Abstract
Background
Diabetes mellitus is characterized by hyperglycemia which displays insufficiency or resistance to insulin. One of the complications of diabetes is the increased risk of fracture and the impairment of bone repair and regulation. There have been evidences from previous studies that mesenchymal stem cells (MSCs) from bone marrow promote cartilage and callous formation. In addition, IL-10, an anti-inflammatory cytokine, has been observed to relieve inflammation-related complications in diabetes.
Methods
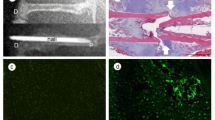
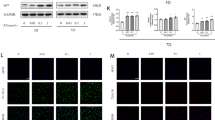
In this study, the role of IL-10-overexpressing bone marrow-derived MSCs (BM-MSCs) was examined in the diabetic mice model with femur fracture. MSCs were isolated from the BALB/c mice and IL-10 over expression was conducted with lentivirus transduction. The streptozotocin (STZ)-induced diabetes model with femoral fracture was established. BM-MSCs with IL-10 over expression were transplanted into the fracture area. The expressions of inflammatory factors IL-6, TNF-α and INF-γ were examined by qPCR and immunoblot; the biomechanical strength of the fracture site of the mice was examined and evaluated.
Results
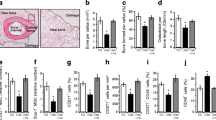
Data showed that IL-10 overexpressed BM-MSCs transplantation decreased inflammatory response, promoted bone formation, and increased the strength of the fracture site in STZ-induced diabetic mice with femoral fracture.
Conclusion
IL-10 overexpressed BM-MSCs transplantation accelerated fracture repair in STZ-induced diabetic mice, which in turn provides potential clinical application prospects.





Similar content being viewed by others
References
Alharbi, M. A., C. Zhang, C. Lu, T. N. Milovanova, L. Yi, J. D. Ryu, H. Jiao, G. Dong, J. P. O’Connor, and D. T. Graves. FOXO1 deletion reverses the effect of diabetic-induced impaired fracture healing. Diabetes 67:2682–2694, 2018. https://doi.org/10.2337/db18-0340.
Blaber, E. A., N. Dvorochkin, C. Lee, J. S. Alwood, R. Yousuf, P. Pianetta, R. K. Globus, B. P. Burns, and E. A. Almeida. Microgravity induces pelvic bone loss through osteoclastic activity, osteocytic osteolysis, and osteoblastic cell cycle inhibition by CDKN1a/p21. PLoS ONE 8:e61372, 2013. https://doi.org/10.1371/journal.pone.0061372.
Brown, M. L., K. Yukata, C. W. Farnsworth, D. G. Chen, H. Awad, M. J. Hilton, R. J. O’Keefe, L. Xing, R. A. Mooney, and M. J. Zuscik. Delayed fracture healing and increased callus adiposity in a C57BL/6J murine model of obesity-associated type 2 diabetes mellitus. PLoS ONE 9:e99656, 2014. https://doi.org/10.1371/journal.pone.0099656.
Cao, J. J. Effects of obesity on bone metabolism. J Orthop Surg Res 6:30, 2011. https://doi.org/10.1186/1749-799X-6-30.
Choi, J. J., S. A. Yoo, S. J. Park, Y. J. Kang, W. U. Kim, I. H. Oh, and C. S. Cho. Mesenchymal stem cells overexpressing interleukin-10 attenuate collagen-induced arthritis in mice. Clin Exp Immunol 153:269–276, 2008. https://doi.org/10.1111/j.1365-2249.2008.03683.x.
Gao, F., T. R. Lv, J. C. Zhou, and X. D. Qin. Effects of obesity on the healing of bone fracture in mice. J Orthop Surg Res 13:145, 2018. https://doi.org/10.1186/s13018-018-0837-7.
Gong, Z., and R. H. Muzumdar. Pancreatic function, type 2 diabetes, and metabolism in aging. Int J Endocrinol 2012:320482, 2012. https://doi.org/10.1155/2012/320482.
Granero-Molto, F., J. A. Weis, M. I. Miga, B. Landis, T. J. Myers, L. O’Rear, L. Longobardi, E. D. Jansen, D. P. Mortlock, and A. Spagnoli. Regenerative effects of transplanted mesenchymal stem cells in fracture healing. Stem Cells 27:1887–1898, 2009. https://doi.org/10.1002/stem.103.
Hamann, C., C. Goettsch, J. Mettelsiefen, V. Henkenjohann, M. Rauner, U. Hempel, R. Bernhardt, N. Fratzl-Zelman, P. Roschger, S. Rammelt, K. P. Gunther, and L. C. Hofbauer. Delayed bone regeneration and low bone mass in a rat model of insulin-resistant type 2 diabetes mellitus is due to impaired osteoblast function. Am J Physiol Endocrinol Metab 301:E1220–1228, 2011. https://doi.org/10.1152/ajpendo.00378.2011.
Imam, M. A., J. Holton, L. Ernstbrunner, W. Pepke, F. Grubhofer, A. Narvani, and M. Snow. A systematic review of the clinical applications and complications of bone marrow aspirate concentrate in management of bone defects and nonunions. Int Orthop 41:2213–2220, 2017. https://doi.org/10.1007/s00264-017-3597-9.
Jiang, Y., B. N. Jahagirdar, R. L. Reinhardt, R. E. Schwartz, C. D. Keene, X. R. Ortiz-Gonzalez, M. Reyes, T. Lenvik, T. Lund, M. Blackstad, J. Du, S. Aldrich, A. Lisberg, W. C. Low, D. A. Largaespada, and C. M. Verfaillie. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418:41–49, 2002. https://doi.org/10.1038/nature00870.
Knight, M. N., and K. D. Hankenson. Mesenchymal stem cells in bone regeneration. Adv Wound Care (New Rochelle) 2:306–316, 2013. https://doi.org/10.1089/wound.2012.0420.
Krishnamurthy, P., J. Rajasingh, E. Lambers, G. Qin, D. W. Losordo, and R. Kishore. IL-10 inhibits inflammation and attenuates left ventricular remodeling after myocardial infarction via activation of STAT3 and suppression of HuR. Circ Res 104:e9–18, 2009. https://doi.org/10.1161/CIRCRESAHA.108.188243.
Lecka-Czernik, B., L. A. Stechschulte, P. J. Czernik, and A. R. Dowling. High bone mass in adult mice with diet-induced obesity results from a combination of initial increase in bone mass followed by attenuation in bone formation; implications for high bone mass and decreased bone quality in obesity. Mol Cell Endocrinol 410:35–41, 2015. https://doi.org/10.1016/j.mce.2015.01.001.
Lotfy, M., J. Adeghate, H. Kalasz, J. Singh, and E. Adeghate. Chronic complications of diabetes mellitus: a mini review. Curr Diabetes Rev 13:3–10, 2017. https://doi.org/10.2174/1573399812666151016101622.
Mao, C., and H. Yan. Roles of elevated intravitreal IL-1beta and IL-10 levels in proliferative diabetic retinopathy. Indian J Ophthalmol 62:699–701, 2014. https://doi.org/10.4103/0301-4738.136220.
Moseley, K. F. Type 2 diabetes and bone fractures. Curr Opin Endocrinol Diabetes Obes 19:128–135, 2012. https://doi.org/10.1097/MED.0b013e328350a6e1.
Pantalone, K. M., T. M. Hobbs, B. J. Wells, S. X. Kong, M. W. Kattan, J. Bouchard, C. Yu, B. Sakurada, A. Milinovich, W. Weng, J. M. Bauman, and R. S. Zimmerman. Clinical characteristics, complications, comorbidities and treatment patterns among patients with type 2 diabetes mellitus in a large integrated health system. BMJ Open Diabetes Res Care 3:e000093, 2015. https://doi.org/10.1136/bmjdrc-2015-000093.
Papatheodorou, K., M. Banach, E. Bekiari, M. Rizzo, and M. Edmonds. Complications of diabetes 2017. J Diabetes Res 2018:3086167, 2018. https://doi.org/10.1155/2018/3086167.
Schwarz, S., I. Mrosewski, S. Silawal, and G. Schulze-Tanzil. The interrelation of osteoarthritis and diabetes mellitus: considering the potential role of interleukin-10 and in vitro models for further analysis. Inflamm Res 67:285–300, 2018. https://doi.org/10.1007/s00011-017-1121-8.
Shu, L., E. Beier, T. Sheu, H. Zhang, M. J. Zuscik, E. J. Puzas, B. F. Boyce, R. A. Mooney, and L. Xing. High-fat diet causes bone loss in young mice by promoting osteoclastogenesis through alteration of the bone marrow environment. Calcif Tissue Int 96:313–323, 2015. https://doi.org/10.1007/s00223-015-9954-z.
Soleimani, M., and S. Nadri. A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. Nat Protoc 4:102–106, 2009. https://doi.org/10.1038/nprot.2008.221.
Stabley, J. N., R. D. Prisby, B. J. Behnke, and M. D. Delp. Type 2 diabetes alters bone and marrow blood flow and vascular control mechanisms in the ZDF rat. J Endocrinol 225:47–58, 2015. https://doi.org/10.1530/JOE-14-0514.
Tesch, G. H., and T. J. Allen. Rodent models of streptozotocin-induced diabetic nephropathy. Nephrology (Carlton) 12:261–266, 2007. https://doi.org/10.1111/j.1440-1797.2007.00796.x.
Wang, Y., D. W. Malcolm, and D. S. W. Benoit. Controlled and sustained delivery of siRNA/NPs from hydrogels expedites bone fracture healing. Biomaterials 139:127–138, 2017. https://doi.org/10.1016/j.biomaterials.2017.06.001.
Wu, K. K., and Y. Huan. Streptozotocin-induced diabetic models in mice and rats. Curr Protoc Pharmacol 2008. https://doi.org/10.1002/0471141755.ph0547s40.
Yan, W., and X. Li. Impact of diabetes and its treatments on skeletal diseases. Front Med 7:81–90, 2013. https://doi.org/10.1007/s11684-013-0243-9.
Yao, Y. D., T. M. Sun, S. Y. Huang, S. Dou, L. Lin, J. N. Chen, J. B. Ruan, C. Q. Mao, F. Y. Yu, M. S. Zeng, J. Y. Zang, Q. Liu, F. X. Su, P. Zhang, J. Lieberman, J. Wang, and E. Song. Targeted delivery of PLK1-siRNA by ScFv suppresses Her2+ breast cancer growth and metastasis. Sci Transl Med 4:130, 2012. https://doi.org/10.1126/scitranslmed.3003601.
Yuan, N., H. F. Zhang, Q. Wei, P. Wang, and W. Y. Guo. Expression of CD4+CD25+Foxp3+ regulatory T cells, interleukin 10 and transforming growth factor beta in newly diagnosed type 2 diabetic patients. Exp Clin Endocrinol Diabetes 126:96–101, 2018. https://doi.org/10.1055/s-0043-113454.
Acknowledgments
This work was supported by the Natural Science Foundation of Hainan Province, China (819MS146) and the Seed Fund Program of Shanghai University of Medicine & Health Sciences (No. 2017N0612).
Conflict of interest
Keze Cui, Yuanliang Chen, Haibo Zhong, Nan Wang, Lihui Zhou and Fusong Jiang declare that they have no conflict of interest.
Ethical Approval
No human studies were carried out by the authors for this article. All animal studies were carried out in accordance with the guideline issued by Shanghai Jiao Tong University Affiliated Sixth People’s Hospital and approved by Shanghai Jiao Tong University Affiliated Sixth People’s Hospital.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Edward Sander oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cui, K., Chen, Y., Zhong, H. et al. Transplantation of IL-10-Overexpressing Bone Marrow-Derived Mesenchymal Stem Cells Ameliorates Diabetic-Induced Impaired Fracture Healing in Mice. Cel. Mol. Bioeng. 13, 155–163 (2020). https://doi.org/10.1007/s12195-019-00608-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-019-00608-w