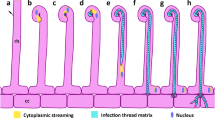
Abstract
In rhizobial symbiosis with legume plant hosts, the symbiotic tissue in the root nodules of indeterminate type is localized to the basal part of the nodule where the symbiotic zones contain infected cells (IC) interspersed with uninfected cells (UC) that are devoid of rhizobia. Although IC are easily distinguished in nodule sections using standard histochemical techniques, their observation in intact nodules is hampered by nodule tissue characteristics. Tagging of Rhizobium leguminosarum bv. viciae strain 128C30 with a constitutively expressed gene for green fluorescent protein (nonshifted mutant form cycle3) in combination with the advantages of the tiny nodules formed by Vicia tetrasperma (L.) Schreb. allowed for vital observation of symbiotic tissue using fluorescence microscopy. Separation of a red-shifted background channel and digital image stacking along z-axis enabled us to construct a nodule image in a classical fluorescence microscopy of nodules exceeding 1 mm in diameter. In parallel, visualization of nodule bacteria inside the symbiotic tissue by confocal microscopy at the excitation wavelength 488 nm clearly distinguished IC/UC pattern in the nodule virtual sections and revealed red-shifted fluorescence of nonrhizobial origin. This signal was located on the periphery of IC and increased with their degradation, thus suggesting accumulation of secondary metabolites, presumably flavonoids. The simultaneous detection of bacteria and secondary metabolites can be used for monitoring changes to intact nodule physiology in the model legumes. The advantage of V. tetrasperma as a suggested laboratory model for pea cross-inoculation group has been demonstrated.
Similar content being viewed by others
Abbreviations
- GFP:
-
green fluorescent protein
- IC:
-
infected cells
- UC:
-
uninfected cells
References
Beijerinck M.W.: Die Bacterien der Papilionaceenknöllchen. Bot.Z. 46, 725–804 (1888).
Boivin C., Camut S., Malpica C.A., Truchet G., Rosenberg C.: Rhizobium meliloti genes encoding catabolism of trigonelline are induced under symbiotic conditions. Plan Cell 2, 1157–1170 (1990).
Chovanec P., Novák K.: Visualization of nodulation gene activity on the early stages of Rhizobium leguminosarum bv. viciae symbiosis. Folia Microbiol. 50, 323–331 (2005).
Crameri A., Whitehorn E.A., Tate E., Stemmer W.P.C.: Improved green fluorescent protein by molecular evolution using DNA shuffling. Nature Biotechnol. 14, 315–319 (1996).
Cubitt A.B., Heim R., Adams S.R., Boyd A.E., Gross L.A., Tsien R.Y.: Understanding, improving and using green fluorescent proteins. Trends Biochem.Sci. 20, 448–455 (1995).
DeBilly F., Barker D.G., Gallasci P., Truchet G.: Leghemoglobin gene transcription is triggered in a single cell layer in the indeterminate nitrogen-fixing root nodule of alfalfa. Plant J. 1, 27–35 (1991).
Figurski D.H., Helinski D.R.: Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc.Nat.Acad.Sci.USA 76, 1648–1652 (1979).
Franssen H.J., Vijn I., Yang W.C., Bisseling T.: Developmental aspects of the Rhizobium-legume symbiosis. Plant Mol.Biol. 19, 89–107 (1992).
Gage D.J., Bobo T., Long S.R.: Use of green fluorescent protein to visualize the early events of symbiosis between Rhizobium meliloti and alfalfa (Medicago sativa). J.Bacteriol. 178, 7159–7166 (1996).
Handberg K., Stougaard J.: Lotus japonicus, an autogamous, diploid legume species for classical and molecular genetics. Plant J. 2, 487–496 (1992).
Harborne J.B.: The comparative biochemistry of phytoalexin induction in plants. Biochem.Syst.Ecol. 27, 335–367 (1999).
Herrero M., de Lorenzo V., Timmis K.N.: Transposon vectors containing non-antibiotic resistance selection markers for cloning and stable chromosomal insertion of foreign genes in Gram-negative bacteria. J.Bacteriol. 172, 6557–6567 (1990).
Leyva A., Palacios J.N., Ruiz-Argueso T.: Conserved plasmid hydrogen-uptake (hup)-specific sequences within Hup+ Rhizobium leguminosarum strains. Appl.Environ.Microbiol. 53, 2539–2543 (1987).
Masterson R.V., Prakash R.K., Atherly A.G.: Conservation of symbiotic nitrogen fixation gene sequences in Rhizobium japonicum and Bradyrhizobium japonicum. J.Bacteriol. 163, 21–26 (1985).
Mathesius U., Bayliss C., Weinman J.J., Schlaman H.R.M., Spaink H.P., Rolfe B.G., McCully M.E., Djordjevic M.A.: Flavonoids synthesized in cortical cells during nodule initiation are early developmental markers in white clover. Mol.Plant-Microbe Interact. 11, 1223–1232 (1998).
Mellor R.B.: Bacteriods in the Rhizobium-legume symbiosis inhabit a plant internal lytic compartment: implications for other microbial endosymbioses. J.Exp.Bot. 40, 831–839 (1989).
Newcomb W.: A correlated light and electron microscopic study of symbiotic growth and differentiation in Pisum sativum root nodules. Can.J.Bot. 54, 2163–2186 (1976).
Niehaus K., Kapp D., Pühler A.: Plant defense and delayed infection of alfalfa pseudonodules induced by an exopolysaccharide (EPS I)-defficient Rhizobium meliloti mutant. Planta 190, 415–425 (1993).
Novák K., Chovanec P., Škrdleta V., Kropáčová M., Lisá L., Němcová M.: Effect of exogenous flavonoids on nodulation of pea (Pisum sativum L.). J.Exp.Bot. 53, 1735–1745 (2002).
Novák K., Lisá L., Škrdleta V.: Rhizobial nod gene-inducing activity in pea nodulation mutants: dissociation of nodulation and flavonoid response. Physiol.Plant. 120, 546–555 (2004).
Oke V., Long S.R.: Bacterial genes induced within the nodule during the Rhizobium-legume symbiosis. Mol.Microbiol. 32, 837–849 (1999).
Parniske M., Fischer H.-M., Hennecke H., Werner D.: Accumulation of the phytoalexin glyceollin I in soybean nodules infected by a Bradyrhizobium japonicum nifA mutant. Z.Naturforsch. 46c, 318–320 (1991).
Penmetsa R.V., Cook D.R.: A legume ethylene-insensitive mutant hyperinfected by its rhizobial symbiont. Science 275, 527–530 (1997).
Quandt H.J., Pühler A., Broer I.: Transgenic root nodules of Vicia hirsuta — a fast and efficient system for the study of gene expression in indeterminate-type nodules. Mol.Plant-Microbe Interact. 6, 699–706 (1993).
Recourt K., Schripsema J., Kijne J.W., Van Brussel A.A.N., Lugtenberg B.J.J.: Inoculation of Vicia sativa subsp. nigra roots with Rhizobium leguminosarum biovar viciae results in release of nod gene activating flavanones and chalcones. Plant Mol.Biol. 16, 841–852 (1991).
Sambrook J., Fritsch E.F., Maniatis T.: Molecular Cloning: a Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor (USA) 1989.
Scheres B., Van Engelen F., Van der Knaap E., Van de Wiel C., Van Kammen A., Bisseling T.: Sequential induction of nodulin gene expression in the developing pea nodule. Plant Cell 2, 687–700 (1990).
Schmidt P.E., Broughton W.J., Werner D.: Nod factors of Bradyrhizobium japonicum and Rhizobium sp. NGR234 induce flavonoid accumulation in soybean root exudate. Mol.Plant-Microbe Interact. 7, 384–390 (1994).
Sharma S.B., Signer E.R.: Temporal and spatial regulation of the symbiotic genes of Rhizobium meliloti in planta revealed by transposon Tn5-gusA. Genes & Dev. 4, 344–356 (1990).
Simon R., Priefer U., Pühler A.: A broad host range mobilization system for in vivo genetic engineering: transposon mutagenesis in Gram-negative bacteria. Bio/Technology 1, 784–791 (1983).
Spaink H.P.: Root nodulation and infection factors produced by rhizobial bacteria. Ann.Rev.Microbiol. 54, 257–288 (2000).
Stuurman N., Bras C.P., Schlaman H.R.M., Wijfjes A.H.M., Bloemberg G., Spaink H.P.: Use of green fluorescent protein color variants expressed on stable broad-host-range vectors to visualize rhizobia interacting with plants. Mol.Plant-Microbe Interact. 13, 1163–1169 (2000).
Tsyganov V.E., Voroshilova V.A., Priefer U.B., Borisov A.Y., Tikhonovich I.A.: Genetic dissection of the initiation of the infection process and nodule tissue development in the Rhizobium-pea (Pisum sativum L.) symbiosis. Ann.Bot. 89, 357–366 (2002).
Van Brussel A.A.N., Tak T., Wetselaar A., Pees E., Wijffelman C.A.: Small Leguminosae as test plants for nodulation of Rhizobium leguminosarum and other rhizobia and agrobacteria harboring a leguminosarum Sym-plasmid. Plant Sci.Lett. 27, 317–325 (1982).
Vance C.P., Boylan K.L.M., Stade S.: Host plant determinants of legume nodule function: similarities to plant disease situation, pp. 271–287 in S. Nishimura, C.P. Vance, N. Doke (Eds): Molecular Determinants of Plant Diseases. Japan Sci. Soc. Press-Springer, Tokyo-Berlin 1987.
Vasse J., De Billy F., Truchet G.: Abortion of infection during the Rhizobium meliloti—alfalfa symbiotic interaction is accompanied by a hypersensitive reaction. Plant J. 4, 555–566 (1993).
Vincent J.M.: A Manual for the Practical Study of the Root Nodule Bacteria. Blackwell, Oxford (UK) 1970.
Voroshilova V.A., Boesten B., Tsyganov V.E., Borisov A.Y., Tikhonovich I.A., Priefer U.B.: Effect of mutations in Pisum sativum L. genes blocking different stages of nodule development on the expression of late symbiotic genes in Rhizobium leguminosarum bv. viciae. Mol.Plant-Microbe Interact. 14, 471–476 (2001).
Wilson K.J.: Molecular techniques for the study of rhizobial ecology in the field. Soil Biol.Biochem. 27, 501–514 (1995).
Wolff A.B., Werner D.: Defense reactions in Rhizobium-legume symbiosis: phytoalexin concentration in Vicia faba nodules is affected by the host plant genotype. Z.Naturforsch 45c, 958–962 (1990).
Xi C., Lambrecht M., Vanderleyden J., Michiels J.: Bi-functional gfp- and gusA-containing mini-Tn5 transposon derivatives for combined gene expression and bacterial localization studies. J.Microbiol.Meth. 35, 85–92 (1999).
Xi C., Dirix G., Hofkens J., De Schryver F.C., Vanderleyden J., Michiels J.: Use of dual marker transposons to identify new symbiosis genes in Rhizobium. Microbial Ecol. 41, 325–332 (2001).
Yang W.C., Cremers H.C.J.C., Hogendijk P., Katinakis P., Wijffelman C.A., Franssen H., Van Kammen A., Bisseling T.: In situ localization of chalcone synthase messenger RNA in pea root nodule development. Plant J. 2, 143–151 (1992).
Zaat S.A.J., Wijffelman C.A., Mulders I.H.M., Van Brussel A.A.N., Lugtenberg B.J.J.: Root exudates of various host plants of Rhizobium leguminosarum contain different sets of inducers of Rhizobium nodulation genes. Plant Physiol. 86, 1298–1303 (1988).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chovanec, P., Hovorka, O. & Novák, K. Visualization of symbiotic tissue in intact root nodules of Vicia tetrasperma using GFP-marked Rhizobium leguminosarum bv. viciae . Folia Microbiol 53, 139–146 (2008). https://doi.org/10.1007/s12223-008-0020-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-008-0020-3