Abstract
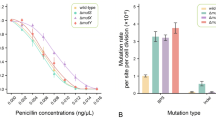
The effect of H2O2 on the induction of ciprofloxacin (CFL) resistant mutants of Salmonella enterica subsp. enterica serovar Typhimurium was evaluated and determinants of CFL resistance in the mutants were analyzed. Factors associated with CFL resistance in H2O2-induced mutants included (i) mutations in gyrA gene, predominantly (63 %) Asp87→Asn and less (37 %) Ser83→Phe substitutions, (ii) mutations in the regulatory genes of MarRAB or SoxRS or in the individual structural genes of these operons. Such mutations are induced by H2O2 in a much lower extent. Reduced OmpF expression simultaneously with enhanced efflux was detected only in one mutant strain and 20 % of mutant strains had increased CFL efflux from the cells.
Similar content being viewed by others
Abbreviations
- AS-PCR-RFLP:
-
allele specific-PCR-restriction fragment length polymorphism
- CFL:
-
ciprofloxacin
- MIC(s):
-
minimal inhibitory concentration(s)
- OMP:
-
outer-membrane protein
- TCS:
-
3,3′,4′,5-tetrachlorosalicylanilide
References
Ames B.N., Shigenaga M.K., Hagen T.M.: Oxidants, antioxidants, and the degenerative disease of aging. Proc.Nat.Acad.Sci.USA 90, 7915–7922 (1993).
Barnass S., Franklin J., Tabaqchali S.: The successful treatment of multiresistant non-enteric salmonellosis with seven day oral ciprofloxacin. J.Antimicrob.Chemother. 25, 299–300 (1990).
Brown J.C., Thomson C.J., Aymes S.G.B.: Mutations of the gyrA gene of clinical isolates of Salmonella typhimurium and three other Salmonella species leading to decreased susceptibilities to 4-quinolone drugs. J.Antimicrob.Chemother. 37, 351–356 (1996).
Chou J.H., Greenberg J.T., Demple B.: Posttranscriptional repression of Escherichia coli OmpF protein in response to redox stress: positive control of the micF antisense RNA by the soxRS locus. J.Bacteriol. 175, 1026–1030 (1993).
Conrad S., Oethinger M., Kaifel K., Klotz R., Marre R., Kera E.V.: gyrA mutations in high-level fluoroquinolone-resistant clinical isolates of Escherichia coli. J.Antimicrob.Chemother. 38, 443–455 (1996).
Eriksson S., Bjorkman J., Borg S., Pettersson S., Andersson D.I., Rhen M.: Salmonella typhimurium mutants that downregulate phagocyte nitric oxide production. Cell Microbiol. 2, 239–250 (2000).
Giraud E., Brisabois A., Martel J.L., Chaslus-Dancla E.: Comparative studies of mutations in animal isolates and experimental in vitro-and in vivo-selected mutants of Salmonella spp. suggest a counterselection of highly fluoroquinolone-resistant strains in the field. Antimicrob.Agents Chemother. 43, 2131–2137 (1999).
Giraud E., Cloeckaert A., Kerboeuf D., Chaslus-Dancla E.: Evidence for active efflux as the primary mechanism of resistance to ciprofloxacin in Salmonella enterica serovar Typhimurium. Antimicrob.Agents Chemother. 44, 1223–1228 (2000).
Hirai K., Aoyama H., Suzue S., Irikura T., Iyobe S., Mitsuhashi S.: Isolation and characterization of norfloxacin-resistant mutants of Escherichia coli K-12. Antimicrob.Agents Chemother. 30, 248–253 (1986).
Koutsolioutsou A., Martins E.A., White D.G., Levy S.B., Demple B.: A soxRS-constitutive mutation contributing to antibiotic resistance in a clinical isolate of Salmonella enterica (serovar typhimurium). Antimicrob.Agents Chemother. 45, 38–43 (2001).
Oethinger M., Kern V., Jellen-Ritter A.S., Mcmurry L.M., Levy S.B.: Inefectiveness of topoisomerase mutations in mediating clinically significant fluoroquinolone resistance in Escherichia coli in the absence of the AcrAB efflux pump. Antimicrob. Agents Chemother. 44, 10–13 (2000).
Okusu H., Ma D., Nikaido H.: AcrAB efflux pumps plays a major role in the antibiotic resistance phenotype of Escherichia coli multiple-antibiotic-resistance (Mar) mutants. J.Bacteriol. 178, 306–308 (1996).
Piddock L.J.V.: Fluoroquinolone resistance in Salmonella serovars isolated from humans and food animals. FEMS Microbiol.Rev. 26, 3–16 (2002).
Randall L.P., Woodward M.J.: Multiple antibiotic resistance (mar) locus in Salmonella enterica serovar typhimurium DT104. Appl. Environ.Microbiol. 67, 1190–1197 (2001).
Sande M., Chambers H.: Antimicrobial agents, general considerations, in J. Hardman, L. Limbird, P. Molinoff, R.W. Ruddon, A.G. Gilman (Eds): Goodman and Gilmans’s the Pharmacological Basis of Therapeutics, 9th ed., section IX: Chemotherapy of microbial diseases. McGraw-Hill, New York 1996.
Shiloh M.U., Nathan C.F.: Reactive nitrogen intermediates and the pathogenesis of Salmonella and mycobacteria. Curr.Opin.Microbiol. 3, 35–42 (2000).
Tavío M.M., Villa J., Ruiz J., Martín-Sánchez A.M., Jiménez de Anta M.T.: Mechanisms involved in the development of resistance to fluoroquinolones in Escherichia coli isolates. J.Antimicrob.Chemother. 44, 735–742 (1999).
Wang D., Kreutzer D.A., Essigmann J.M.: Mutagenicity and repair of oxidative DNA damage: insight from studies using defined lesions. Mutat.Res. 480, 99–115 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Birošová, L., Mikulášová, M. The mechanism of ciprofloxacin resistance in dihydrogen peroxide-induced mutants of Salmonella enterica subsp. enterica serovar typhimurium consists mainly in mutations in gyrA gene and less in mutations affecting ciprofloxacin uptake. Folia Microbiol 53, 368–372 (2008). https://doi.org/10.1007/s12223-008-0057-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-008-0057-3