Abstract
Introduction
Notch signaling is amongst the key intrinsic mechanisms regulating satellite cell fate, promoting the transition of activated satellite cells to highly proliferative myogenic progenitor cells and preventing their premature differentiation. Although much is known about the biochemical milieu that drives myogenic progression, less is known about the spatial cues providing spatiotemporal control of skeletal muscle repair in the context of Notch signaling.
Methods
Using a murine injury model, we quantified in vivo biophysical changes that occur within the skeletal muscle during regeneration. Employing tunable poly(ethylene glycol)—based hydrogel substrates, we modeled the measured changes in bulk stiffness in the context of Notch ligand signaling, which are present in the regenerative milieu at the time of injury.
Results
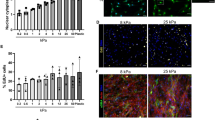
Following injury, there is a transient increase in the bulk stiffness of the tibialis anterior muscle that may be explained in part by changes in extracellular matrix deposition. When presented to primary myoblasts, Jagged-1, Jagged-2, and Dll1 in a tethered format elicited greater degrees of Notch activity compared to their soluble form. Only tethered Jagged-1 effects were tuned by substrate stiffness, with the greatest Notch activation observed on stiff hydrogels matching the stiffness of regenerating muscle. When exposed to tethered Jagged-1 on stiff hydrogels, fewer primary myoblasts expressed myogenin, and pharmacological inhibitor studies suggest this effect is Notch and RhoA dependent.
Conclusion
Our study proposes that tethered Jagged-1 presented in the context of transient tissue stiffening serves to tune Notch activity in myogenic progenitors during skeletal muscle repair and delay differentiation.




Similar content being viewed by others
Abbreviations
- bFGF:
-
Basic fibroblast growth factor
- Dll1:
-
Delta-like 1
- DM:
-
Differentiation media
- ECM:
-
Extracellular matrix
- GM:
-
Growth media
- IgG1:
-
Immunoglobulin G subtype 1
- Jag1:
-
Jagged-1
- Jag2:
-
Jagged-2
- MyoG:
-
Myogenin
- NICD:
-
Notch intracellular domain
- PEG:
-
Polyethylene glycol
- PBS:
-
Phosphate buffered saline
- ROCK:
-
Rho-associated, coiled-coil containing protein kinase
- TBST:
-
Tris-buffered saline plus Tween-20
References
Bjornson, C. R. R., T. H. Cheung, L. Liu, P. V. Tripathi, K. M. Steeper, and T. A. Rando. Notch signaling is necessary to maintain quiescence in adult muscle stem cells. Stem Cells 30:232–242, 2012.
Blau, H. M., and C. Webster. Isolation and characterization of human muscle cells. Proc. Natl. Acad. Sci. USA. 78:5623–5627, 1981.
Buas, M. F., and T. Kadesch. Regulation of skeletal myogenesis by Notch. Exp. Cell Res. 316:3028–3033, 2010.
Calve, S., J. Isaac, J. P. Gumucio, and C. L. Mendias. Hyaluronic acid, HAS1, and HAS2 are significantly upregulated during muscle hypertrophy. Am. J. Physiol. Cell Physiol. 303:C577–C588, 2012.
Conboy, I. M., and T. A. Rando. The regulation of Notch signaling controls satellite cell activation and cell fate determination in postnatal myogenesis. Dev. Cell 3:397–409, 2002.
Cordey, M., M. Limacher, S. Kobel, V. Taylor, and M. P. Lutolf. Enhancing the reliability and throughput of neurosphere culture on hydrogel microwell arrays. Stem Cells 26:2586–2594, 2008.
Cosgrove, B. D., P. M. Gilbert, E. Porpiglia, F. Mourkioti, S. P. Lee, S. Y. Corbel, M. E. Llewellyn, S. L. Delp, and H. M. Blau. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat. Med. 20:255–264, 2014.
Davoudi, S., and P. M. Gilbert. Optimization of satellite cell culture through biomaterials. Methods. Mol. Biol. 1556:329–341, 2017.
Engler, A. J., M. A. Griffin, S. Sen, C. G. Bönnemann, H. L. Sweeney, and D. E. Discher. Myotubes differentiate optimally on substrates with tissue-like stiffness: pathological implications for soft or stiff microenvironments. J. Cell Biol. 166:877–887, 2004.
Engler, A. J., S. Sen, H. L. Sweeney, and D. E. Discher. Matrix elasticity directs stem cell lineage specification. Cell 126:677–689, 2006.
Faux, C. H., A. M. Turnley, R. Epa, R. Cappai, and P. F. Bartlett. Interactions between growth factors and Notch regulate neuronal differentiation. J Neurosci. 21:5587–5596, 2001.
Gerdin, B., and R. Hällgren. Dynamic role of hyaluronan (HYA) in connective tissue activation and inflammation. J. Intern. Med. 242:49–55, 1997.
Gilbert, P. M., K. L. Havenstrite, K. E. G. Magnusson, A. Sacco, N. A. Leonardi, P. Kraft, N. K. Nguyen, S. Thrun, M. P. Lutolf, and H. M. Blau. Substrate elasticity regulates skeletal muscle stem cell self-renewal in culture. Science 329:1078–1081, 2010.
Gordon, W. R., D. Vardar-Ulu, G. Histen, C. Sanchez-Irizarry, J. C. Aster, and S. C. Blacklow. Structural basis for autoinhibition of Notch. Nat Struct Mol Biol 14:295–300, 2007.
Gordon, W. R., B. Zimmerman, L. He, L. J. Miles, J. Huang, K. Tiyanont, D. G. McArthur, J. C. Aster, N. Perrimon, J. J. Loparo, and S. C. Blacklow. Mechanical allostery: evidence for a force requirement in the proteolytic activation of Notch. Dev. Cell 33:729–736, 2015.
Griffith, L. G., and M. A. Swartz. Capturing complex 3D tissue physiology in vitro. Nat Rev Mol Cell Biol 7:211–224, 2006.
Günther, S., J. Kim, S. Kostin, C. Lepper, C. M. Fan, and T. Braun. Myf5-positive satellite cells contribute to Pax7-dependent long-term maintenance of adult muscle stem cells. Cell Stem Cell 13:590–601, 2013.
Hisada, Y., M. Yasunaga, S. Hanaoka, S. Saijou, T. Sugino, A. Tsuji, T. Saga, K. Tsumoto, S. Manabe, J. Kuroda, J. Kuratsu, and Y. Matsumura. Discovery of an uncovered region in fibrin clots and its clinical significance. Sci. Rep. 3:2604, 2013.
Jahnsen, E. D., A. Trindade, H. C. Zaun, S. Lehoux, A. Duarte, and E. A. V. Jones. Notch1 is pan-endothelial at the onset of flow and regulated by flow. PLoS ONE 10:1–14, 2015.
Jiang, W. R., G. Cady, M. M. Hossain, Q. Q. Huang, X. Wang, and J. P. Jin. Mechanoregulation of h2-calponin gene expression and the role of notch signaling. J. Biol. Chem. 289:1617–1628, 2014.
Kondoh, K., K. Sunadome, and E. Nishida. Notch signaling suppresses p38 MAPK activity via induction of MKP-1 in myogenesis. J. Biol. Chem. 282:3058–3065, 2007.
Kuhl, P. R., and L. G. Griffith-Cima. Tethered epidermal growth factor as a paradigm for growth factor–induced stimulation from the solid phase. Nat. Med. 2:1022–1027, 1996.
Lepper, C., S. J. Conway, and C.-M. Fan. Adult satellite cells and embryonic muscle progenitors have distinct genetic requirements. Nature 460:627–631, 2009.
Lutolf, M. P., R. Doyonnas, K. Havenstrite, K. Koleckar, and H. M. Blau. Perturbation of single hematopoietic stem cell fates in artificial niches. Integr Biol 1:59–69, 2009.
Lutolf, M. P., and J. A. Hubbell. Synthesis and physicochemical characterization of end-linked poly(ethylene glycol)-co-peptide hydrogels formed by Michael-type addition. Biomacromolecules 4:713–722, 2003.
Mauro, A., and W. R. Adams. The structure of the sarcolemma of the frog skeletal muscle fiber. J. Biophys. Biochem. Cytol. 10:177–185, 1961.
Morrissey, J. B., R. Y. Cheng, S. Davoudi, and P. M. Gilbert. Biomechanical origins of muscle stem cell signal transduction. J. Mol. Biol. 428:1441–1454, 2015.
Quarta, M., J. O. Brett, R. DiMarco, A. De Morree, S. C. Boutet, R. Chacon, M. C. Gibbons, V. A. Garcia, J. Su, J. B. Shrager, S. Heilshorn, and T. A. Rando. An artificial niche preserves the quiescence of muscle stem cells and enhances their therapeutic efficacy. Nat. Biotechnol. 34:752–759, 2016.
Seo, D., K. M. Southard, J. Kim, J. Cheon, Z. J. Gartner, Y. Jun, D. Seo, K. M. Southard, J. Kim, H. J. Lee, J. Farlow, and J. Lee. A mechanogenetic toolkit for interrogating cell signaling in space and time. Cell 165:1507–1518, 2016.
Tierney, M. T., and A. Sacco. Inducing and Evaluating Skeletal Muscle Injury by Notexin and Barium Chloride. Methods in Molecular Biology, Clifton: Springer, 2016, pp. 53–60.
Trensz, F., F. Lucien, V. Couture, T. Söllrald, G. Drouin, A.-J. Rouleau, M. Grandbois, G. Lacraz, and G. Grenier. Increased microenvironment stiffness in damaged myofibers promotes myogenic progenitor cell proliferation. Skelet. Muscle 5:5, 2015.
Tsivitse, S. Notch and Wnt signaling, physiological stimuli and postnatal myogenesis. Int. J. Biol. Sci. 6:268–281, 2010.
Tu, J., Y. Li, and Z. Hu. Notch1 and 4 signaling responds to an increasing vascular wall shear stress in a rat model of arteriovenous malformations. Biomed. Res. Int. 2014. doi:10.1155/2014/368082.
Urciuolo, A., M. Quarta, V. Morbidoni, F. Gattazzo, S. Molon, P. Grumati, F. Montemurro, F. S. Tedesco, B. Blaauw, G. Cossu, G. Vozzi, T. A. Rando, and P. Bonaldo. Collagen VI regulates satellite cell self-renewal and muscle regeneration. Nat. Commun. 4:1964, 2013.
Varnum-Finney, B., L. Wu, M. Yu, C. Brashem-Stein, S. Staats, D. Flowers, J. D. Griffin, and I. D. Bernstein. Immobilization of Notch ligand, Delta-1, is required for induction of notch signaling. J. Cell Sci. 113(Pt 23):4313–4318, 2000.
Vieira, N. M., I. Elvers, M. S. Alexander, Y. B. Moreira, A. Eran, J. P. Gomes, J. L. Marshall, E. K. Karlsson, S. Verjovski-Almeida, K. Lindblad-Toh, L. M. Kunkel, and M. Zatz. Jagged 1 rescues the duchenne muscular dystrophy phenotype. Cell 163:1204–1213, 2015.
von Maltzahn, J., N. C. Chang, C. F. Bentzinger, and M. A. Rudnicki. Wnt signaling in myogenesis. Trends Cell Biol. 22:602–609, 2012.
von Maltzahn, J., A. E. Jones, R. J. Parks, and M. A. Rudnicki. Pax7 is critical for the normal function of satellite cells in adult skeletal muscle. Proc. Natl. Acad. Sci. USA 110:16474–16479, 2013.
Wang, X., and T. Ha. Defining single molecular forces required to activate integrin and notch signaling. Science 340:991–994, 2013.
Yin, H., F. Price, and M. A. Rudnicki. Satellite cells and the muscle stem cell niche. Physiol. Rev. 93:23–67, 2013.
Zhu, J.-H., C.-L. Chen, S. Flavahan, J. Harr, B. Su, and N. A. Flavahan. Cyclic stretch stimulates vascular smooth muscle cell alignment by redox-dependent activation of Notch3. Am. J. Physiol. Heart Circ. Physiol. 300:H1770–H1780, 2011.
Acknowledgments
We are grateful to Dr. Yasuhiro Matsumura (National Cancer Center Hospital, Kashiwa City, Japan) for providing us with his custom antibody to analyze fibrin clot formation in our study.
Author Contributions
HS, MAB, CAS, and PMG conceived the study and designed experiments. HS, SD, RYC, AJM, and EWL performed experiments, analyzed data, performed statistical analyses, and prepared figures. HS, MAB, SD, RYC, AJM, EWL, CAS, and PMG wrote, assembled, and revised the manuscript. All authors reviewed and approved the submission.
Funding
This study was funded by the Natural Sciences and Engineering Research Council (USRA fellowship to H.S., CREATE ToEP fellowship to S.D., RGPIN 327627-06 to C.A.S., RGPIN 435724-13 and Canada Research Chair 950-231201 to P.M.G.); Toronto Musculoskeletal Centre (Graduate Scholarships to M.A.B. and R.Y.C.); Barbara and Frank Milligan Foundation (R.Y.C.); Ontario Provincial Government (OGS-visa to M.A.B., 31390 and ER15-11-073 to P.M.G.); Canada Foundation for Innovation (31390 to P.M.G.); Krembil Foundation (Scholarship to M.A.B.); Toronto Western Arthritis Program (to P.M.G.); and Canadian Institutes of Health Research (MOP-302041 to C.A.S. and ONM-137370 to P.M.G.)
Conflict of interest
All authors declared that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Alyssa Panitch oversaw the review of this article.
Penney Gilbert is an Assistant Professor at the University of Toronto in the Institute of Biomaterials and Biomedical Engineering. She holds cross-appointments in the Department of Biochemistry and the Donnelly Centre for Cellular and Biomolecular Research. Gilbert received her PhD from the University of Pennsylvania under the mentorship of Valerie Weaver and then pursued Postdoctoral studies with Helen Blau at Stanford University. Her research team is focused on engineering human skeletal muscle culture models and uncovering niche cues that drive muscle stem cell fate changes in vivo. She is recipient of an Ontario Early Researcher Award and a BMES-CMBE Conference ‘Rising Star’ Award. She holds a Tier II Canada Research Chair in Endogenous Repair.

This article is part of the 2017 CMBE Young Innovators special issue.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Safaee, H., Bakooshli, M.A., Davoudi, S. et al. Tethered Jagged-1 Synergizes with Culture Substrate Stiffness to Modulate Notch-Induced Myogenic Progenitor Differentiation. Cel. Mol. Bioeng. 10, 501–513 (2017). https://doi.org/10.1007/s12195-017-0506-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-017-0506-7