Abstract
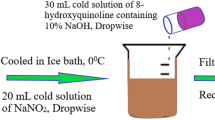
Anthrone- and aniline-based azo dyes (4a-4i) are synthesized and well characterized. These dyes are further applied to wool, nylon, polyester, and acrylic fabrics. Fastness properties, including light, wash, and rub for dyed fabrics, were studied and found to be better for polyester and nylon than wool and acrylic substrate. Better sublimation fastness was obtained on polyester than nylon at 210 °C. The ultraviolet protection factor (UPF) of all dyed textiles was detected to block ultraviolet radiation (UVR) in the range of 93–97%. Nylon, wool, and acrylic dyed fabric showed excellent, while polyester showed good ultraviolet protection activity. All the dyes are effective against “(S. aureus) gram + ve” and “(K. pneumoniae) gram −ve” bacteria. The fabric dyed with the azo dye obtained from the p-COOH (4a), p-CH3 (4d), p-NO2 (4 g), and p-NHCOCH3 (4i) substituted anilne derivatives were as found to have ≥ 98% potent inhibition. The HOMO–LUMO gap and other chemical reactivity descriptors, such as electrophilicity index, chemical hardness, and chemical potential, are calculated using the density functional theory (DFT) method.










Similar content being viewed by others
References
K.B. Schloss, S.E. Palmer, Attent. Percept. Psychophys. 73, 551 (2011)
E. Hagan, J. Poulin, Herit. Sci. 9, 33 (2011)
S. Popli, U.D. Patel, Int. J. Environ. Sci. Tech. 12, 405 (2015)
M. Berradi, R. Hsissou, M. Khudhair, M. Assouag, O. Cherkaoui, A. El Bachiri, and A. El Harfi; Heliyon., 5 (2019).
J.O. Otutu, E. Osabohien, E.M. Efurhievwe, Orient. J. Chem. 26, 31 (2010)
S. Benkhaya, S. M’rabet, and A El Harfi; Heliyon., 6, (2020).
S. Benkhaya, S. El Harfi, A. El Harfi, Appl. J. Envir. Eng. Sci. 3, 311 (2017)
A. Mohammadi, B. Khalili, M. Tahavor, Spectrochim. Acta. Part A Mol. Biomol. Spectrosc. 150, 799 (2015)
J.O. Otutu, Int. J. Recent Res. Appl. Stud. 15, 292 (2013)
F.L. Coelho, C.A. Braga, G.M. Zanotto, E.S. Gil, L.F. Campo, P.F.B. Gonçalves, F.S. Rodembusch, S.F. de Santos, Sens. Actuators B Chem. 259, 514 (2018)
P. Prajongtat, S. Suramitr, S. Nokbin, K. Nakajima, K. Mitsuke, S. Hannongbua, J. Mol. Graph Model. 76, 551 (2017)
N.N. Ayare, M.C. Sreenath, S. Chitrambalam, I.H. Joe, N. Sekar, Mol. Phys. 118, 1 (2020)
S. Manickasundaram, P. Kannan, Q.M.A. Hassan, P.K. Palanisamy, J. Mater. Sci. Mater. Electron. 19, 1045 (2008)
B. Hemmateenejad, M Shamsipur, R. Miria, M. Elyasia, F. Foroghiniaa, and H. Sharghi; Anal. Chim. Acta., 25, 0 (2008).
H. Prinz, P. Schmidt, K.J. Böhm, S. Baasner, K. Müller, M. Gerlach, E.G. Günther, E. Unger, Bioorganic Med. Chem. 19, 4183 (2011)
C. Roma-rodrigues, D. Peixoto, L.M. Ferreira, P.V. Baptista, A.R. Fernandes, P.S. Branco, Bioorg. Chem. 99, 103849 (2020)
H.M. Dardeer, A.S. Assran, S.A. Al-Hussain, A. Toghan, J. Mol. Struct. 1226, 129359 (2021)
J.D. Porter, O. Vivas, C.D. Weaver, A. Alsafran, E. Dimilo, L.A. Arnold, J.A. Dickson, C. Dockendroff, Bioorg. Med. Chem. Lett. 29, 126681 (2019)
G. Yen, P. Duh, D. Chuang, Food Chem. 70, 437 (2000)
H.M. Dardeer, E.E. Elboray, G.S. Mohamed, Polycycl. Aromat. Compd. 40, 622 (2020)
V.A. Online, E.J. Dufek, M. Hoffman, A.G. Sykes, RSC Adv. 00, 1 (2013)
V.E. Turula, T. Gore, S. Singh, R.G. Arumugham, Anal. Chem. 82, 1786 (2010)
A. Laurentin, C.A. Edwards, Anal. Biochem. 315, 143 (2003)
P. M. Cheuka, G. Mayoka, P. Mutai, and K. Chibale; Molecules, 22 (2017).
M. Al-Tamimi, S.M. Al-Massarani, A.A. El-Gamal, O.A. Basudan, M.S. Abdel-Kader, W.M. Mageed, Plants 9, 1 (2020)
M. M. Mohammed; Nat. Prod. Chem. Res., 4 (2016).
Z.H. Wu, D. Liu, Y. Xu, J.L. Chen, W.H. Lin, Chin. J. Nat. Med. 16, 219 (2018)
R. Penthala, G. Heo, H. Kim, I.Y. Lee, E.H. Ko, Y.A. Son, J. CO2 Util. 38, 49 (2020)
S. Benkhaya, S. M’rabet, A. El Harfi, Inorg. Chem. Commun. 115, 107891 (2020)
N.G. Khaligh, S.B.A. Hamid, H. Hazarkhani, Inorg. Nano-Metal Chem. 47, 10 (2017)
C.C. Hsueh, B.Y. Chen, C.Y. Yen, J. Hazard. Mater. 167, 995 (2009)
V.R. Mishra, N. Sekar, J. Fluoresc. 27, 1101 (2017)
N.N. Ayare, S.H. Ramugade, N. Sekar, Dyes Pigm. 163, 692 (2019)
K.H. Meyer, Org. Synth. 8, 8 (1928)
A.B. Pawar, G. Mahajan, R.V. Adivarekar, Indian J. Sci. Res. 14, 2 (2017)
H.R. Maradiya, Turkish J. Chem. 25, 441 (2001)
R.D. Kale, A. Pratap, P.B. Kane, V.G. Gorade, Indian J. Sci. Res. 14, 257 (2017)
M. H. Imran; J. Text. Sci. Eng., 07, (2017).
G. B. Ramaiah, A. P. Ari; AIP Conf. Proc., 2162 (2019).
ISO 105-B02: Colour fastness to artificial light: Xenon arc fading lamp test (2014).
ISO 105-C06: Colour fastness to domestic and commercial laundering (2010).
ISO 105-X11: Colour fastness to hot pressing (1996).
AATCC 116: Colorfastness to Crocking: Rotary Vertical Crockmeter: AATCC Technical Manual (2018).
AS/NZS 4399 Australia/New Zealand Standard – Sun Protective Clothing – Evaluation and Classification (1996).
AATCC 112–2004: Antibacterial Finishes on Textile Materials: Assessment of, AATCC Technical Manual (2007).
H.E. Fierz-David, L. Blange, Fundamental processes of dye chemistry, 5th edn. (Interscience Publishers, INC, New York, 1949)
Y. Hirao, T. Saito, H. Kurata, T. Kubo, Angew. Chem. Int. Ed. 54, 2402 (2015)
C. Hansch, A. Leo, R.W. Taft, Chem. Rev. 91, 2 (1991)
P. Hammett, Louis. J. Am. Chem. Soc. 59, 1 (1937)
M. Clark; Handbook of Textile and Industrial Dyeing, Vol. 2, Applications of dyes Woodhead Publishing Series in Textiles (2011).
M.A. Rauf, S. Hisaindee, N. Saleh, RSC Adv. 5, 18097 (2015)
P.M. Hans-Gert Korth, J. Org. Chem. 78, 7674 (2013)
B. Yoshikazu, S.M.T. Iho, Ind. Chem. Mag. 67, 177 (1964)
E.R. Silva, E.L. Dall’Oglio, L.G. Vasconcelos, E.B. Morais, Braz. J. Biol. 82, e237386 (2021)
W. Liu, W. Hu, and J. Liu; AIP Conf. Proc., 1794 (2017).
B.Y. Chen, K.W. Lin, Y.M. Wang, C.Y. Yen, J Hazard Mater. 166, 187 (2009)
D.R. Roy, R. Parthasarathi, B. Maiti, V. Subramanian, P.K. Chattaraj, Bioorg. Med. Chem. 13, 3405 (2005)
V. Sahu, P. Sharma, A. Kumar, J. Chil. Chem. Soc. 59, 1 (2014)
R.G. Parr, L. von Szentpaly, S. Liu, J. Am. Chem. Soc. 121, 1922 (1999)
C.W. Ghanavatkar, V.R. Mishra, N. Sekar, Spectrochim Acta - Part A Mol. Biomol. Spectrosc. 230, 118064 (2020)
V.R. Mishra, C.W. Ghanavatkar, N. Sekar, Spectrochim Acta - Part A Mol. Biomol. Spectrosc. 223, 117353 (2019)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patil, P.S., Gupta, P.O., Ingole, G.S. et al. Anthrone-Based Carbocyclic Azo Dyes: Synthesis, Dyeing, UV Protection, Anti-microbial Activity and Computational Study. Fibers Polym 24, 1285–1296 (2023). https://doi.org/10.1007/s12221-023-00054-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12221-023-00054-6