Abstract
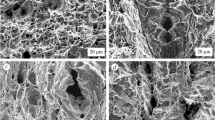
Various efforts have been made to improve the safety of high-pressure gas cylinders for hydrogen or natural gas with high strength steel liners. Metal liners with high tensile strength have a safety concern, particularly with hydrogen gas or hydrogen generating environments. The hydrogen can permeate into the liner material, and make the material brittle, causing hydrogen damage. This study investigated resistance to hydrogen damage for two kinds of 34CrMo4 steel with different strength levels. Hydrogen was charged with the electrochemical method, and the material strength was measured by the small punch testing technique. Hydrogen concentration of the specimen was also measured for every testing condition, with various charging periods. The specimens with high tensile strength absorbed more hydrogen than the regular tensile strength specimens. The absorbed hydrogen caused internal damage of intergranular cracking and blistering. Material ductility at failure decreased, as the hydrogen concentration of the specimen increased. But the hydrogen concentration had virtually no effect on the strength of the materials with hydrogen. These results confirm that the susceptibility to hydrogen damage of the high tensile strength materials is much higher than that of the materials with regular strength. If the metal liner of a hoop-wrapped cylinder vessel of type II has high tensile strength, general corrosion at the liner surface can cause a hydrogen rich environment, and the cylinder can suffer hydrogen damage and embrittlement. Therefore, controlling the strength level under an optimal level is critical for the safety of a cylinder made with 34CrMo4 steel.
Similar content being viewed by others
References
ANS ANSI/CSA NGV2-2000, Basic requirements for compressed Natural gas vehicle (NGV) fuel containers, CSA International, 8501E, Cleveland, OH (2000).
G. Bhattacharjee, S. Bhattacharya, S. Neogi and S. K. Das, CNG cylinder in a bus during gas filling–Lesson learned, Safety Sci., 48 (10) (2010) 1516–1519.
S. C. Kim, S. H. Lee and K. B. Yoon, Thermal characteristics during hydrogen fueling process of type IV cylinder, Int. J. of Hydrogen Energ., 35 (2010) 6830–6835.
O. Comond, D. Perreux, F. Thiebaud and M. Weber, Methodology to improve the lifetime of type III HP tank with a steel liner, Int. J. of Hydrogen Energ., 34 (13) (2009) 3077–3090.
B. Somerday, P. Sofronis and R. Jones, Effects of hydrogen on materials, Proceedings of the 2008 International Hydrogen Conference, ASM International (2009).
N. Eliaz, A. Shachar, B. Tal and D. Eliezer, Characteristics of hydrogen embrittlement, stress corrosion cracking and tempered martensite embrittlement in high-strength steels, Eng. Fail. Anal., 9 (2) (2002) 167–184.
Fractography Atlas of steel weldments, Japan Welding Society (1982).
B. Swieczko-Zurek, S. Sobieszczyk, J. Cwiek and A. Zielinski, Evaluation of susceptibility of high-strength steels to hydrogen delayed cracking, J. Achiev. Mater. Manuf. Eng., 18 (1–2) (2006) 243–246.
J. K. Yoon and K. B. Yoon, Suggestions for safety improvement of CNG bus based on accident and failure analysis, J. Korean Inst. Gas (KIGAS), 12 (2) (2008) 69–76.
K. B. Yoon, Failure analysis of high pressure vessel of type II for CNG bus, 5th International Conference on Engineering Failure Analysis, July 1-4 (2012).
K. Murakami, N. Yabe, H. Suzuki, K. Takai, Y. Hagihara and Y. Wada, Substitution of high-pressure charge by electrolysis charge and hydrogen environment embrittlement susceptibilities for Inconel 625 and SUS 316L, 2006 ASME Pressure Vessels and Piping Division Conference, PVP2006-ICPVT-11-93397 (2006) 1–8.
H. U. Seo, Y. W. Ma and K. B. Yoon, Evaluation of hydrogen embrittlement behavior in Inconel alloy 617 by small punch test, Trans. Korean Hydrogen and New Energy Society, 21 (4) (2010) 340–345.
D. J. Brookfield, W. Li, B. Rodgers, J. E. Mottershead, T. K. Hellen, J. Jarvis, R. Lohr, R. Howard-Hildige, A. Carlton and M. Whelan, Material properties from small specimens using the punch and bulge test, J. Strain Anal. Eng., 34 (6) (1999) 423–436.
Y. W. Ma and K. B. Yoon, Assessment of tensile strength using small punch test for transversely isotropic aluminum 2024 alloy produced by equal channel angular pressing, Mater. Sci. Eng. A, 527 (16–17) (2010) 3630–3638.
Y. W. Ma, J. W. Choi and K. B. Yoon, Change of anisotropic tensile strength due to amount of severe plastic deformation in Aluminum 2024 Alloy, Mater. Sci. Eng. A, 529 (2011) 1–8.
X. Mao and H. Takahashi, Development of a furtherminiaturized specimen of 3 mm diameter for TEM disk small punch tests, J. Nucl. Mater., 150 (1) (1987) 42–52.
J. D. Parker and J. D. James, Developments in a progressingtechnology, ASME PVP, 279 (1994) 167–172.
Y. W. Ma and K. B. Yoon, Assessment of power law creep constants of Gr91 steel using small punch creep tests, Fatigue Fract. Eng. M, 32 (12) (2009) 951–960.
ASTM Standard E8M-11, Standard test methods for tension testing of metallic materials, ASTM International, West Conshohocken, PA (2011) Doi: 10.1520/E0008-E0008M-11.
M. P. Manahan, A. S. Argon and O. K. Harling, The development of a miniaturized disk bend test for the determination of post-irradiation mechanical properties, J. Nucl. Mater., 103–104 (1981) 1545–1550.
NACE Standard TM0177-2005, Standard test method: Laboratory testing of metals for resistance to sulfide stress cracking and stress corrosion cracking in H2S environments, NACE Int., Houston, TX (2005).
S. Dey, A. M. Mandhyan, S. K. Sondhi and I. Chattoraj, Hydrogen entry into pipeline steel under freely corroding conditions in two corroding media, Corros Sci., 48 (9) (2006) 2676–2688.
N. N. Tun, H. S. Yang, J. M. Yu and K. B. Yoon, Creep crack growth analysis using Ct-parameter for internal circumferential and external axial surface cracks in a pressurized cylinder, J. Mech. Sci. Technol., 30 (12) (2016) 5447–5458.
Author information
Authors and Affiliations
Corresponding author
Additional information
Recommended by Editor Chongdu Cho
Seok Jeong Yoon received his M.S. in Chemical Engineering from KAIST (Korea Advanced Institute of Science and Technology). He is currently a Vice President in Samsung Institute of Safety & Environment in charge of utility and chemicals team. His interests are plant risk management and chemicals safety.
Ho Jun Lee received his M.S. in Mechanical Engineering from Chung-Ang University. He is currently a Project Researcher at KEIT (Korea Evaluation Institute of Industrial Technology). His research interests are hydrogen embrittlement and mechanical behavior of materials.
Kee Bong Yoon received his M.S. in Mechanical Engineering from KAIST and Ph.D. from Georgia Institute of Technology. He is currently a Professor at Chung-Ang University. His research interests are high temperature fracture mechanics, failure analysis and risk based management of facilities in semiconductor industry as well as the conventional power and process plants.
Young Wha Ma received his Ph.D. degree in Mechanical Engineering from Chung-Ang University, Korea in 2007. After that, he worked at Georgia Institute of Technology, U.S.A. as a postdoc. Dr. Ma is currently Principal R&PD Engineer at Doosan Heavy Industries & Construction Co. His research interests are application of high temperature fracture mechanics to residual life assessment of structural materials including anisotropic materials such as gas turbine blade.
Un Bong Baek received his Ph.D. degree in Mechanical Engineering from Kyungpook National University in 2001. He worked at Georgia Institute of Technology, U.S.A. as a postdoc during 2002-2003. Dr. Baek is currently Director of Center for Energy and Material Metrology of KRISS (Korea Research Institute of Standards and Science). His research interest is mechanical behavior of materials under high pressure hydrogen environment.
Rights and permissions
About this article
Cite this article
Yoon, S.J., Lee, H.J., Yoon, K.B. et al. Hydrogen damage in 34CrMo4 pressure vessel steel with high tensile strength. J Mech Sci Technol 32, 637–646 (2018). https://doi.org/10.1007/s12206-018-0111-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12206-018-0111-7