Abstract
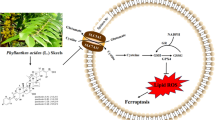
Doxorubicin (DOX) is a chemotherapeutic drug for a variety of malignancies, while its application is restricted by the cardiovascular toxic effects characterized by oxidative stress. Ferroptosis is a novel iron-dependent regulated cell death driven by lipid peroxidation. Our study aimed to investigate the role of Elabela (ELA) in DOX-induced oxidative stress and ferroptosis. In cultured rat aortic adventitial fibroblasts (AFs), stimulation with DOX dramatically induced cytotoxicity with reduced cell viability and migration ability, and enhanced lactate dehydrogenase (LDH) activity. Importantly, ELA and ferrostatin-1 (Fer-1) mitigated DOX-mediated augmentation of reactive oxygen species (ROS) in rat aortic AFs, accompanied by upregulated levels of Nrf2, SLC7A11, GPX4, and GSH. In addition, ELA reversed DOX-induced dysregulation of apoptosis- and inflammation-related factors including Bax, Bcl2, interleukin (IL)-1β, IL6, IL-10, and CXCL1. Intriguingly, knockdown of Krüppel-like factor 15 (KLF15) by siRNA abolished ELA-mediated alleviation of ROS production and inflammatory responses. More importanly, KLF15 siRNA impeded the beneficial roles of ELA in DOX-pretreated rat aortic AFs by suppressing the Nrf2/SLC7A11/GPX4 signaling. In conclusion, ELA prevents DOX-triggered promotion of cytotoxicity, and exerts anti-oxidative and anti-ferroptotic effects in rat aortic AFs via activation of the KLF15/GPX4 signaling, indicating a promising therapeutic value of ELA in antagonizing DOX-mediated cardiovascular abnormality and disorders.








Similar content being viewed by others
Data availability
The data that support the findings of this study are available on reasonable request from the corresponding author.
References
Alhazzani K, Alotaibi MR, Alotaibi FN, Aljerian K, As Sobeai HM, Alhoshani AR, Alanazi AZ, Alanazi WA, Alswayyed M (2021) Protective effect of valsartan against doxorubicin-induced cardiotoxicity: histopathology and metabolomics in vivo study. J Biochem Mol Toxicol 35:e22842. https://doi.org/10.1002/jbt.22842
Bosman M, Krüger DN, Favere K, Wesley CD, Neutel CHG, Van Asbroeck B, Diebels OR, Faes B, Schenk TJ, Martinet W, De Meyer GRY, Van Craenenbroeck EM, Guns PJDF (2021) Doxorubicin impairs smooth muscle cell contraction: novel insights in vascular toxicity. Int J Mol Sci 22. https://doi.org/10.3390/ijms222312812
Burridge PW, Li YF, Matsa E, Wu H, Ong SG, Sharma A, Holmström A, Chang AC, Coronado MJ, Ebert AD, Knowles JW, Telli ML, Witteles RM, Blau HM, Bernstein D, Altman RB, Wu JC (2016) Human induced pluripotent stem cell-derived cardiomyocytes recapitulate the predilection of breast cancer patients to doxorubicin-induced cardiotoxicity. Nat Med 22:547–556. https://doi.org/10.1038/nm.4087
Carvalho C, Santos RX, Cardoso S, Correia S, Oliveira PJ, Santos MS, Moreira PI (2009) Doxorubicin: the good, the bad and the ugly effect. Curr Med Chem 16:3267–3285. https://doi.org/10.2174/092986709788803312
Catanzaro MP, Weiner A, Kaminaris A, Li C, Cai F, Zhao F, Kobayashi S, Kobayashi T, Huang Y, Sesaki H, Liang Q (2019) Doxorubicin-induced cardiomyocyte death is mediated by unchecked mitochondrial fission and mitophagy. FASEB J 33:11096–11108. https://doi.org/10.1096/fj.201802663R
Chen CC, Hong HJ, Hao WR, Cheng TH, Liu JC, Sung LC (2019) Nicorandil prevents doxorubicin-induced human umbilical vein endothelial cell apoptosis. Eur J Pharmacol 859:172542. https://doi.org/10.1016/j.ejphar.2019.172542
Chen X, Li J, Kang R, Klionsky DJ, Tang D (2021) Ferroptosis: machinery and regulation. Autophagy 17:2054–2081. https://doi.org/10.1080/15548627.2020.1810918
Chen D, Yu W, Zhong C, Hong Q, Huang G, Que D, Wang Y, Yang Y, Rui B, Zhuang Z, Liang M, Ye Z, Yan X, Lv J, Zhang R, Yan J, Yang P (2022) Elabela ameliorates doxorubicin-induced cardiotoxicity by promoting autophagic flux through TFEB pathway. Pharmacol Res 178:106186. https://doi.org/10.1016/j.phrs.2022.106186
Dodson M, Castro Portuguez R, Zhang DD (2019) NRF2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol 23:101107. https://doi.org/10.1016/j.redox.2019.101107
Doll S, Freitas FP, Shah R, Aldrovandi M, da Silva MC, Ingold I, Goya Grocin A, Xavier da Silva TN, Panzilius E, Scheel CH, Mourão A, Buday K, Sato M, Wanninger J, Vignane T, Mohana V, Rehberg M, Flatley A, Schepers A, Kurz A, White D, Sauer M, Sattler M, Tate EW, Schmitz W, Schulze A, O’Donnell V, Proneth B, Popowicz GM, Pratt DA, Angeli JPF, Conrad M (2019) FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575:693–698. https://doi.org/10.1038/s41586-019-1707-0
Dong H, Xia Y, Jin S, Xue C, Wang Y, Hu R, Jiang H (2021) Nrf2 attenuates ferroptosis-mediated IIR-ALI by modulating TERT and SLC7A11. Cell Death Dis 12:1027. https://doi.org/10.1038/s41419-021-04307-1
Gao L, Guo Y, Liu X, Shang D, Du Y (2017) KLF15 protects against isoproterenol-induced cardiac hypertrophy via regulation of cell death and inhibition of Akt/mTOR signaling. Biochem Biophys Res Commun 487:22–27. https://doi.org/10.1016/j.bbrc.2017.03.087
Gao M, Fan K, Chen Y, Zhang G, Chen J, Zhang Y (2022) Understanding the mechanistic regulation of ferroptosis in cancer: the gene matters. J Genet Genomics S1673–8527(22):00160–00166. https://doi.org/10.1016/j.jgg.2022.06.002
He S, Lu Y, Guo Y, Li S, Lu X, Shao S, Zhou H, Wang R, Wang J, Gao P, Li X (2021) Krüppel-like factor 15 modulates CXCL1/CXCR2 signaling-mediated inflammatory response contributing to angiotensin II-induced cardiac remodeling. Front Cell Dev Biol 9:644954. https://doi.org/10.3389/fcell.2021.644954
Hou K, Shen J, Yan J, Zhai C, Zhang J, Pan JA, Zhang Y, Jiang Y, Wang Y, Lin RZ, Cong H, Gao S, Zong WX (2021) Loss of TRIM21 alleviates cardiotoxicity by suppressing ferroptosis induced by the chemotherapeutic agent doxorubicin. EBioMedicine 69:103456. https://doi.org/10.1016/j.ebiom.2021.103456
Jain A, Rani V (2018) Mode of treatment governs curcumin response on doxorubicin-induced toxicity in cardiomyoblasts. Mol Cell Biochem 442:81–96. https://doi.org/10.1007/s11010-017-3195-6
Kuwabara JT, Tallquist MD (2017) Tracking adventitial fibroblast contribution to disease: a review of current methods to identify resident fibroblasts. Arterioscler Thromb Vasc Biol 37:1598–1607. https://doi.org/10.1161/ATVBAHA.117.308199
Li L, Xu W, Zhang L (2021) KLF15 regulates oxidative stress response in cardiomyocytes through NAD. Metabolites 11:620. https://doi.org/10.3390/metabo11090620
Liu B, Xu L, Yu X, Li W, Sun X, Xiao S, Guo M, Wang H (2018) Protective effect of KLF15 on vascular endothelial dysfunction induced by TNF-α. Mol Med Rep 18:1987–1994. https://doi.org/10.3892/mmr.2018.9195
Liu X, Yang Y, Song J, Li D, Liu X, Li C, Ma Z, Zhong J, Wang L (2022) Knockdown of forkhead box protein P1 alleviates hypoxia reoxygenation injury in H9c2 cells through regulating Pik3ip1/Akt/eNOS and ROS/mPTP pathway. Bioengineered 13:1320–1334. https://doi.org/10.1080/21655979.2021.2016046
Lu YY, Li XD, Zhou HD, Shao S, He S, Hong MN, Liu JC, Xu YL, Wu YJ, Zhu DL, Wang JG, Gao PJ (2019) Transactivation domain of Krüppel-like factor 15 negatively regulates angiotensin II-induced adventitial inflammation and fibrosis. FASEB J 33:6254–6268. https://doi.org/10.1096/fj.201801809R
Ma Z, Song JJ, Martin S, Yang XC, Zhong JC (2021) The Elabela-APJ axis: a promising therapeutic target for heart failure. Heart Fail Rev 26:1249–1258. https://doi.org/10.1007/s10741-020-09957-5
Matt S, Hofmann TG (2016) The DNA damage-induced cell death response: a roadmap to kill cancer cells. Cell Mol Life Sci 73:2829–2850. https://doi.org/10.1007/s00018-016-2130-4
Mišúth S, Uhrinová M, Klimas J, Vavrincová Yaghi D, Vavrinec P (2021) Vildagliptin improves vascular smooth muscle relaxation and decreases cellular senescence in the aorta of doxorubicin-treated rats. Vascul Pharmacol 138:106855. https://doi.org/10.1016/j.vph.2021.106855
Octavia Y, Tocchetti CG, Gabrielson KL, Janssens S, Crijns HJ, Moens AL (2012) Doxorubicin-induced cardiomyopathy: from molecular mechanisms to therapeutic strategies. J Mol Cell Cardiol 52:1213–1225. https://doi.org/10.1016/j.yjmcc.2012.03.006
Pan JA, Zhang H, Lin H, Gao L, Zhang HL, Zhang JF, Wang CQ, Gu J (2021) Irisin ameliorates doxorubicin-induced cardiac perivascular fibrosis through inhibiting endothelial-to-mesenchymal transition by regulating ROS accumulation and autophagy disorder in endothelial cells. Redox Biol 46:102120. https://doi.org/10.1016/j.redox.2021.102120
Park E, Chung SW (2019) ROS-mediated autophagy increases intracellular iron levels and ferroptosis by ferritin and transferrin receptor regulation. Cell Death Dis 10:822. https://doi.org/10.1038/s41419-019-2064-5
Qi W, Boliang W, Xiaoxi T, Guoqiang F, Jianbo X, Gang W (2020) Cardamonin protects against doxorubicin-induced cardiotoxicity in mice by restraining oxidative stress and inflammation associated with Nrf2 signaling. Biomed Pharmacother 122:109547. https://doi.org/10.1016/j.biopha.2019.109547
Rawat PS, Jaiswal A, Khurana A, Bhatti JS, Navik U (2021) Doxorubicin-induced cardiotoxicity: An update on the molecular mechanism and novel therapeutic strategies for effective management. Biomed Pharmacother 139:111708. https://doi.org/10.1016/j.biopha.2021.111708
Renu K, Abilash VG, Tirupathi Pichiah PB, Arunachalam S (2018) Molecular mechanism of doxorubicin-induced cardiomyopathy - an update. Eur J Pharmacol 818:241–253. https://doi.org/10.1016/j.ejphar.2017.10.043
Rivankar S (2014) An overview of doxorubicin formulations in cancer therapy. J Cancer Res Ther 10:853–858. https://doi.org/10.4103/0973-1482.139267
Seibt TM, Proneth B, Conrad M (2019) Role of GPX4 in ferroptosis and its pharmacological implication. Free Radic Biol Med 133:144–152. https://doi.org/10.1016/j.freeradbiomed.2018.09.014
Sirangelo I, Sapio L, Ragone A, Naviglio S, Iannuzzi C, Barone D, Giordano A, Borriello M (2020) Vanillin prevents doxorubicin-induced apoptosis and oxidative stress in rat H9c2 cardiomyocytes. Nutrients 12:2317. https://doi.org/10.3390/nu12082317
Song JJ, Yang M, Liu Y, Song JW, Wang J, Chi HJ, Liu XY, Zuo K, Yang XC, Zhong JC (2020) MicroRNA-122 aggravates angiotensin II-mediated apoptosis and autophagy imbalance in rat aortic adventitial fibroblasts via the modulation of SIRT6-elabela-ACE2 signaling. Eur J Pharmacol 883:173374. https://doi.org/10.1016/j.ejphar.2020.173374
Song JJ, Yang M, Liu Y, Song JW, Liu XY, Miao R, Zhang ZZ, Liu Y, Fan YF, Zhang Q, Dong Y, Yang XC, Zhong JC (2021) Elabela prevents angiotensin II-induced apoptosis and inflammation in rat aortic adventitial fibroblasts via the activation of FGF21-ACE2 signaling. J Mol Histol 52:905–918. https://doi.org/10.1007/s10735-021-10011-3
Songbo M, Lang H, Xinyong C, Bin X, Ping Z, Liang S (2019) Oxidative stress injury in doxorubicin-induced cardiotoxicity. Toxicol Lett 307:41–48. https://doi.org/10.1016/j.toxlet.2019.02.013
Sonowal H, Pal P, Shukla K, Saxena A, Srivastava SK, Ramana KV (2018) Aldose reductase inhibitor, fidarestat prevents doxorubicin-induced endothelial cell death and dysfunction. Biochem Pharmacol 150:181–190. https://doi.org/10.1016/j.bcp.2018.02.018
Soulet F, Bodineau C, Hooks KB, Descarpentrie J, Alves I, Dubreuil M, Mouchard A, Eugenie M, Hoepffner JL, López JJ, Rosado JA, Soubeyran I, Tomé M, Durán RV, Nikolski M, Villoutreix BO, Evrard S, Siegfried G, Khatib AM (2020) ELA/APELA precursor cleaved by furin displays tumor suppressor function in renal cell carcinoma through mTORC1 activation. JCI Insight 5:e129070. https://doi.org/10.1172/jci.insight.129070
Speth PA, van Hoesel QG, Haanen C (1988) Clinical pharmacokinetics of doxorubicin. Clin Pharmacokinet 15:15–31. https://doi.org/10.2165/00003088-198815010-00002
Steinberg JS, Cohen AJ, Wasserman AG, Cohen P, Ross AM (1987) Acute arrhythmogenicity of doxorubicin administration. Cancer 60:1213–1218. https://doi.org/10.1002/1097-0142(19870915)60:6%3c1213::aid-cncr2820600609%3e3.0.co;2-v
Swain SM, Whaley FS, Ewer MS (2003) Congestive heart failure in patients treated with doxorubicin: a retrospective analysis of three trials. Cancer 97:2869–2879. https://doi.org/10.1002/cncr.11407
Wallace KB, Sardão VA, Oliveira PJ (2020) Mitochondrial determinants of doxorubicin-induced cardiomyopathy. Circ Res 126:926–941. https://doi.org/10.1161/CIRCRESAHA.119.314681
Wang Z, Yu D, Wang M, Wang Q, Kouznetsova J, Yang R, Qian K, Wu W, Shuldiner A, Sztalryd C, Zou M, Zheng W, Gong DW (2015) Elabela-apelin receptor signaling pathway is functional in mammalian systems. Sci Rep 5:8170. https://doi.org/10.1038/srep08170
Wang B, Xu H, Kong J, Liu D, Qin W, Bai W (2021) Krüppel-like factor 15 reduces ischemia-induced apoptosis involving regulation of p38/MAPK signaling. Hum Gene Ther 32:1471–1480. https://doi.org/10.1089/hum.2021.075
Xu R, Zhang ZZ, Chen LJ, Yu HM, Guo SJ, Xu YL, Oudit GY, Zhang Y, Chang Q, Song B, Chen DR, Zhu DL, Zhong JC (2016) Ascending aortic adventitial remodeling and fibrosis are ameliorated with Apelin-13 in rats after TAC via suppression of the miRNA-122 and LGR4-β-catenin signaling. Peptides 86:85–94. https://doi.org/10.1016/j.peptides.2016.10.005
Xue CC, Li MH, Zhao Y, Zhou J, Hu Y, Cai KY, Zhao Y, Yu SH, Luo Z (2020) Tumor microenvironment-activatable Fe-doxorubicin preloaded amorphous CaCO3 nanoformulation triggers ferroptosis in target tumor cells. Sci Adv 6:eaax1346. https://doi.org/10.1126/sciadv.aax1346
Yu Y, Yan Y, Niu F, Wang Y, Chen X, Su G, Liu Y, Zhao X, Qian L, Liu P, Xiong Y (2021) Ferroptosis: a cell death connecting oxidative stress, inflammation and cardiovascular diseases. Cell Death Discov 7:193. https://doi.org/10.1038/s41420-021-00579-w
Zhang X, Zhu JX, Ma ZG, Wu HM, Xu SC, Song P, Kong CY, Yuan YP, Deng W, Tang QZ (2019) Rosmarinic acid alleviates cardiomyocyte apoptosis via cardiac fibroblast in doxorubicin-induced cardiotoxicity. Int J Biol Sci 15:556–567. https://doi.org/10.7150/ijbs.29907
Zhang X, Hu C, Kong CY, Song P, Wu HM, Xu SC, Yuan YP, Deng W, Ma ZG, Tang QZ (2020) FNDC5 alleviates oxidative stress and cardiomyocyte apoptosis in doxorubicin-induced cardiotoxicity via activating AKT. Cell Death Differ 27:540–555. https://doi.org/10.1038/s41418-019-0372-z
Zhao L, Tao X, Qi Y, Xu L, Yin L, Peng J (2018) Protective effect of dioscin against doxorubicin-induced cardiotoxicity via adjusting microRNA-140-5p-mediated myocardial oxidative stress. Redox Biol 16:189–198. https://doi.org/10.1016/j.redox.2018.02.026
Zhao Y, Song W, Wang L, Rane MJ, Han F, Cai L (2019) Multiple roles of KLF15 in the heart: underlying mechanisms and therapeutic implications. J Mol Cell Cardiol 129:193–196. https://doi.org/10.1016/j.yjmcc.2019.01.024
Zheng X, Zhong T, Ma Y, Wan X, Qin A, Yao B, Zou H, Song Y, Yin D (2020) Bnip3 mediates doxorubicin-induced cardiomyocyte pyroptosis via caspase-3/GSDME. Life Sci 242:117186. https://doi.org/10.1016/j.lfs.2019.117186
Zhou M, Wu Y (2022) Effects and signaling pathways of Elabela in the cardiovascular system. Peptides 147:170674. https://doi.org/10.1016/j.peptides.2021.170674
Zhou L, Han Y, Yang Q, Xin B, Chi M, Huo Y, Guo C, Sun X (2022) Scutellarin attenuates doxorubicin-induced oxidative stress, DNA damage, mitochondrial dysfunction, apoptosis and autophagy in H9c2 cells, cardiac fibroblasts and HUVECs. Toxicol in Vitro 82:105366. https://doi.org/10.1016/j.tiv.2022.105366
Acknowledgements
This study was supported by the General Program and the National Major Research Plan Training Program of the National Natural Science Foundation of China (No. 92168117; 81770253; 91849111), Beijing Natural Science Foundation (7222068), Clinical Research Incubation Program of Beijing Chaoyang Hospital Affiliated to Capital Medical University (CYFH202209) and Reform and Development Program of Beijing Institute of Respiratory Medicine (ysrh2022002). We appreciate all graduate students and the doctors who participated in this study.
Funding
This study was supported by the General Program and the National Major Research Plan Training Program of the National Natural Science Foundation of China (No. 92168117; 81770253; 91849111), Beijing Natural Science Foundation (7222068), Clinical Research Incubation Program of Beijing Chaoyang Hospital Affiliated to Capital Medical University (CYFH202209) and Reform and Development Program of Beijing Institute of Respiratory Medicine (ysrh2022002).
Author information
Authors and Affiliations
Contributions
Mi-Wen Zhang and Jiu-Chang Zhong: writing—original draft, methodology, supervision, writing—review and editing, read and approved the final manuscript. Xue-Ting Li, Zhen-Zhou Zhang, and Ying Liu: collected and recorded the samples, methodology, read and approved the final manuscript. Jia-Wei Song and Xin-Ming Liu: collected and recorded the samples, read and approved the final manuscript. Yihang Chen, Ning Wang, and Ying Guo: formal analysis, read and approved the final manuscript. Li-Rong Liang and Jiu-Chang Zhong: Writing—review and editing, supervision, methodology, read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, MW., Li, XT., Zhang, ZZ. et al. Elabela blunts doxorubicin-induced oxidative stress and ferroptosis in rat aortic adventitial fibroblasts by activating the KLF15/GPX4 signaling. Cell Stress and Chaperones 28, 91–103 (2023). https://doi.org/10.1007/s12192-022-01317-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-022-01317-6