Abstract
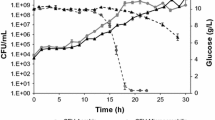
Growth patterns of Cryptococcus neoformans submerged culture in different culture volumes, intensity of agitation and types of sealing were evaluated to better understand the physiological role of hypoxia response in this yeast. When low intensity agitation was set at high culture volumes and air exchange between the cultivation vessel and external environment was not abolished completely, the cells proliferated slowly but steadily. On the other hand, when the intensity of agitation was high but the vessel was withheld from fresh air supply, the cells first proliferated rapidly, then arrested completely and finally died. Therefore, the central strategy of C. neoformans here seems to lie in its proliferation-rate adjustment to the available oxygen levels and not in its capacity to survive under anoxia. The data support the opinion that the cultures grown under limited aeration (even though not completely withheld from fresh air supply) are much closer to the real cryptococcal life in human tissues than conventional well-aerated exponential cultures.
Similar content being viewed by others
References
Chang Y.C., Stins M.F., Mccaffery M.J., Miller G.F., Pare D.R., Dam T., Paul-Satyaseela M., Kim K.S., Kwon-Chung K.J.: Cryptococcal yeast cells invade the central nervous system via transcellular penetration of the blood-brain barrier. Infect. Immun.72, 4985–4995 (2004).
Chang Y.C., Bien C.M., Lee H., Espenshade P.J., Kwon-Chung K.J.: Sre1p, a regulator of oxygen sensing and sterol homeostasis, is required for virulence in Cryptococcus neoformans. Mol.Microbiol.64, 614–629 (2007).
Chun C.D., Liu O.W., Madhani H.D.: A link between virulence and homeostatic responses to hypoxia during infection by the human fungal pathogen Cryptococcus neoformans. PLoS Pathogens3, e22 (2007).
Ernst J.F., Tielker D.: Responses to hypoxia in fungal pathogens. Cell MicrobiolDOI: 10.1111/j.1462-5822.2008.01259.x (2008).
Isett K., George H., Herber W., Amanullah A.: Twenty-four-well plate miniature bioreactor high-throughput system: assessment for microbial cultivations. Biotechnol.Bioeng.98, 1017–1028 (2007).
Matsushita T., Brendzel A.M., Shotola M.A., Groh K.R.: Electrical determination of viability in saline-treated mouse myeloma cells. Biophys.J.39, 41–47 (1982).
Odds F.C., DE Backer T., Dams G., Vranckx L., Woestenborghs F.: Oxygen as limiting nutrient for growth of Cryptococcus neoformans. J.Clin.Microbiol.33, 995–997 (1995).
Ohkusu M., Hata K., Takeo K.: Bud emergence is gradually delayed from S to G2 with progression of growth phase in Cryptococcus neoformans. FEMS Microbiol.Lett.194, 251–255 (2001a).
Ohkusu M., Raclavský V., Takeo K.: Deficit in oxygen causes G(2) budding and unbudded G(2) arrest in Cryptococcus neoformans. FEMS Microbiol.Lett.204, 29–32 (2001b).
Raclavský V., Pavlíček J., Novotný R., Moráňová Z., Ohkusu M., Trtková J., Takeo K., Kawamoto S.: Peculiar clusters of daughter cells observed in Cryptococcus neoformans grown in sealed microtiter plates. Folia Microbiol.54, 369–371 (2009).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raclavský, V., Husičková, V., Moráňová, Z. et al. Growth strategy of the pathogenic yeast Cryptococcus neoformans submerged culture under different cultivation formats. Folia Microbiol 54, 349–352 (2009). https://doi.org/10.1007/s12223-009-0044-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-009-0044-3