastic paresis and axonal degeneration in Biozzi ABH mice. J Neuropathol Exp Neurol 66:295–304
Ilyas AA, Chen ZW, Cook SD (2003) Antibodies to sulfatide in cerebrospinal fluid of patients with multiple sclerosis. J Neuroimmunol 139:76–80
Jianping L, Zhibing Y, Wei Q, Zhikai C, Jie X, Jinbiao L (2006) Low avidity and level of serum anti-Abeta antibodies in Alzheimer disease. Alzheimer Dis Assoc Disord 20:127–132
Jimenez-Jimenez FJ, Hernanz A, Medina-Acebron S, de Bustos F, Zurdo JM, Alonso H, Puertas I, Barcenilla B, Sayed Y, Cabrera-Valdivia F (2005) Tau protein concentrations in cerebrospinal fluid of patients with amyotrophic lateral sclerosis. Acta Neurol Scand 111:114–117
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Lim ET, Sellebjerg F, Jensen CV, Altmann DR, Grant D, Keir G, Thompson EJ, Giovannoni G (2005) Acute axonal damage predicts clinical outcome in patients with multiple sclerosis. Mult Scler 11:532–536
Lublin FD, Reingold SC (1996) Defining the clinical course of multiple sclerosis: results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis. Neurology 46:907–911
Martinez-Yelamos A, Saiz A, Bas J, Hernandez JJ, Graus F, Arbizu T (2004) Tau protein in cerebrospinal fluid: a possible marker of poor outcome in patients with early relapsing-remitting multiple sclerosis. Neurosci Lett 363:14–17
Matheus S, Deparis X, Labeau B, Lelarge J, Morvan J, Dussart P (2005a) Discrimination between primary and secondary dengue virus infection by an immunoglobulin G avidity test using a single acute-phase serum sample. J Clin Microbiol 43:2793–2797
Matheus S, Deparis X, Labeau B, Lelarge J, Morvan J, Dussart P (2005b) Use of four dengue virus antigens for determination of dengue immune status by enzyme-linked immunosorbent assay of immunoglobulin G avidity. J Clin Microbiol 43:5784–5786
McDonald WI, Compston A, Edan G, Goodkin D, Hartung HP, Lublin FD, McFarland HF, Paty DW, Polman CH, Reingold SC, Sandberg-Wollheim M, Sibley W, Thompson A, van den Noort S, Weinshenker BY, Wolinsky JS (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Narita M, Yamada S, Matsuzono Y, Itakura O, Togashi T, Kikuta H (1996) Immunoglobulin G avidity testing in serum and cerebrospinal fluid for analysis of measles virus infection. Clin Diagn Lab Immunol 3:211–215
Poser CM, Paty DW, Scheinberg L, McDonald WI, Davis FA, Ebers GC, Johnson KP, Sibley WA, Silberberg DH, Tourtellotte WW (1983) New diagnostic criteria for multiple sclerosis: guidelines for research protocols. Ann Neurol 13:227–231
Rosenmann H, Meiner Z, Geylis V, Abramsky O, Steinitz M (2006) Detection of circulating antibodies against tau protein in its unphosphorylated and in its neurofibrillary tangles-related phosphorylated state in Alzheimer's disease and healthy subjects. Neurosci Lett 410:90–93
Silber E, Semra YK, Gregson NA, Sharief MK (2002) Patients with progressive multiple sclerosis have elevated antibodies to neurofilament subunit. Neurology 58:1372–1381
Skoda D, Kranda K, Bojar M, Glosova L, Baurle J, Kenney J, Romportl D, Pelichovska M, Cvachovec K (2006) Antibody formation against beta-tubulin class III in response to brain trauma. Brain Res Bull 68:213–216
Sussmuth SD, Reiber H, Tumani H (2001) Tau protein in cerebrospinal fluid (CSF): a blood–CSF barrier related evaluation in patients with various neurological diseases. Neurosci Lett 300:95–98
Svarcova J, Fialova L, Bartos A, Steinbachova M, Malbohan I (2008) Cerebrospinal fluid antibodies to tubulin are elevated in the patients with multiple sclerosis. Eur J Neurol 15:1173–1179
Vyshkina T, Kalman B (2008) Autoantibodies and neurodegeneration in multiple sclerosis. Lab Invest 88:796–807
Zaffaroni M (2003) Biological indicators of the neurodegenerative phase of multiple sclerosis. Neurol Sci 24(Suppl 5):S279–282
Zetterberg H, Jacobsson J, Rosengren L, Blennow K, Andersen PM (2007) Cerebrospinal fluid neurofilament light levels in amyotrophic lateral sclerosis: impact of SOD1 genotype. Eur J Neurol 14:1329–1333
Acknowledgments
The study was supported by a grant from the Ministry of Health No NS 10369-3.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fialová, L., Švarcová, J., Bartos, A. et al. Avidity of anti-neurocytoskeletal antibodies in cerebrospinal fluid and serum. Folia Microbiol 57, 415–419 (2012). https://doi.org/10.1007/s12223-012-0105-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-012-0105-x