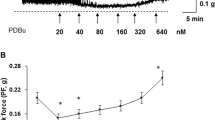
Abstract
We and other laboratories previously demonstrated that multiple signaling pathways are at work in SMC responses to applied mechanical stimuli, indicating the need for a high-throughput approach to study the complex mechanotransduction mechanism. In the present study, the feasibility of proteomic techniques to examine the mechanotransduction of bladder smooth muscle cells (SMC) was tested for the first time. Specifically, two-dimensional (2D) gel electrophoresis and image analyses were employed to separate and identify protein profiles in control and in cells exposed to cyclic pressure (0.1 Hz, 40 cmH2O) for 1 h. The results provide evidence that 20 (out of 155) spots exhibited changes (up- or down-regulation) in intensity between control and cyclic pressure groups. The mass spectrometry analysis provided sequences for 118 tryptic peptides from these 20 spots, and peptide mapping provided a positive match of peptide sequences to 24 distinct proteins. Moreover, the results of Western analysis independently confirmed up-regulation of nuclear matrix protein, lamin A/C in SMC exposed to cyclic pressure. Since the protein samples were predominantly of the cytosolic fraction, we concluded that the observed up-regulation was due to the translocation of either membrane-bound or nuclear localized proteins into the cytosol. Based on these observations, the present study demonstrated that the proteomic approach can be a powerful analytical tool for mechanobiology research that allows analysis of a large number of variables, thereby supporting development of new hypotheses to guide future investigation.



Similar content being viewed by others
References
Adam, R.M., S.H. Eaton, C. Estrada, A. Nimgaonkar, S.C. Shih, L.E. Smith, I.S. Kohane, D. Bagli, and M.R. Freeman, Mechanical stretch is a highly selective regulator of gene expression in human bladder smooth muscle cells. Physiol Genomics, 2004. 20(1): p. 36–44.
Adam, R.M., J.A. Roth, H.L. Cheng, D.C. Rice, J. Khoury, S.B. Bauer, C.A. Peters, and M.R. Freeman, Signaling through PI3 K/Akt mediates stretch and PDGF-BB-dependent DNA synthesis in bladder smooth muscle cells. J Urol, 2003. 169(6): p. 2388–93.
Backhaus, B.O., M. Kaefer, K.M. Haberstroh, K. Hile, J. Nagatomi, R.C. Rink, M.P. Cain, A. Casale, and R. Bizios, Alterations in the molecular determinants of bladder compliance at hydrostatic pressures less than 40 cm.H 2 O. J Urol, 2002. 168(6): p. 2600–4.
Biering-Sorensen, F., P. Bagi, and N. Hoiby, Urinary tract infections in patients with spinal cord lesions: treatment and prevention. Drugs, 2001. 61(9): p. 1275–87.
Davies, P.F., J.A. Spaan, and R. Krams, Shear stress biology of the endothelium. Ann Biomed Eng, 2005. 33(12): p. 1714–8.
Ewalt, D.H., P.S. Howard, B. Blyth, H.M. Snyder, 3rd, J.W. Duckett, R.M. Levin, and E.J. Macarak, Is lamina propria matrix responsible for normal bladder compliance? J Urol, 1992. 148(2 Pt 2): p. 544–9.
Frye, S.R., A. Yee, S.G. Eskin, R. Guerra, X. Cong, and L.V. McIntire, cDNA microarray analysis of endothelial cells subjected to cyclic mechanical strain: importance of motion control. Physiol Genomics, 2005. 21(1): p. 124–30.
Galvin, D.J., R.W. Watson, J.I. Gillespie, H. Brady, and J.M. Fitzpatrick, Mechanical stretch regulates cell survival in human bladder smooth muscle cells in vitro. Am J Physiol Renal Physiol, 2002. 283(6): p. F1192–9.
Haberstroh, K.M., M. Kaefer, and R. Bizios, Inhibition of pressure induced bladder smooth muscle cell hyperplasia using CRM197. J Urol, 2000. 164(4): p. 1329–33.
Haberstroh, K.M., M. Kaefer, A.B. Retik, M.R. Freeman, and R. Bizios, The effects of sustained hydrostatic pressure on select bladder smooth muscle cell functions. J Urol, 1999. 162(6): p. 2114–8.
Ingber, D.E., Mechanobiology and diseases of mechanotransduction. Ann Med, 2003. 35(8): p. 564–77.
Lammerding, J., P.C. Schulze, T. Takahashi, S. Kozlov, T. Sullivan, R.D. Kamm, C.L. Stewart, and R.T. Lee, Lamin A/C deficiency causes defective nuclear mechanics and mechanotransduction. J Clin Invest, 2004. 113(3): p. 370–8.
Lee, M.S., K.W. Kim, Y.H. Kim, and G.M. Lee, Proteome analysis of antibody-expressing CHO cells in response to hyperosmotic pressure. Biotechnol Prog, 2003. 19(6): p. 1734–41.
Nagatomi, J., B.P. Arulanandam, D.W. Metzger, A. Meunier, and R. Bizios, Frequency- and duration-dependent effects of cyclic pressure on select bone cell functions. Tissue Eng, 2001. 7(6): p. 717–28.
Nguyen, H.T., R.M. Adam, S.H. Bride, J.M. Park, C.A. Peters, and M.R. Freeman, Cyclic stretch activates p38 SAPK2-, ErbB2-, and AT1-dependent signaling in bladder smooth muscle cells. Am J Physiol Cell Physiol, 2000. 279(4): p. C1155–67.
Orsola, A., R.M. Adam, C.A. Peters, and M.R. Freeman, The decision to undergo DNA or protein synthesis is determined by the degree of mechanical deformation in human bladder muscle cells. Urology, 2002. 59(5): p. 779–83.
Park, J.M., R.M. Adam, C.A. Peters, P.D. Guthrie, Z. Sun, M. Klagsbrun, and M.R. Freeman, AP-1 mediates stretch-induced expression of HB-EGF in bladder smooth muscle cells. Am J Physiol, 1999. 277(2 Pt 1): p. C294–301.
Park, J.M., J.G. Borer, M.R. Freeman, and C.A. Peters, Stretch activates heparin-binding EGF-like growth factor expression in bladder smooth muscle cells. Am J Physiol, 1998. 275(5 Pt 1): p. C1247–54.
Park, J.M., T. Yang, L.J. Arend, J.B. Schnermann, C.A. Peters, M.R. Freeman, and J.P. Briggs, Obstruction stimulates COX-2 expression in bladder smooth muscle cells via increased mechanical stretch. Am J Physiol, 1999. 276(1 Pt 2): p. F129–36.
Peters, D.G., X.C. Zhang, P.V. Benos, E. Heidrich-O’Hare, and R.E. Ferrell, Genomic analysis of immediate/early response to shear stress in human coronary artery endothelial cells. Physiol Genomics, 2002. 12(1): p. 25–33.
Rosenberg, I.M., Protein Analysis and Purification—Benchtop Techniques. 2nd ed. 2006, Boston: Birkhauser. 231–235.
Shin, H.Y., M.L. Smith, K.J. Toy, P.M. Williams, R. Bizios, and M.E. Gerritsen, VEGF-C mediates cyclic pressure-induced endothelial cell proliferation. Physiol Genomics, 2002. 11(3): p. 245–51.
Stover, J. and J. Nagatomi, Cyclic Pressure Stimulates DNA Synthesis through the PI3 K/Akt Signaling Pathway in Rat Bladder Smooth Muscle Cells. Ann Biomed Eng, 2007. 35(9): p. 1585–94.
Yalla, S.V., A.B. Rossier, and B. Fam, Dyssynergic vesicourethral responses during bladder rehabilitation in spinal cord injury patients: effects of suprapubic percussion, crede method and bethanechol chloride. J Urol, 1976. 115(5): p. 575–9.
Acknowledgments
The authors wish to thank Dr. Bruce Gao, Department of Bioengineering, Clemson University, for providing the rats used as the source of bladder cells. The funding for this research was provided by Paralyzed Veterans of America (2289-02) and by Clemson University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagatomi, J., Wu, Y. & Gray, M. Proteomic Analysis of Bladder Smooth Muscle Cell Response to Cyclic Hydrostatic Pressure. Cel. Mol. Bioeng. 2, 166–173 (2009). https://doi.org/10.1007/s12195-009-0043-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-009-0043-0