Abstract
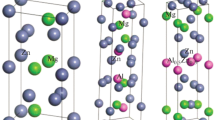
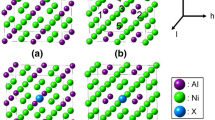
The energetic and electronic structures of precipitates on the Cu-rich side of Cu-Ni-Si alloys were investigated by using the first-principle calculations based on plane-wave pseudopotential method. The negative formation heats and the cohesive energies of these precipitates were estimated with electronic structure calculations, and their structural stability was also analyzed. The results show that δ-Ni2Si, γ-Ni5Si2 and β-Ni3Si precipitates all have great alloying ability and structural stability, which, after comparing their density of states (DOS), is found attributed to the pseudogap effect near the Fermi level (E F) and strong hybridization between the Ni-3d and Si-3p states. Compared with the other two precipitates, the δ-Ni2Si precipitate has the greatest structural stability, which is resulted from its lower DOS at E F and the main bonding peaks slightly moving to the low energy region.
Similar content being viewed by others
References
Robertson W D, Grenier E G, Nole V F. The structure and associated properties of an age hardening copper alloy [J]. Transactions of the Metallurgical Society of AIME, 1961, 221: 503–512.
Lockyer S A, Noble F W. Precipitate structure in a Cu-Ni-Si alloy [J]. Journal of Materials Science, 1994, 29(1): 218–226.
Fujiwara H, Sato T, Kamio A. Effect of alloy composition on precipitation behavior in Cu-Ni-Si alloys [J]. Nippon Kinzoku Gakkaishi/Journal of the Japan Institute of Metals, 1998, 62(4): 30–35.
Huang F, Ma J, Ning H, et al. Precipitation in Cu-Ni-Si-Zn alloy for lead frame [J]. Materials Letters, 2003, 57(13–14): 2135–2139.
Zhao D M, Dong Q M, Liu P, et al. Structure and strength of the age hardened Cu-Ni-Si alloy [J]. Materials Chemistry and Physics, 2003, 79(1): 81–86.
Suzuki S, Shibutani N, Mimura K, et al. Improvement in strength and electrical conductivity of Cu-Ni-Si alloys by aging and cold rolling [J]. Journal of Alloys and Compounds, 2006, 417(1–2): 116–120.
Oelson W, von Samson-Himmelstjerna H O. The enthalpy of formation of nickel-silicon alloys and liquid [J]. Mitt Kaiser-Wilhelm Inst Eisenforch Düsseldorf, 1936, 18: 131–133.
Meschel S V, Kleppa O J. Standard enthalpies of formation of some 3d transition metal silicides by-high temperature direct synthesis calorimetry [J]. Journal of Alloys Compounds, 1998, 267(1): 128–135.
Lindhòlm M, Sundman B. A thermodynamic evaluation of the nickel-silicon system [J]. Metallurgical and Materials Transactions A: Physical Metallurgy and Materials Science, 1996, 27: 2897–2903.
Acker J, Bohmhammel K. Optimization of thermodynamic data of the Ni-Si system [J]. Thermochimica Acta, 1999, 337(1–2): 187–193.
Miettinen J. Thermodynamic description of the Cu-Ni-Si system in the copper-rich corner above 700° [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 2005, 29(3): 212–221.
Tokunaga T, Nishio K, Ohtani H, et al. Thermodynamic assessment of the Ni-Si system by incorporating ab initio energetic calculations into the CALPHAD approach [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 2003, 27(2): 161–168.
Derlet P M, Andersen S J, Marioara C D, et al. A first-principles study of the β″-phase in Al-Mg-Si alloys [J]. Journal of Physics: Condensed Matter, 2002, 14(15): 4011–4024.
Frøseth A G, Hoier R, Derlet P M, et al. Bonding in MgSi and Al-Mg-Si compounds relevant to Al-Mg-Si alloys [J]. Condens. Matter Physical Review B: Condensed Matter, 2003, 67: 1–11.
Ravi C, Wolverton C. First-principles study of crystal structure and stability of Al-Mg-Si-(Cu) precipitates [J]. Acta Materialia, 2004, 52(12): 4213–4227.
Toman K. The structure of Ni2Si [J]. Acta Crystallographica Section B: Structural Science, 1952, 5: 329–331.
Frank K, Schubert K. Crystal structure of. Ni31Si12 [J]. Acta Crystallographica Section B: Structural Science, 1971, B27(5): 916–920.
Ochiai S, Mishima Y, Suzuki T. Lattice parameter data of Ni, Ni3Al and Ni3Ga solid solutions with additions of transition and B-subgroup elements [J]. Acta Metallurgica, 1985, 33(6): 1161–1169.
Segall M D, Lindan P L D, Probert M J, et al. First-principles simulation: Ideas, illustrations and the CASTEP code [J]. Journal of Physics: Condensed Matter, 2002, 14(11): 2717–2743.
Kohn W, Sham L J. Self-consistent equations including exchange and correlation effects [J]. Physical Review Letters, 1965, 140(4A): 1133–1138.
Payne M C, Teter M P, Allan D C, et al. Iterative minimization techniques for ab initio total-energy calculations: Molecular dynamics and conjugate gradients [J]. Reviews of Modern Physics, 1992, 64(4): 1045–1094.
Perdew J P. Accurate density functional for the energy real-space cut off of the gradient expansion for the exchange hole [J]. Physical Review Letters, 1985, 55(16): 1665–1668.
Perdew J P, Burke K, Ernzerhof M. Generalized gradient approximation made simple [J]. Physical Review Letter, 1996, 77(18): 3865–3868.
Ceperley D M, Alder B J. Ground state of the electron gas by a stochastic method [J]. Physical Review Letter, 1980, 45(7): 566–569.
Pack J D, Monkhorst H J. Special points for Brillouin-zone integrations—A reply [J]. Physical Review B: Condensed Matter, 1977, 16(4–15): 1748–1749.
Vanderbilt D. Soft self-consistent pseudopotentials in a generalized eigen value formalism [J]. Physical Review B: Condensed Matter, 1990, 43(11): 7892–7895.
Pfrommer B G, Cote M, Louie S G, et al. Relaxation of crystals with the quasi-Newton method [J]. Journal of Computational Physics, 1997, 131(1): 233–240.
Faraoun H I, Zhang Y D, Esling C, et al. Crystalline, electronic, and magnetic structures of θ-Fe3C, χ-Fe5C2, and η-Fe2C from first principle calculation [J]. Journal of Applied Physics, 2006, 99(9): 1–8.
Zubov V I, Tretiakov N P, Teixeira J N, et al. Calculations of the thermal expansion, cohesive energy and thermodynamic stability of a Van der Waals crystal-fullerene C60 [J]. Physics Letters A, 1994, 194(3): 223–227.
Song Y, Guo Z X, Yang R, et al. A first principles study of the theoretical strength and bulk modulus of hcp metals [J]. Acta Materialia, 2001, 49(8): 1647–1654.
Iotova D, Kioussis N, Lim S P. Electronic structure and elastic properties of the Ni3X (X=Mn, Al, Ga, Si, Ge) intermetallic [J]. Physical Review B: Condensed Matter, 1996, 54(20): 14413–14421.
Xu J X, Min B I, Freeman A J, et al. Phase stability and magnetism of Ni3Al [J]. Physical Review B: Condensed Matter, 1990, 41(8): 5010–5016.
Carlsson A E, Meschter P J. Ab initio calculations in intermetallic compounds-principles and practice [M]. Chichester: John Wiley & Sons, 1995: 55–76.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: the National Natural Science Foundation of China (No. 50571035), and the National High Technology Research and Development Project (863) of China (No. 2006AA032528)
Rights and permissions
About this article
Cite this article
Long, Yq., Liu, P., Liu, Y. et al. First-principle investigation of the structural stability and electronic property of precipitates on the Cu-rich side of Cu-Ni-Si alloys. J. Shanghai Jiaotong Univ. (Sci.) 16, 266–271 (2011). https://doi.org/10.1007/s12204-011-1141-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12204-011-1141-4