Abstract
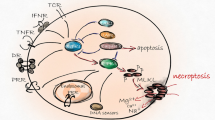
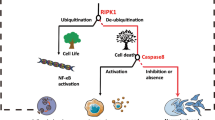
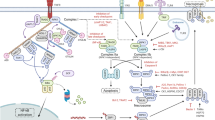
Necroptosis is a new type of programmed cell death discovered in recent years, playing an important role in various diseases. Since it was conceptualized in 2005, research on necroptosis has developed rapidly. However, few bibliometric analyses have provided a comprehensive overview of the field. This study aimed to employ a bibliometric analysis to assess necroptosis research’s current status and hotspot, highlight landmark findings, and orientate future research. A total of 3993 publications from the WoSCC were collected for this study. Multiple tools were used for bibliometric analysis and data visualization, including an online website, VOSviewer, CiteSpace, and HistCite. Publications related to necroptosis have increased significantly annually, especially in the last 5 years. Globally, the USA and Harvard University are the most outstanding countries and institutions in this field, respectively. The academic groups managed by Peter Vandenabeele and Junying Yuan both have permanent and intensive research on necroptosis. Cell Death and Differentiation is the most vital journal in this field. The molecular mechanisms of necroptosis and its role in disease are the focus of current research, while the crosstalk between programmed cell death is an emerging direction in the field. The “reactive oxygen species”, “innate immunity”, and “programmed cell death” may be potential research hotspots. Our results present a comprehensive knowledge map and explore research trends. Researchers and funding agencies on necroptosis can obtain helpful references from our study.









Similar content being viewed by others
References
Anderton H, Wicks IP, Silke J (2020) Cell death in chronic inflammation: breaking the cycle to treat rheumatic disease. Nat Rev Rheumatol 16(9):496–513
Cai ZY, Jitkaew S, Zhao J, Chiang HC, Choksi S, Liu J, Ward Y, Wu LG, Liu ZG (2014) Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis. Nat Cell Biol 16(1):55-+
Cannon G, Kiang JG (2022) A review of the impact on the ecosystem after ionizing irradiation: wildlife population. Int J Radiat Biol 98(6):1054–1062
Chaudhary GR, Yadav PK, Yadav AK, Tiwari M, Gupta A, Sharma A, Sahu K, Pandey AN, Pandey AK, Chaube SK (2019) Necrosis and necroptosis in germ cell depletion from mammalian ovary. J Cell Physiol 234(6):8019–8027
Chen C (2004) Searching for intellectual turning points: progressive knowledge domain visualization. Proc Natl Acad Sci U S A 1(Suppl 1):5303–5310
Chen Y, Li Y, Guo L, Hong J, Zhao W, Hu X, Chang C, Liu W, Xiong K (2020) Bibliometric analysis of the inflammasome and pyroptosis in brain. Front Pharmacol 11:626502
Chen Y, Long T, Xu Q, Zhang C (2021) Bibliometric analysis of ferroptosis in stroke from 2013 to 2021. Front Pharmacol 12:817364
Cho Y, Challa S, Moquin D, Genga R, Ray TD, Guildford M, Chan FKM (2009) Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137(6):1112–1123
Conos SA, Chen KW, De Nardo D, Hara H, Whitehead L, Núñez G, Masters SL, Murphy JM, Schroder K, Vaux DL (2017) Active MLKL triggers the NLRP3 inflammasome in a cell-intrinsic manner. Proc Natl Acad Sci 114(6):E961–E969
Cui Y, Yin K, Gong Y, Qu Y, Liu H, Lin H (2019) Atrazine induces necroptosis by miR-181-5p targeting inflammation and glycometabolism in carp lymphocytes. Fish Shellfish Immunol 94:730–738
Degterev A, Hitomi J, Germscheid M, Ch'en IL, Korkina O, Teng X, Abbott D, Cuny GD, Yuan C, Wagner G, Hedrick SM, Gerber SA, Lugovskoy A, Yuan J (2008) Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat Chem Biol 4(5):313–321
Degterev A, Huang ZH, Boyce M, Li YQ, Jagtap P, Mizushima N, Cuny GD, Mitchison TJ, Moskowitz MA, Yuan JY (2005) Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol 1(2):112–119
Feng X, Song Q, Yu A, Tang H, Peng Z, Wang X (2015) Receptor-interacting protein kinase 3 is a predictor of survival and plays a tumor suppressive role in colorectal cancer. Neoplasma 62(4):592–601
Frank D, Vince JE (2019) Pyroptosis versus necroptosis: similarities, differences, and crosstalk. Cell Death Differ 26(1):99–114
Friedmann Angeli JP, Schneider M, Proneth B, Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch A, Eggenhofer E (2014) Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat Cell Biol 16(12):1180–1191
Fritsch M, Günther SD, Schwarzer R, Albert MC, Schorn F, Werthenbach JP, Schiffmann LM, Stair N, Stocks H, Seeger JM, Lamkanfi M, Krönke M, Pasparakis M, Kashkar H (2019) Caspase-8 is the molecular switch for apoptosis, necroptosis and pyroptosis. Nature 575(7784):683–687
Galluzzi L, Kepp O, Chan FK, Kroemer G (2017) Necroptosis: mechanisms and relevance to disease. Annu Rev Pathol 12:103–130
Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P, Alnemri ES, Altucci L, Amelio I, Andrews DW, Annicchiarico-Petruzzelli M, Antonov AV, Arama E, Baehrecke EH, Barlev NA, Bazan NG, Bernassola F, Bertrand MJM, Bianchi K et al (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ 25(3):486–541
Garfield E, Paris S, Stock WG (2006) HistCiteTM: a software tool for informetric analysis of citation linkage. Information Wissenschaft und Praxis 57(8):391
Ge Y, Yang H, Zhao L, Luo S, Zhang H, Chen S (2018) Structural and functional conservation of half-smooth tongue sole Cynoglossus semilaevis RIP3 in cell death signalling. Fish Shellfish Immunol 82:573–578
Gong Y, Fan Z, Luo G, Yang C, Huang Q, Fan K, Cheng H, Jin K, Ni Q, Yu X, Liu C (2019) The role of necroptosis in cancer biology and therapy. Mol Cancer 18(1):100
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140(6):883–899
Gutierrez KD, Davis MA, Daniels BP, Olsen TM, Ralli-Jain P, Tait SW, Gale M, Oberst A (2017) MLKL activation triggers NLRP3-mediated processing and release of IL-1β independently of gasdermin-D. J Immunol 198(5):2156–2164
He S, Wang L, Miao L, Wang T, Du F, Zhao L, Wang X (2009) Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell 137(6):1100–1111
Hsu SK, Chang WT, Lin IL, Chen YF, Padalwar NB, Cheng KC, Teng YN, Wang CH, Chiu CC (2020) The role of necroptosis in ROS-mediated cancer therapies and its promising applications. Cancers (Basel) 12(8)
Kang YJ, Bang B-R, Han KH, Hong L, Shim E-J, Ma J, Lerner RA, Otsuka M (2015) Regulation of NKT cell-mediated immune responses to tumours and liver inflammation by mitochondrial PGAM5-Drp1 signalling. Nat Commun 6(1):1–15
Karunakaran D, Nguyen MA, Geoffrion M, Vreeken D, Lister Z, Cheng HS, Otte N, Essebier P, Wyatt H, Kandiah JW, Jung R, Alenghat FJ, Mompeon A, Lee R, Pan C, Gordon E, Rasheed A, Lusis AJ, Liu P et al (2021) RIPK1 expression associates with inflammation in early atherosclerosis in humans and can be therapeutically silenced to reduce NF-κB activation and atherogenesis in mice. Circulation 143(2):163–177
Khoury MK, Gupta K, Franco SR, Liu B (2020) Necroptosis in the pathophysiology of disease. Am J Pathol 190(2):272–285
Koo GB, Morgan MJ, Lee DG, Kim WJ, Yoon JH, Koo JS, Kim SI, Kim SJ, Son MK, Hong SS, Levy JM, Pollyea DA, Jordan CT, Yan P, Frankhouser D, Nicolet D, Maharry K, Marcucci G, Choi KS et al (2015) Methylation-dependent loss of RIP3 expression in cancer represses programmed necrosis in response to chemotherapeutics. Cell Res 25(6):707–725
Li D, Meng L, Xu T, Su Y, Liu X, Zhang Z, Wang X (2017) RIPK1-RIPK3-MLKL-dependent necrosis promotes the aging of mouse male reproductive system. Elife 6
Li S, Zhang J, Liu C, Wang Q, Yan J, Hui L, Jia Q, Shan H, Tao L, Zhang M (2021) The role of mitophagy in regulating cell death. Oxidative Med Cell Longev 2021
Liao S, Apaijai N, Chattipakorn N, Chattipakorn SC (2020) The possible roles of necroptosis during cerebral ischemia and ischemia/reperfusion injury. Arch Biochem Biophys 695:108629
Linkermann A, Green DR (2014) Necroptosis. N Engl J Med 370(5):455–465
Lukenaite B, Griciune E, Leber B, Strupas K, Stiegler P, Schemmer P (2022) Necroptosis in solid organ transplantation: a literature overview. Int J Mol Sci 23(7):3677
Ma D, Yang B, Guan B, Song L, Liu Q, Fan Y, Zhao L, Wang T, Zhang Z, Gao Z, Li S, Xu H (2021) A bibliometric analysis of pyroptosis from 2001 to 2021. Front Immunol 12:731933
Mann J, Yang N, Montpetit R, Kirschenman R, Lemieux H, Goping IS (2020) BAD sensitizes breast cancer cells to docetaxel with increased mitotic arrest and necroptosis. Sci Rep 10(1):355
Miao L, Zhang J, Zhang Z, Wang S, Tang F, Teng M, Li Y (2022) A bibliometric and knowledge-map analysis of CAR-T cells from 2009 to 2021. Front Immunol 13:840956
Moriwaki K, Balaji S, McQuade T, Malhotra N, Kang J, Chan FK-M (2014) The necroptosis adaptor RIPK3 promotes injury-induced cytokine expression and tissue repair. Immunity 41(4):567–578
Newton K, Manning G (2016) Necroptosis and inflammation. Annu Rev Biochem 85:743–763
Pasparakis M, Vandenabeele P (2015) Necroptosis and its role in inflammation. Nature 517(7534):311–320
Place DE, Kanneganti T-D (2020) The innate immune system and cell death in autoinflammatory and autoimmune disease. Curr Opin Immunol 67:95–105
Polykratis A, Martens A, Eren RO, Shirasaki Y, Yamagishi M, Yamaguchi Y, Uemura S, Miura M, Holzmann B, Kollias G (2019) A20 prevents inflammasome-dependent arthritis by inhibiting macrophage necroptosis through its ZnF7 ubiquitin-binding domain. Nat Cell Biol 21(6):731–742
Qi J, Liu Y, Yang P, Chen T, Liu XZ, Yin Y, Zhang J, Wang F (2015) Heat shock protein 90 inhibition by 17-dimethylaminoethylamino-17-demethoxygeldanamycin protects blood-brain barrier integrity in cerebral ischemic stroke. Am J Transl Res 7(10):1826
Qin XD, Ma YX, Tan HY, Wang and Z Cai (2019) The role of necroptosis in cancer: a double-edged sword? Biochimica et Biophysica Acta (BBA)-Reviews on Cancer 1871(2): 259-266
Remijsen Q, Goossens V, Grootjans S, Van den Haute C, Vanlangenakker N, Dondelinger Y, Roelandt R, Bruggeman I, Goncalves A, Bertrand MJ, Baekelandt V, Takahashi N, Berghe TV, Vandenabeele P (2014) Depletion of RIPK3 or MLKL blocks TNF-driven necroptosis and switches towards a delayed RIPK1 kinase-dependent apoptosis. Cell Death Dis 5(1):e1004
Ren J, Long Y, Liu R, Song G, Li Q, Cui Z (2021) Characterization of biological pathways regulating acute cold resistance of zebrafish. Int J Mol Sci 22(6)
Schwarzer R, Laurien L, Pasparakis M (2020) New insights into the regulation of apoptosis, necroptosis, and pyroptosis by receptor interacting protein kinase 1 and caspase-8. Curr Opin Cell Biol 63:186–193
Sepand MR, Aliomrani M, Hasani-Nourian Y, Khalhori MR, Farzaei MH, Sanadgol N (2020) Mechanisms and pathogenesis underlying environmental chemical-induced necroptosis. Environ Sci Pollut Res Int 27(30):37488–37501
Shen J, Shen H, Ke L, Chen J, Dang X, Liu B, Hua Y (2022) Knowledge mapping of immunotherapy for hepatocellular carcinoma: a bibliometric study. Front Immunol 13:815575
Sun LM, Wang HY, Wang ZG, He SD, Chen S, Liao DH, Wang L, Yan JC, Liu WL, Lei XG, Wang XD (2012) Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell 148(1-2):213–227
Tang D, Kang R, Berghe TV, Vandenabeele P, Kroemer G (2019) The molecular machinery of regulated cell death. Cell Res 29(5):347–364
Tian Q, Qin B, Gu Y, Zhou L, Chen S, Zhang S, Zhang S, Han Q, Liu Y, Wu X (2020) ROS-mediated necroptosis is involved in iron overload-induced osteoblastic cell death. Oxidative Med Cell Longev 2020:1295382
van Eck NJ, Waltman L (2010) Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics 84(2):523–538
Vandenabeele P, Galluzzi L, Vanden Berghe T, Kroemer G (2010) Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat Rev Mol Cell Biol 11(10):700–714
Vanlangenakker N, Vanden Berghe T, Bogaert P, Laukens B, Zobel K, Deshayes K, Vucic D, Fulda S, Vandenabeele P, Bertrand MJM (2011) cIAP1 and TAK1 protect cells from TNF-induced necrosis by preventing RIP1/RIP3-dependent reactive oxygen species production. Cell Death Different 18(4):656–665
Wang F, Xu L, Wen B, Song S, Zhou Y, Wu H, Wu Q, Wang Y, Tong X, Yan H (2023) Ultrasound-excited temozolomide sonosensitization induces necroptosis in glioblastoma. Cancer Lett 554:216033
Wang H, Sun L, Su L, Rizo J, Liu L, Wang LF, Wang FS, Wang X (2014) Mixed lineage kinase domain-like protein MLKL causes necrotic membrane disruption upon phosphorylation by RIP3. Mol Cell 54(1):133–146
Wang L, Wang L, Shi X, Xu S (2020) Chlorpyrifos induces the apoptosis and necroptosis of L8824 cells through the ROS/PTEN/PI3K/AKT axis. J Hazard Mater 398:122905
Weinlich R, Oberst A, Beere HM, Green DR (2017) Necroptosis in development, inflammation and disease. Nat Rev Mol Cell Biol 18(2):127–136
Wu H, Wang Y, Tong L, Yan H, Sun Z (2021a) Global research trends of ferroptosis: a rapidly evolving field with enormous potential. Front Cell Dev Biol 9:646311
Wu H, Zhou Y, Wang Y, Tong L, Wang F, Song S, Xu L, Liu B, Yan H, Sun Z (2021b) Current state and future directions of intranasal delivery route for central nervous system disorders: a scientometric and visualization analysis. Front Pharmacol 12:717192
Wu Z, Geng Y, Lu X, Shi Y, Wu G, Zhang M, Shan B, Pan H, Yuan J (2019) Chaperone-mediated autophagy is involved in the execution of ferroptosis. Proc Natl Acad Sci U S A 116(8):2996–3005
Xie X, Zhao Y, Ma CY, Xu XM, Zhang YQ, Wang CG, Jin J, Shen X, Gao JL, Li N, Sun ZJ, Dong DL (2015) Dimethyl fumarate induces necroptosis in colon cancer cells through GSH depletion/ROS increase/MAPKs activation pathway. Br J Pharmacol 172(15):3929–3943
Yan WT, Lu S, Yang YD, Ning WY, Cai Y, Hu XM, Zhang Q, Xiong K (2021) Research trends, hot spots and prospects for necroptosis in the field of neuroscience. Neural Regen Res 16(8):1628–1637
Yang Z, Wang Y, Zhang Y, He X, Zhong C-Q, Ni H, Chen X, Liang Y, Wu J, Zhao S (2018) RIP3 targets pyruvate dehydrogenase complex to increase aerobic respiration in TNF-induced necroptosis. Nat Cell Biol 20(2):186–197
Yu X, Deng Q, Li W, Xiao L, Luo X, Liu X, Yang L, Peng S, Ding Z, Feng T, Zhou J, Fan J, Bode AM, Dong Z, Liu J, Cao Y (2015) Neoalbaconol induces cell death through necroptosis by regulating RIPK-dependent autocrine TNFα and ROS production. Oncotarget 6(4):1995–2008
Yuan J, Amin P, Ofengeim D (2019) Necroptosis and RIPK1-mediated neuroinflammation in CNS diseases. Nat Rev Neurosci 20(1):19–33
Zhang D-W, Shao J, Lin J, Zhang N, Lu B-J, Lin S-C, Dong M-Q, Han J (2009) RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325(5938):332–336
Zhang Y, Huo L, Wei Z, Tang Q, Sui H (2022) Hotspots and frontiers in inflammatory tumor microenvironment research: a scientometric and visualization analysis. Front Pharmacol 13:862585
Zhang Y, Su SS, Zhao S, Yang Z, Zhong CQ, Chen X, Cai Q, Yang ZH, Huang D, Wu R, Han J (2017) RIP1 autophosphorylation is promoted by mitochondrial ROS and is essential for RIP3 recruitment into necrosome. Nat Commun 8:14329
Zhao J, Jitkaew S, Cai Z, Choksi S, Li Q, Luo J, Liu ZG (2012) Mixed lineage kinase domain-like is a key receptor interacting protein 3 downstream component of TNF-induced necrosis. Proc Natl Acad Sci U S A 109(14):5322–5327
Zhou Y, Liao J, Mei Z, Liu X, Ge J (2021a) Insight into crosstalk between ferroptosis and necroptosis: novel therapeutics in ischemic stroke. Oxidative Med Cell Longev 2021:9991001
Zhou Q, Wu F, Zhao M, Yang M (2021b) Bibliometric evaluation of 2012-2020 publications on ferroptosis in cancer treatment. Front Cell Dev Biol 9:793347
Zyoud SH, Al-Jabi SW, Amer R, Shakhshir M, Shahwan M, Jairoun AA, Akkawi M, Abu Taha A (2022) Global research trends on the links between the gut microbiome and cancer: a visualization analysis. J Transl Med 20(1):83
Acknowledgements
We thank Dr. Zhang Xueqian for his guidance on the language of this paper.
Funding
Chengdu Municipal Health Commission (2021095); Langfang Science and Technology Programme Support Project (2023013070)
Author information
Authors and Affiliations
Contributions
Conceptualization: FC.W. and L.S.
Methodology: D.M. and X.W.
Investigation: D.M. and J.L.
Visualization: D.M. and X.W.
Funding acquisition: Y.C. and D.M.
Project administration: FC.W.
Supervision: L.S.
Writing—original draft: D.M. and FC.W.
Writing—review & editing: FC.W.
Corresponding author
Ethics declarations
Data availability
All data are available in the main text or supplementary materials.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ma, D., Wang, X., Liu, J. et al. The development of necroptosis: what we can learn. Cell Stress and Chaperones 28, 969–987 (2023). https://doi.org/10.1007/s12192-023-01390-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-023-01390-5