Abstract
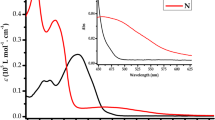
D-π-A system 3 has been designed and synthesized from 4-hydroxy benzaldehyde and isophorone. The dye 3 exhibited high selectivity for cyanide ion detection over other anions with the association constant of the dye 3-CN− complexes were found to be of 4.65×103 M−1. 1H NMR studies indicated that the cyanide ion sensing by the dye 3 is due to the formation of H-bonds between CN− ions and OH moiety of the dye 3. In the presence of CN− ion the color of dye 3 is purple. After the addition of Cu2+ ion, the purple dye 3-CN solution has been turn to yellow color with the binding constant to be 6.17×103 M−1. Besides, dye 3 has also been sense volatile organic compounds (VOCs) such as ethylamine, diethylamine and triethylamine. The order of the binding constant of dye 3 with various amines are ethylamine>diethylamine> triethylamine. The structural and electronics properties of the dye 3 and their cyanide complexes were also investigated using Material Studio 4.4 program.
Similar content being viewed by others
References
D. M. Burland, R. D. Miller, and C. A. Walsh, Chem. Rev., 94, 31 (1994).
D. R. Kanis, T. J. Marks, and M. A. Ratner, Chem. Rev., 94, 195 (1994).
A. P. D. Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher, and T. E. Rice, Chem. Rev., 97, 1515 (1997).
A. Mishra, R. K. Behera, P. K. Behera, B. K. Mishra, and G. B. Behera, Chem. Rev., 100, 1973 (2000).
Z. R. Grabowski, K. Rotkiewicz, and W. Rettig, Chem. Rev., 103, 3899 (2003).
C. R. Moylan, S. Ermer, S. M. Lovejoy, I. H. McComb, D. S. Leung, R. Wortmann, P. Krdmer, and R. J. Twieg, J. Am. Chem. Soc., 118, 12950 (1996).
Y. Suzuki and K. Yokoyama, J. Am. Chem. Soc., 127, 17799 (2005).
Z. Q. Guo, W. H. Zhu, and L. J. H. Shen Tian, Angew. Chem. Int. Ed., 46, 5549 (2007).
Y. S. Yao, J. X. Xiao, S. Wang, Z. B. Deng, and B. W. Zhang, Adv. Funct. Mater., 16, 709 (2006).
B. Liu, W. Zhu, Q. Zhang, W. Wu, M. Xu, Z. Ning, Y. Xie, and H. Tian, Chem. Commun., 1766 (2009).
M. Meyer, J. C. Mialocq, and B. Perly, J. Phys. Chem., 94, 98 (1990).
S. K. Pal, D. Sukul, D. Mandal, and K. Bhattacharyya, J. Phys. Chem. B, 104, 4529 (2000).
B. Boldrini, E. Cavalli, A. Painelli, and F. Terenziani, J. Phys. Chem. A, 106, 6286 (2002).
L. Chen, G. Qian, Y. Cui, X. Jin, Z. Wang, and M. Wang, J. Phys. Chem. B, 110, 19176 ( 2006).
T. Suksai and T. Tuntulani, Chem. Soc. Rev., 32, 192 (2003).
A. Bianchi, K. Bowman-James, and E. García-Espana, “Supramolecular Chemistry of Anions”, pp.1–44, Wiley-VCH, New York, 1997.
S. Park and H. J. Kim, Chem. Commun., 46, 9197 (2010).
N. Kumari, S. Jha, and S. Bhattacharya, J. Org. Chem., 76, 8215 (2011).
S. Wang and S. H. Kim, Curr. Appl. Phys., 9, 783 (2009).
Y. A. Son, J. H. Hwang, S. Wang, J. S. Bae, and S. H. Kim, Fiber. Polym., 10, 272 (2009).
Y. A. Son, S. Y. Gwon, and S. H. Kim, Mol. Cryst. Liquid. Cryst., 538, 327 (2011).
E. M. Lee, S. Y. Gwon, B. C. Ji, S. Wang, and S. H. Kim, Dyes Pigments., 92, 542 (2011).
S. H. Kim, S. Y. Gwon, J. S. Bae, and Y. A. Son, Spectroc. Acta Pt. A-Molec. Biomolec. Spectr., 78, 234 (2011).
R. M. Wachter, D. Yarbrough, K. Kallio, and S. J. Remington, J. Mol. Biol., 301, 157 (2000).
M. A. Khan, A. W. McCulloch, and A. G. McInnes, Can. J. Chem., 63, 2119 (1985).
D. K. Smith, Org. Biomol. Chem., 1, 3874 (2003).
E. M. Lee, S. Y. Gwon, B. C. Ji, and S. H. Kim, J. Lumines, 131, 1211 (2011).
B. Delley, J. Chem. Phys., 92, 508 (1990).
B. Delley, J. Chem. Phys., 113, 7756 (2000).
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett., 77, 3865 (1996).
A. D. Boese and N. C. Handy, J. Chem. Phys., 114, 5497 (2001).
Z. Xu, X. Chen, H. N. Kim, and J. Yoon, Chem. Soc. Rev., 39, 127 (2010).
P. Kaur, S. Kaur, and K. Singh, Inorg. Chem. Commun., 12, 978 (2009).
X. Lou, L. Zhang, J. Qin, and Z. Li, Chem. Commun., 5848 (2008).
Q. Zeng, P. Cai, Z. Li, J. Qin, and B. Z. Tang, Chem. Commun., 1094 (2008).
Z. Li, X. Lou, H. Yu, Z. Li, and J. Qin, Macromolecules, 41, 7433 (2008).
H. J. Kim, J. E. Park, M. G. Choi, S. Ahn, and S. K. Chang, Dyes Pigment., 84, 54 (2010).
J. R. Jadhav, C. H. Bae, and H. S. Kim, Tetrahedron Lett., 52, 1623 (2011).
L. Argyro, L. Euripides, F. Kostas, K. Alexandros, and L. Maria, Environ. Sci. Technol., 35, 1982 (2001).
J. N. Smith, K. C. Barsanti, H. R. Friedli, M. Ehn, K. Kulmala, D. R. Collins, J. H. Scheckman, B. J. Williams, and P. H. McMurry, Proc. Nat. Acad. Sci. USA, 107, 6634 (2010).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Gwon, SY., Angupillai, S., Son, YA. et al. The synthesis and spectral properties of a stimuli-responsive D-π-A charge transfer dye based on phenol donor and isophorone acceptor moiety. Fibers Polym 16, 1605–1610 (2015). https://doi.org/10.1007/s12221-015-5288-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12221-015-5288-8