Abstract
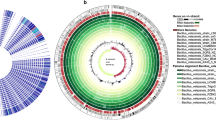
Fifty fluorescent pseudomonads were isolated from rhizospheric soil of green gram from nearby area of Kaziranga, Assam, India and assayed for their extracellular proteinase production. Out of these isolates, 20 were found to be prominent in proteinase production. Genetic diversity of the 20 isolates were analyzed through BOX-PCR fingerprinting and 16S rDNA-RFLP along with three reference strains, viz., Pseudomonas fluorescens (NCIM2099T), Pseudomonas aureofaciens (NCIM2026T), and Pseudomonas aeruginosa (MTCC2582T). BOX-PCR produced two distinct clusters at 56% similarity coefficient and seven distinct BOX profiles. 16S rDNA-RFLP with three tetra-cutters restriction enzymes (HaeIII, AluI, and MspI) revealed two major clusters A and B; cluster A contained only single isolate FPS9 while the rest of 22 isolates belonged to the cluster B. Based on phenotypic characters and 16S rDNA sequence similarity, all the eight highly proteinase-producing strains were affiliated with P. aeruginosa. The proteinase was extracted from two most prominent strains (KFP1 and KFP2), purified by a three-step process involving (NH4)2SO4 precipitation, gel filtration, and ion exchange chromatography. The enzyme had an optimal pH of 8.0 and exhibit highest activity at 60°C and 37°C by KFP1 and KFP2 respectively. The specific activities were recorded as 75,050 (for KFP1) and 81,320 U/mg (for KFP2). The purified enzyme was migrated as a single band on native and SDS-PAGE with a molecular mass of 32 kDa. Zn2+, Cu2+, and Ni2+ ion inhibited the enzyme activity. Enzyme activity was also inhibited by EDTA established as their metallo-proteinase nature.






Similar content being viewed by others
Abbreviations
- EDTA:
-
Ethylenediaminetetraacetic acid
- PMSF:
-
Phenylmethylsulfonylfluoride
- TAE:
-
Tris-acetate ethylenediaminetetraacetic acid
- DEAE:
-
Diethylaminoethyl
- SDS-PAGE:
-
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis
References
Bergey DH, Holt JG et al (1994) Bergey's manual of determinative bacteriology. Williams & Wilkins, Baltimore
Davelos AL, Xiao K, Samac DA, Martin AP, Kinkel LL (2004) Spatial variation in Streptomyces genetic composition and diversity in a Prairie soil. Microb Ecol 448:601–612
Edwards U, Rogall TH, Blocker H, Emde M, Bottger EC (1989) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res 17:7843–7853
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Huessen C, Dowdle EB (1980) Electrophoretic analysis of plasminogen activators in polyacrylamide gels containing sodium dodecyl sulfate and copolymerized substrates. Anal Biochem 102:196–202
Jukes TH, Cantor CR (1969) Evolution of protein molecules. In: Munro HN (ed) Mammalian protein metabolism, vol 3. Academic, New York
Kalaiarasi K, Sunitha P (2009) Optimization of alkaline protease production from Pseudomonas fluorescens isolated from meat waste contaminated soil. Afr J Biotechnol 8:7035–7041
Kamath P, Subrahmanyam VM, Venkata Rao J, Vasanth Raj P (2010) Optimization of cultural conditions for protease production by a fungal species. Indian J Pharm Sci 72:161–166
Kanekar PP, Nilegaonkar SS, Sarnaik SS, Kelkar AS (2002) Optimization of protease activity of alkaliphilic bacteria isolated from an alkaline lake in India. Biores Technol 85:87–93
Louws FJ, Fulbright DW, Stephens CT, de Bruijn FJ (1994) Specific genomic fingerprints of phytopathogenic Xanthomonas and Pseudomonas pathovars and strains generated with repetitive sequences and PCR. Appl Environ Microbiol 60:2286–2295
Mc Arthur JV, Kovacic DA, Smith MH (1988) Genetic diversity in natural populations of a soil bacterium across a landscape gradient. Proc Natl Acad Sci USA 85:9621–9624
Mohen FN, Dileep D, Deepthi D (2005) Potential application of protease isolated from Pseudomonas auriginosa PD100. Biotechnol Ind 8:197–203
Myers N, Russel AM, Cristina G, Gustavo-Foneca AB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Ochman H, Lawrence JG, Groisman EA (2000) Lateral gene transfer and the nature of bacterial innovation. Nature 405:299–304
Oliver A, Canton R, Campo P, Baquero F, Blazquez J (2000) High frequency of hypermutable Pseudomonas aeruginosa in cystic fibrosis lung infection. Science 288:1251–1254
Patel R, Dodia M, Singh S (2005) Extracellular alkaline protease from a newly isolated haloalkaliphilic Bacillus sp.: production and optimization. Process Biochem 40:3569–3575
Rao MB, Tanksale AM, Ghatge MS, Deshpande VV (1998) Molecular and biotechnological aspects of microbial protease. Microbiol Mol Biol Rev 62:597–635
Saikia R, Gogoi DK, Mazumder S, Yadav A, Sarma RK, Bora TC, Gogoi BK (2011a) Brevibacillus laterosporus strain BPM3, a potential biocontrol agent isolated from a natural hot water spring of Assam, India. Microbiol Res 166:216–225
Saikia R, Sarma RK, Yadav A, Bora TC (2011b) Genetic and functional diversity among the antagonistic potential fluorescent pseudomonads isolated from tea rhizosphere. Curr Microbiol 62:434–444
Salwan R, Gulati A, Kasana R (2010) Phylogenetic diversity of alkaline protease-producing psychrotrophic bacteria from glacier and cold environment of Lahaul and Spiti, India. J Basic Microbiol 50:150–159
Shimogaki H, Takeuchi K, Nishino T, Ohdera M, Kudo T, Ohba K, Iwama M, Irie M (1991) Purification and properties of a novel surface active agent and alkaline-resistant protease from Bacillus sp. Agric Biol Chem 55:2251–2258
Vazquez S, Coria S, Cormack W (2004) Extracellular protease from eight psychrotolerant Antarctic strains. Microbiol Res 159:157–166
Acknowledgments
The work is supported by a Network Project (AMAAS), sponsored by Indian Council of Agricultural Research (ICAR), Government of India, New Delhi. The authors are thankful to the funding agency. The authors are also thankful to P.G. Rao, Director, North-East Institute of Science and Technology (CSIR), Jorhat, India for providing necessary facilities to carry out the work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Supplementary material 1
Phenotypic and molecular traits of fluorescent pseudomonads (Pseudomonas aeruginosa) (DOC 39 kb)
Supplementary material 2
Fig. a, b, c Restriction patterns of PCR amplified fragment of 16S rDNA digested with HaeIII, AluI, and MspI, respectively. Lane name with respective isolates: M, 100 bp molecular marker; 1, FPS 26; 2, KFP 5; 3, FPS 9; 4, FPS 324; 5, FPS 14; 6, KFP 7; 7, KFP8; 8, FPS 37; 9, NAM 7; 10, FPS 22; 11, FPS 211; 12, KFP1; 13, FPS 5; 14, FPS 24; 15, KFP3; 16, KFP 9; 17 KFP 2; 18, KFP 357; 19, FPS 18; 20, KFP4; 21, P. fluorescens NCIM2099T; 22, P. aeruginosa MTCC2582T; and 23, P. aureofaciens NCIM 2026T (DOC 180 kb)
Rights and permissions
About this article
Cite this article
Sarma, R.K., Debnath, R., Saikia, R. et al. Phylogenetic analysis of alkaline proteinase producing fluorescent pseudomonads associated with green gram (Vigna radiata L.) rhizosphere. Folia Microbiol 57, 129–137 (2012). https://doi.org/10.1007/s12223-012-0097-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-012-0097-6