Abstract
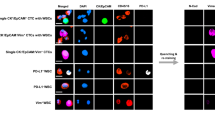
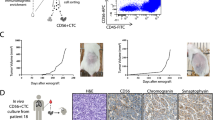
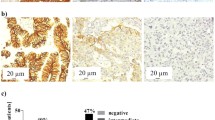
Circulating tumour cells (CTCs) were enriched in the peripheral blood of four patients with Stage I non-small cell lung cancer (NSCLC). Octamer-binding transcription factor-4 positive (OCT4+) and negative (OCT4−) CTCs were identified and captured by interphase fluorescence in situ hybridisation (iFISH). Single cell whole exome sequencing (WES) was performed and the corresponding bioinformatics data were analysed. OCT4+ cells were successfully detected in peripheral blood collected from all four Stage I lung cancer patients. Moreover, the tumour mutational burden (TMB) values observed for OCT4+ samples from the same patients were slightly smaller than those of the OCT4− samples; the difference was not statistically significant (P> 0.05). Thirteen and six characteristic mutations were found in negative samples and positive samples, respectively. The findings indicate that this methodology provides a potential diagnostic index for the early detection of NSCLC.
Similar content being viewed by others
References
SIEGEL R L, MILLER K D, JEMAL A. Cancer statistics, 2019 [J]. CA: A Cancer Journal for Clinicians, 2019, 69(1): 7–34.
HERBST R S, MORGENSZTERN D, BOSHOFF C. The biology and management of non-small cell lung cancer [J]. Nature, 2018, 553: 446–454.
HIRSCH F R, SUDA K, WIENS J, et al. New and emerging targeted treatments in advanced non-small-cell lung cancer [J]. The Lancet, 2016, 388(10048): 1012–1024.
LUNDIN A, DRISCOLL B. Lung cancer stem cells: Progress and prospects [J]. Cancer Letters, 2013, 338(1): 89–93.
KLONISCH T, WIECHEC E, HOMBACH-KLONISCH S, et al. Cancer stem cell markers in common cancers: Therapeutic implications [J]. Trends in Molecular Medicine, 2008, 14(10): 450–460.
CHAMBERS I, COLBY D, ROBERTSON M, et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells [J]. Cell, 2003, 113(5): 643–655.
MITSUI K, TOKUZAWA Y, ITOH H, et al. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells [J]. Cell, 2003, 113(5): 631–642.
VAN SCHAIJIK B, DAVIS P F, WICKREMESEKERA A C, et al. Subcellular localisation of the stem cell markers OCT4, SOX2, NANOG, KLF4 and c-MYC in cancer: A review [J]. Journal of Clinical Pathology, 2018, 71: 88–91.
VILLODRE E S, KIPPER F C, PEREIRA M B, et al. Roles of OCT4 in tumorigenesis, cancer therapy resistance and prognosis [J]. Cancer Treatment Reviews, 2016, 51: 1–9.
ZHANG X Y, DONG Q G, HUANG J S, et al. The expression of stem cell-related indicators as a prognostic factor in human lung adenocarcinoma [J]. Journal of Surgical Oncology, 2010, 102(7): 856–862.
ZHANG X Y, ZHANG Y W, XU J L, et al. Antigen presentation of the Oct4 and Sox2 peptides by CD154-activated B lymphocytes enhances the killing effect of cytotoxic T lymphocytes on tumor stem-like cells derived from cisplatin-resistant lung cancer cells [J]. Journal of Cancer, 2018, 9(2): 367–374.
ZHANG X Y, HU F, LI C H, et al. OCT4&SOX2-specific cytotoxic T lymphocytes plus programmed cell death protein 1 inhibitor presented with synergistic effect on killing lung cancer stem-like cells in vitro and treating drug-resistant lung cancer mice in vivo [J]. Journal of Cellular Physiology, 2019, 234(5): 6758–6768.
LI R, HUANG J S, MA M L, et al. Two-stage induced differentiation of OCT4+/Nanog+ stem-like cells in lung adenocarcinoma [J]. Oncotarget, 2016, 7(42): 68360–68370.
MATIKAS A, SYRIGOS K N, AGELAKI S. Circulating biomarkers in non-small-cell lung cancer: Current status and future challenges [J]. Clinical Lung Cancer, 2016, 17(6): 507–516.
PAOLETTI C, HAYES D F. Circulating tumor cells [M]//STEARNS V. Advances in Experiment Medicine and Biology. Cham, Switzerland: Springer, 2016: 235–258.
CHUDASAMA D, BARR J, BEESON J, et al. Detection of circulating tumour cells and survival of patients with non-small cell lung cancer [J]. Anticancer Research, 2017, 37: 169–173.
HEEKE S, MOGRABI B, ALIX-PANABIÈRES C, et al. Never travel alone: The crosstalk of circulating tumor cells and the blood microenvironment [J]. Cells, 2019, 8(7): 714.
PRABAVATHY D, RAMADOSS N. Heterogeneity of small cell lung cancer stem cells [M]//BIRBRAIR A. Stem cells heterogeneity in cancer. Cham, Switzerland: Springer, 2019: 41–57.
NEL I, JEHN U, GAULER T, et al. Individual profiling of circulating tumor cell composition in patients with non-small cell lung cancer receiving platinum based treatment [J]. Translational Lung Cancer Research, 2014, 3(2): 100–106.
MOHIUDDIN I S, WEI S J, KANG M H. Role of OCT4 in cancer stem-like cells and chemotherapy resistance [J]. BBA-Molecular Basis of Disease, 2020, 1866(4): 165432.
JEN J, TANG Y A, LU Y H, et al. Oct4 transcriptionally regulates the expression of long non-coding RNAs NEAT1 and MALAT1 to promote lung cancer progression [J]. Molecular Cancer, 2017, 16(1): 104.
KOBAYASHI I, TAKAHASHI F, NURWIDYA F, et al. Oct4 plays a crucial role in the maintenance of gefitinib-resistant lung cancer stem cells [J]. Biochemical and Biophysical Research Communications, 2016, 473(1): 125–132.
LI S, CHEN Q, LI H, et al. Mesenchymal circulating tumor cells (CTCs) and OCT4 mRNA expression in CTCs for prognosis prediction in patients with non-small-cell lung cancer [J]. Clinical and Translational Oncology, 2017, 19: 1147–1153.
RIZVI N A, HELLMANN M D, SNYDER A, et al. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer [J]. Science, 2015, 348(6230): 124–128.
GANDARA D R, PAUL S M, KOWANETZ M, et al. Blood-based tumor mutational burden as a predictor of clinical benefit in non-small-cell lung cancer patients treated with atezolizumab [J]. Nature Medicine, 2018, 24: 1441–1448.
GUO J C, LI C Q, WANG Q Y, et al. Protein-coding genes combined with long non-coding RNAs predict prognosis in esophageal squamous cell carcinoma patients as a novel clinical multi-dimensional signature [J]. Molecular BioSystems, 2016, 12(11): 3467–3477.
MATSUBARA J, HONDA K, ONO M, et al. Identification of adipophilin as a potential plasma biomarker for colorectal cancer using label-free quantitative mass spectrometry and protein microarray [J]. Cancer Epidemiology, Biomarkers & Prevention, 2011, 20(10): 2195–2203.
MORRISSEY J J, MOBLEY J, FIGENSHAU R S, et al. Urine Aquaporin 1 and Perilipin 2 differentiate renal carcinomas from other imaged renal masses and bladder and prostate cancer [J]. Mayo Clinic Proceedings, 2015, 90(1): 35–42.
AMBROSIO M R, PICCALUGA P P, PONZONI M, et al. The alteration of lipid metabolism in Burkitt lymphoma identifies a novel marker: Adipophilin [J]. PLoS One, 2012, 7(8): e44315.
ZHANG X D, LI W, ZHANG N, et al. Identification of adipophilin as a potential diagnostic tumor marker for lung adenocarcinoma [J]. International Journal of Clinical and Experimental Medicine, 2014, 7(4): 1190–1196.
CONTE M, FRANCESCHI C, SANDRI M, et al. Perilipin 2 and age-related metabolic diseases: A new perspective [J]. Trends in Endocrinology & Metabolism, 2016, 27(12): 893–903.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Foundation item
the National Natural Science Foundation of China (No. 81773273)
Rights and permissions
About this article
Cite this article
Yan, B., Fu, S., Chang, Y. et al. Mutational Analysis of OCT4+ and OCT4− Circulating Tumour Cells by Single Cell Whole Exome Sequencing in Stage I Non-Small Cell Lung Cancer Patients. J. Shanghai Jiaotong Univ. (Sci.) 26, 40–46 (2021). https://doi.org/10.1007/s12204-021-2258-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12204-021-2258-8
Key words
- non-small cell lung cancer (NSCLC)
- octamer-binding transcription factor-4 (OCT4)
- circulating tumour cells (CTCs)
- whole exome sequencing (WES)