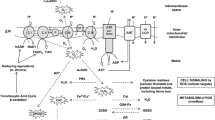
Abstract
The poultry industry plays a significant role in boosting the economy of several countries, particularly developing countries, and acts as a good, cheap, and affordable source of animal protein. A stress-free environment is the main target in poultry production. There are several stressors, such as cold stress, heat stress, high stocking density, and diseases that can affect birds and cause several deleterious changes. Stress reduces feed intake and growth, as well as impairs immune response and function, resulting in high disease susceptibility. These effects are correlated with higher corticosteroid levels that modulate several immune pathways such as cytokine-cytokine receptor interaction and Toll-like receptor signaling along with induction of excessive production of reactive oxygen species (ROS) and thus oxidative stress. Several approaches have been considered to boost bird immunity to overcome stress-associated effects. Of these, dietary supplementation of certain nutrients and management modifications, such as light management, are commonly considered. Dietary supplementations improve bird immunity by improving the development of lymphoid tissues and triggering beneficial immune modulators and responses. Since nano-minerals have higher bioavailability compared to inorganic or organic forms, they are highly recommended to be included in the bird’s diet during stress. Additionally, light management is considered a cheap and safe approach to control stress. Changing light from continuous to intermittent and using monochromatic light instead of the normal light improve bird performance and health. Such changes in light management are associated with a reduction of ROS production and increased antioxidant production. In this review, we discuss the impact of stress on the immune system of birds and the transcriptome of oxidative stress and immune-related genes, in addition, how nano-minerals supplementations and light system modulate or mitigate stress-associated effects.


Similar content being viewed by others
Data Availability
Not applicable
References
Abdo SE, El-Kassas S, El-Nahas AF, Mahmoud S (2017) Modulatory effect of monochromatic blue light on heat stress response in commercial broilers. Oxidative Med Cell Longev 2017:1351945. https://doi.org/10.1155/2017/1351945
Abo-Al-Ela HG (2018) An introduction to selected innate immune-relevant genes in fish. Appl Ecol Env Res 16:955–976. https://doi.org/10.15666/aeer/1602_955976
Abo-Al-Ela HG, Burgos-Aceves MA (2021) Exploring the role of microRNAs in axolotl regeneration. J Cell Physiol 236:839–850. https://doi.org/10.1002/jcp.29920
Abo-Al-Ela HG, Faggio C (2021) MicroRNA-mediated stress response in bivalve species. Ecotoxicol Environ Saf 208:111442. https://doi.org/10.1016/j.ecoenv.2020.111442
Ahmadi M, Ahmadian A, Seidavi A (2018) Effect of different levels of nano-selenium on performance, blood parameters, immunity and carcass characteristics of broiler chickens. Poult Sci J 6:99–108. https://doi.org/10.22069/psj.2018.13815.1276
Ali A, Soliman E, Hamad R, El-Borad O, Hassan R, Helal M (2020) Preventive, behavioral, productive, and tissue modification using green synthesized selenium nanoparticles in the drinking water of two broiler breeds under microbial stress. Braz J Poult Sci 22:eRBCA-2019-1129. https://doi.org/10.1590/1806-9061-2019-1129
Alvino GM, Archer GS, Mench JA (2009) Behavioural time budgets of broiler chickens reared in varying light intensities. Appl Anim Behav Sci 118:54–61. https://doi.org/10.1016/j.applanim.2009.02.003
Archer G (2018) Color temperature of light-emitting diode lighting matters for optimum growth and welfare of broiler chickens. Animal 12:1015–1021. https://doi.org/10.1017/S1751731117002361
Beere HM, Wolf BB, Cain K, Mosser DD, Mahboubi A, Kuwana T, Tailor P, Morimoto RI, Cohen GM, Green DR (2000) Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nat Cell Biol 2:469–475. https://doi.org/10.1038/35019501
Bellezza I, Giambanco I, Minelli A, Donato R (2018) Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res 1865:721–733. https://doi.org/10.1016/j.bbamcr.2018.02.010
Berenjian A, Sharifi SD, Mohammadi-Sangcheshmeh A, Ghazanfari S (2018) Effect of chromium nanoparticles on physiological stress induced by exogenous dexamethasone in Japanese quails. Biol Trace Elem Res 184:474–481. https://doi.org/10.1007/s12011-017-1192-y
Bhakkiyalakshmi E, Sireesh D, Rajaguru P, Paulmurugan R, Ramkumar KM (2015) The emerging role of redox-sensitive Nrf2–Keap1 pathway in diabetes. Pharmacol Res 91:104–114. https://doi.org/10.1016/j.phrs.2014.10.004
Blair R, Newberry RC, Gardiner EE (1993) Effects of lighting pattern and dietary tryptophan supplementation on growth and mortality in broilers. Poult Sci 72:495–502. https://doi.org/10.3382/ps.0720495
Blatchford R, Klasing K, Shivaprasad H, Wakenell P, Archer G, Mench J (2009) The effect of light intensity on the behavior, eye and leg health, and immune function of broiler chickens. Poult Sci 88:20–28. https://doi.org/10.3382/ps.2008-00177
Blatchford R, Archer G, Mench J (2012) Contrast in light intensity, rather than day length, influences the behavior and health of broiler chickens. Poult Sci 91:1768–1774. https://doi.org/10.3382/ps.2011-02051
Boostani A, Sadeghi AA, Mousavi SN, Chamani M, Kashan N (2015) Effects of organic, inorganic, and nano-Se on growth performance, antioxidant capacity, cellular and humoral immune responses in broiler chickens exposed to oxidative stress. Livest Sci 178:330–336. https://doi.org/10.1016/j.livsci.2015.05.004
Borges T, Wieten L, van Herwijnen M, Broere F, Van Der Zee R, Bonorino C, Van Eden W (2012) The anti-inflammatory mechanisms of Hsp70. Front Immunol 3. https://doi.org/10.3389/fimmu.2012.00095
Burgos-Aceves MA, Abo-Al-Ela HG, Faggio C (2021) Physiological and metabolic approach of plastic additive effects: immune cells responses. J Hazard Mater 404:124114. https://doi.org/10.1016/j.jhazmat.2020.124114
Buzea C, Pacheco II, Robbie K (2007) Nanomaterials and nanoparticles: sources and toxicity. Biointerphases 2:MR17–MR71. https://doi.org/10.1116/1.2815690
Cai SJ, Wu CX, Gong LM, Song T, Wu H, Zhang LY (2012) Effects of nano-selenium on performance, meat quality, immune function, oxidation resistance, and tissue selenium content in broilers. Poult Sci J 91:2532–2539. https://doi.org/10.3382/ps.2012-02160
Campo J, Gil M, Davila S, Munoz I (2005) Influence of perches and footpad dermatitis on tonic immobility and heterophil to lymphocyte ratio of chickens. Poult Sci 84:1004–1009. https://doi.org/10.1093/ps/84.7.1004
Cao J, Liu W, Wang Z, Xie D, Jia L, Chen Y (2008) Green and blue monochromatic lights promote growth and development of broilers via stimulating testosterone secretion and myofiber growth. J Appl Poult Res 17:211–218. https://doi.org/10.3382/japr.2007-00043
Chen H, Weiss J, Shahidi F (2006) Nanotechnology in nutraceuticals and functional foods. Food Technol 3:30–36
Chen X, Li R, Geng Z (2015) Cold stress initiates the Nrf2/UGT1A1/L-FABP signaling pathway in chickens. Poult Sci 94:2597–2603. https://doi.org/10.3382/ps/pev253
Chen M, Li X, Shi Q, Zhang Z, Xu S (2019) Hydrogen sulfide exposure triggers chicken trachea inflammatory injury through oxidative stress-mediated FOS/IL8 signaling. J Hazard Mater 368:243–254. https://doi.org/10.1016/j.jhazmat.2019.01.054
Compton MM, Gibbs PS, Johnson LR (1990a) Glucocorticoid activation of deoxyribonucleic acid degradation in bursal lymphocytes. Poult Sci 69:1292–1298. https://doi.org/10.3382/ps.0691292
Compton MM, Gibbs PS, Swicegood LR (1990b) Glucocorticoid-mediated activation of DNA degradation in avian lymphocytes. Gen Comp Endocrinol 80:68–79. https://doi.org/10.1016/0016-6480(90)90149-g
Dawood MAO, Abo-Al-Ela HG, Hasan MT (2020) Modulation of transcriptomic profile in aquatic animals: probiotics, prebiotics and synbiotics scenarios. Fish Shellfish Immunol 97:268–282. https://doi.org/10.1016/j.fsi.2019.12.054
Deep A, Schwean-Lardner K, Crowe TG, Fancher BI, Classen HL (2012) Effect of light intensity on broiler behaviour and diurnal rhythms. Appl Anim Behav Sci 136:50–56. https://doi.org/10.1016/j.applanim.2011.11.002
Deep A, Raginski C, Schwean-Lardner K, Fancher B, Classen H (2013) Minimum light intensity threshold to prevent negative effects on broiler production and welfare. Br Poult Sci 54:686–694. https://doi.org/10.1080/00071668.2013.847526
Dietert RR, Golemboski KA, Austic RE (1994) Environment-immune interactions. Poult Sci 73:1062–1076. https://doi.org/10.3382/ps.0731062
Dohms JE, Metz A (1991) Stress—mechanisms of immunosuppression. Vet Immunol Immunopathol 30:89–109. https://doi.org/10.1016/0165-2427(91)90011-z
El Sabry MI, McMillin KW, Sabliov CM (2018) Nano-technology considerations for poultry and livestock production systems – a review. Ann Anim Sci 18:319–334. https://doi.org/10.1515/aoas-2017-0047
El-Deep MH, Ijiri D, Ebeid TA, Ohtsuka A (2016) Effects of dietary nano-selenium supplementation on growth performance, antioxidative status, and immunity in broiler chickens under thermoneutral and high ambient temperature conditions. J Poult Sci 53:274–283. https://doi.org/10.2141/jpsa.0150133
El-Deep MH, Shabaan M, Assar MH, Attia KM, Sayed MAM (2017) Comparative effects of different dietary selenium sources on productive performance, antioxidative properties and immunity in local laying hens exposed to high ambient temperature. J Animal and Poultry Prod 9:335–343. https://doi.org/10.21608/JAPPMU.2017.45998
El-Kassas S, Abdo SE, El-Naggar K, Abdo W, Kirrella AAK, Nashar TO (2018) Ameliorative effect of dietary supplementation of copper oxide nanoparticles on inflammatory and immune responses in commercial broiler under normal and heat-stress housing conditions. J Therm Biol 78:235–246. https://doi.org/10.1016/j.jtherbio.2018.10.009
El-Kassas S, El-Naggar K, Abdo SE, Abdo W, Kirrella AAK, El-Mehaseeb I, Abu El-Magd M (2020) Dietary supplementation with copper oxide nanoparticles ameliorates chronic heat stress in broiler chickens. Anim Prod Sci 60:254–268. https://doi.org/10.1071/AN18270
El-Katcha M, Soltan MA, El-badry M (2017) Effect of dietary replacement of inorganic zinc by organic or nanoparticles sources on growth performance, immune response and intestinal histopathology of broiler chicken. Alex J Vet Sci 55:129–145. https://doi.org/10.5455/ajvs.266925
El-katcha MI, Soltan MA, Arafa MM, El-Naggar K, Kawarei E-SR (2018) Impact of dietary replacement of inorganic zinc by organic or nano sources on productive performance, immune response and some blood biochemical constituents of laying hens. Alex J Vet Sci 59:48–59. https://doi.org/10.5455/ajvs.301885
El-Katcha MI, Soltan MA, Khalifa E, Fadl SE, Hassan A, El-Shimey OK, El-Naggar K (2020) Growth and immune response of broiler chicks fed on oxidized oil containing diets and supplemented with different copper sources and levels. Alex J Vet Sci 66:15–26. https://doi.org/10.5455/ajvs.98495
El-Kazaz SE, Hafez MH (2020) Evaluation of copper nanoparticles and copper sulfate effect on immune status, behavior, and productive performance of broilers. J Adv Vet Anim Res 7:16–25. https://doi.org/10.5455/javar.2020.g388
El-Lethey H, Huber-Eicher B, Jungi TW (2003) Exploration of stress-induced immunosuppression in chickens reveals both stress-resistant and stress-susceptible antigen responses. Vet Immunol Immunopathol 95:91–101. https://doi.org/10.1016/s0165-2427(02)00308-2
El-Naggar K, El-Kassas S, Abdo SE, Kirrella AAK, Al Wakeel RA (2019) Role of gamma-aminobutyric acid in regulating feed intake in commercial broilers reared under normal and heat stress conditions. J Therm Biol 84:164–175. https://doi.org/10.1016/j.jtherbio.2019.07.004
Failla ML (2003) Trace elements and host defense: recent advances and continuing challenges. J Nutr 133:1443s–1447s. https://doi.org/10.1093/jn/133.5.1443S
Fair J, Ricklefs R (2002) Physiological, growth, and immune responses of Japanese quail chicks to the multiple stressors of immunological challenge and lead shot. Arch Environ Contam Toxicol 42:77–87. https://doi.org/10.1007/s002440010294
Fan Q et al (2020) Influence of mushroom polysaccharide, nano-copper, copper loaded chitosan, and lysozyme on intestinal barrier and immunity of LPS-mediated yellow-feathered chickens. Animals 10:594. https://doi.org/10.3390/ani10040594
Fang J, Zheng Z, Yang Z, Peng X, Zuo Z, Cui H, Ouyang P, Shu G, Chen Z, Huang C (2018) Ameliorative effects of selenium on the excess apoptosis of the jejunum caused by AFB1 through death receptor and endoplasmic reticulum pathways. Toxicol Res 7:1108–1119. https://doi.org/10.1039/c8tx00068a
Fidan ED, Turkyilmaz MK, Nazligul A (2015) Effects of noise and light intensities on stress and fear reactions in broilers. Indian J Anim Sci 85:1375–1378
Fowles JR, Fairbrother A, Fix M, Schiller S, Kerkvliet NI (1993) Glucocorticoid effects on natural and humoral immunity in mallards. Dev Comp Immunol 17:165–177. https://doi.org/10.1016/0145-305X(93)90026-M
Freeman B (1987) The stress syndrome. Worlds Poult Sci J 43:15–19. https://doi.org/10.1079/WPS19870002
Genestra M (2007) Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell Signal 19:1807–1819. https://doi.org/10.1016/j.cellsig.2007.04.009
Gomes A et al (2014) Overcrowding stress decreases macrophage activity and increases Salmonella Enteritidis invasion in broiler chickens. Avian Pathol 43:82–90. https://doi.org/10.1080/03079457.2013.874006
Gongruttananun N (2011) Influence of red light on reproductive performance, eggshell ultrastructure, and eye morphology in Thai-native hens. Poult Sci 90:2855–2863. https://doi.org/10.3382/ps.2011-01652
Gopi M, Pearlin B, Kumar RD, Shanmathy M, Prabakar G (2017) Role of nanoparticles in animal and poultry nutrition: Modes of action and applications in formulating feed additives and food processing. Int J Pharmacol 13:724–731. https://doi.org/10.3923/ijp.2017.724.731
Grüngreiff K, Reinhold D, Wedemeyer H (2016) The role of zinc in liver cirrhosis. Ann Hepatol 15:7–16. https://doi.org/10.5604/16652681.1184191
Gu XH, Hao Y, Wang XL (2012) Overexpression of heat shock protein 70 and its relationship to intestine under acute heat stress in broilers: 2. Intestinal oxidative stress1. Poult Sci 91:790–799. https://doi.org/10.3382/ps.2011-01628
Guo Y, Ma S, Du J, Chen J (2018) Effects of light intensity on growth, anti-stress ability and immune function in yellow feathered broilers. Braz J Poult Sci 20:79–84. https://doi.org/10.1590/1806-9061-2017-0542
Guo Y, Jiang R, Su A, Tian H, Zhang Y, Li W, Tian Y, Li K, Sun G, Han R, Yan F, Kang X (2020) Identification of genes related to effects of stress on immune function in the spleen in a chicken stress model using transcriptome analysis. Mol Immunol 124:180–189. https://doi.org/10.1016/j.molimm.2020.06.004
Gupta A (2011) Ascites syndrome in poultry: a review. Worlds Poult Sci J 67:457–468. https://doi.org/10.1017/S004393391100050X
Hafez A, Nassef E, Fahmy M, Elsabagh M (2020) Impact of dietary nano-zinc oxide on immune response and antioxidant defense of broiler chickens. Environ Sci Pollut Res 27:19108–19114. https://doi.org/10.1007/s11356-019-04344-6
Hajializadeh F, Ghahri H, Talebi A (2017) Effects of supplemental chromium picolinate and chromium nanoparticles on performance and antibody titers of infectious bronchitis and avian influenza of broiler chickens under heat stress condition. Vet Res Forum 8:259–264
Hamal K, Burgess SC, Pevzner I, Erf G (2006) Maternal antibody transfer from dams to their egg yolks, egg whites, and chicks in meat lines of chickens. Poult Sci 85:1364–1372. https://doi.org/10.1093/ps/85.8.1364
Hamidi O, Mohammad C, Hasan G, Ali A, Hassan M (2016) Effects of chromium (III) picolinate and chromium (III) picolinate nanoparticles supplementation on growth performance, organs weight and immune function in cyclic heat stressed broiler chickens. Kafkas Univ Vet Fak Derg 1:10–16. https://doi.org/10.9775/kvfd.2015.14736
Hamidi O, Chamani M, Ghahri H, Sadeghi AA, Malekinejad H, Palangi V (2021) Effects of supplemental chromium nanoparticles on IFN-γ expression of heat stress broilers. Biol Trace Elem Res. https://doi.org/10.1007/s12011-021-02634-0
Hangalapura BN (2006) Cold stress and immunity: do chickens adapt to cold by trading-off immunity for thermoregulation? PhD Thesis. Wageningen Institute of Animal Sciences, Wageningen University and Research Centre.
Hangalapura BN, Nieuwland MG, de Vries RG, Heetkamp MJ, Van den Brand H, Kemp B, Parmentier HK (2003) Effects of cold stress on immune responses and body weight of chicken lines divergently selected for antibody responses to sheep red blood cells. Poult Sci 82:1692–1700. https://doi.org/10.1093/ps/82.11.1692
Hangalapura BN, Nieuwland MGB, Buyse J, Kemp B, Parmentier HK (2004) Effect of duration of cold stress on plasma adrenal and thyroid hormone levels and immune responses in chicken lines divergently selected for antibody responses. Poult Sci 83:1644–1649. https://doi.org/10.1093/ps/83.10.1644
Hassan RA, Soliman ES, Hamad RT, El-Borady OM, Ali AA, Helal MS (2020a) Selenium and nano-selenium ameliorations in two breeds of broiler chickens exposed to heat stress. South Afr J Anim Sci 50:215–232. https://doi.org/10.4314/sajas.v50i2.5
Hassan S, Hassan F-U, Rehman MS-U (2020b) Nano-particles of trace minerals in poultry nutrition: potential applications and future prospects. Biol Trace Elem Res 195:591–612. https://doi.org/10.1007/s12011-019-01862-9
Hayward LS, Wingfield JC (2004) Maternal corticosterone is transferred to avian yolk and may alter offspring growth and adult phenotype. Gen Comp Endocrinol 135:365–371. https://doi.org/10.1016/j.ygcen.2003.11.002
Heckert RA, Estevez I, Russek-Cohen E, Pettit-Riley R (2002) Effects of density and perch availability on the immune status of broilers. Poult Sci 81:451–457. https://doi.org/10.1093/ps/81.4.451
Hofmann T, Schmucker SS, Bessei W, Grashorn M, Stefanski V (2020) Impact of housing environment on the immune system in chickens: a review. Animals 10:1138. https://doi.org/10.3390/ani10071138
Huff GR, Huff WE, Rath NC, de Los Santos FS, Farnell MB, Donoghue AM (2007) Influence of hen age on the response of turkey poults to cold stress, Escherichia coli challenge, and treatment with a yeast extract antibiotic alternative. Poult Sci 86:636–642. https://doi.org/10.1093/ps/86.4.636
Ishii T, Itoh K, Takahashi S, Sato H, Yanagawa T, Katoh Y, Bannai S, Yamamoto M (2000) Transcription factor Nrf2 coordinately regulates a group of oxidative stress-inducible genes in macrophages. J Biol Chem 275:16023–16029. https://doi.org/10.1074/jbc.275.21.16023
Isobe T, Lillehoj HS (1992) Effects of corticosteroids on lymphocyte subpopulations and lymphokine secretion in chickens. Avian Dis 36:590–596. https://doi.org/10.2307/1591753
Itoh K, Wakabayashi N, Katoh Y, Ishii T, Igarashi K, Engel JD, Yamamoto M (1999) Keap1 represses nuclear activation of antioxidant responsive elements by Nrf2 through binding to the amino-terminal Neh2 domain. Genes Dev 13:76–86. https://doi.org/10.1101/gad.13.1.76
Itoh K, Mimura J, Yamamoto M (2010) Discovery of the negative regulator of Nrf2, Keap1: a historical overview. Antioxid Redox Signal 13:1665–1678. https://doi.org/10.1089/ars.2010.3222
Jankowski J, Otowski K, Kozłowski K, Pietrzak P, Ferenc K, Ognik K, Juśkiewicz J, Sawosz E, Zduńczyk Z (2020) Effect of different levels of copper nanoparticles and copper sulfate on morphometric indices, antioxidant status and mineral digestibility in the small intestine of turkeys. Ann Anim Sci 20:975–990. https://doi.org/10.2478/aoas-2020-0013
Janský L, Pospíšilová D, Honzova S, Uličný B, Šrámek P, Zeman V, Kaminkova J (1996) Immune system of cold-exposed and cold-adapted humans. Eur J Appl Physiol Occup Physiol 72:445–450. https://doi.org/10.1007/BF00242274
Janus P, Pakuła-Cis M, Kalinowska-Herok M, Kashchak N, Szołtysek K, Pigłowski W, Widlak W, Kimmel M, Widlak P (2011) NF-κB signaling pathway is inhibited by heat shock independently of active transcription factor HSF1 and increased levels of inducible heat shock proteins. Genes to Cells 16:1168–1175. https://doi.org/10.1111/j.1365-2443.2011.01560.x
Jiang J, Pi J, Cai J (2018) The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg Chem Appl 2018:1062562. https://doi.org/10.1155/2018/1062562
Jiang S, Mohammed AA, Jacobs JA, Cramer TA, Cheng HW (2020) Effect of synbiotics on thyroid hormones, intestinal histomorphology, and heat shock protein 70 expression in broiler chickens reared under cyclic heat stress. Poult Sci 99:142–150. https://doi.org/10.3382/ps/pez571
Kaiser P, Wu Z, Rothwell L, Fife M, Gibson M, Poh TY, Shini A, Bryden W, Shini S (2009) Prospects for understanding immune-endocrine interactions in the chicken. Gen Comp Endocrinol 163:83–91. https://doi.org/10.1016/j.ygcen.2008.09.013
Kamal R, Dey A, Mondal KG, Chandran PC (2018) Impact of environmental stressors on the performance of backyard poultry. Proc Natl Acad Sci, India, Sect B Biol Sci 88:79–84. https://doi.org/10.1007/s40011-016-0741-z
Kaspers B, Schat KA (2012) Avian immunology (Second Edition). Academic Press, Boston. https://doi.org/10.1016/C2011-0-07243-5
Ke YY, Liu WJ, Wang ZX, Chen JL (2011) Effects of monochromatic light on quality properties and antioxidation of meat in broilers. Poult Sci 90:2632–2637. https://doi.org/10.3382/ps.2011-01523
Keum Y-S, Choi BY (2014) Molecular and chemical regulation of the Keap1-Nrf2 signaling pathway. Molecules 19:10074–10089. https://doi.org/10.3390/molecules190710074
Kidd MT (2004) Nutritional modulation of immune function in broilers. Poult Sci 83:650–657. https://doi.org/10.1093/ps/83.4.650
Kjaer JB, Vestergaard K (1999) Development of feather pecking in relation to light intensity. Appl Anim Behav Sci 62:243–254. https://doi.org/10.1016/S0168-1591(98)00217-2
Klasing KC (2007) Nutrition and the immune system. Br Poult Sci 48:525–537. https://doi.org/10.1080/00071660701671336
Kliger CA, Gehad AE, Hulet RM, Roush WB, Lillehoj HS, Mashaly MM (2000) Effects of photoperiod and melatonin on lymphocyte activities in male broiler chickens. Poult Sci 79:18–25. https://doi.org/10.1093/ps/79.1.18
Koutsos EA, Klasing KC (2014) Chapter 17 - factors modulating the avian immune system. In: Schat KA, Kaspers B, Kaiser P (eds) Avian Immunology, 2nd edn. Academic Press, Boston, pp 299–313. https://doi.org/10.1016/B978-0-12-396965-1.00017-0
Lauridsen C (2019) From oxidative stress to inflammation: redox balance and immune system. Poult Sci 98:4240–4246. https://doi.org/10.3382/ps/pey407
Lechner O, Dietrich H, Wiegers GJ, Vacchio M, Wick G (2001) Glucocorticoid production in the chicken bursa and thymus. Int Immunol 13:769–776. https://doi.org/10.1093/intimm/13.6.769
Lee KA, Wikelski M, Robinson WD, Robinson TR, Klasing KC (2008) Constitutive immune defences correlate with life-history variables in tropical birds. J Anim Ecol 77:356–363. https://doi.org/10.1111/j.1365-2656.2007.01347.x
Lewis PD, Morris TR (2000) Poultry and coloured light. Worlds Poult Sci J 56:189–207. https://doi.org/10.1079/Wps20000015
Li Q, Zhang M, Chen Y-J, Wang Y-J, Huang F, Liu J (2011) Oxidative damage and HSP70 expression in masseter muscle induced by psychological stress in rats. Physiol Behav 104:365–372. https://doi.org/10.1016/j.physbeh.2011.04.012
Li S, Muhammad I, Yu H, Sun X, Zhang X (2019) Detection of aflatoxin adducts as potential markers and the role of curcumin in alleviating AFB1-induced liver damage in chickens. Ecotoxicol Environ Saf 176:137–145. https://doi.org/10.1016/j.ecoenv.2019.03.089
Li G-M, Liu LP, Yin B, Liu YY, Dong WW, Gong S, Zhang J, Tan JH (2020) Heat stress decreases egg production of laying hens by inducing apoptosis of follicular cells via activating the FasL/Fas and TNF-α systems. Poult Sci 99:6084–6093. https://doi.org/10.1016/j.psj.2020.07.024
Lina T, Fenghua Z, Huiying R, Jianyang J, Wenli L (2009) Effects of nano-zinc oxide on antioxidant function in broilers. Chin J Anim Nutr 21:534–539
Liu SF, Malik AB (2006) NF-κB activation as a pathological mechanism of septic shock and inflammation. Am J Physiol Lung Cell Mol Physiol 290:L622–L645. https://doi.org/10.1152/ajplung.00477.2005
Liu ZH, Lu L, Li SF, Zhang LY, Xi L, Zhang KY, Luo XG (2011) Effects of supplemental zinc source and level on growth performance, carcass traits, and meat quality of broilers. Poult Sci 90:1782–1790. https://doi.org/10.3382/ps.2010-01215
Liu LL, He JH, Xie HB, Yang YS, Li JC, Zou Y (2014) Resveratrol induces antioxidant and heat shock protein mRNA expression in response to heat stress in black-boned chickens. Poult Sci 93:54–62. https://doi.org/10.3382/ps.2013-03423
Liu W, Yuan Y, Sun C, Balasubramanian B, Zhao Z, An L (2019) Effects of dietary betaine on growth performance, digestive function, carcass traits, and meat quality in indigenous yellow-feathered broilers under long-term heat stress. Animals 9:506. https://doi.org/10.3390/ani9080506
Lushchak VI (2011) Adaptive response to oxidative stress: Bacteria, fungi, plants and animals. Comp Biochem Physiol C-Pharmacol Toxicol Endocrinol 153:175–190. https://doi.org/10.1016/j.cbpc.2010.10.004
Lyu Q, Wawrzyniuk M, Rutten VPMG, van Eden W, Sijts AJAM, Broere F (2020) Hsp70 and NF-kB mediated control of innate inflammatory responses in a canine macrophage cell line. Int J Mol Sci 21:6464. https://doi.org/10.3390/ijms21186464
Ma Q, He X (2012) Molecular basis of electrophilic and oxidative defense: promises and perils of Nrf2. Pharmacol Rev 64:1055–1081. https://doi.org/10.1124/pr.110.004333
Ma Y, Zheng YX, Dong XY, Zou XT (2018a) Effect of mercury chloride on oxidative stress and nuclear factor erythroid 2-related factor 2 signalling molecule in liver and kidney of laying hens. J Anim Physiol Anim Nutr 102:1199–1209. https://doi.org/10.1111/jpn.12920
Ma Y, Zhu M, Miao L, Zhang X, Dong X, Zou X (2018b) Mercuric chloride induced ovarian oxidative stress by suppressing Nrf2-Keap1 signal pathway and its downstream genes in laying hens. Biol Trace Elem Res 185:185–196. https://doi.org/10.1007/s12011-018-1244-y
Martin LB II, Gilliam J, Han P, Lee K, Wikelski M (2005) Corticosterone suppresses cutaneous immune function in temperate but not tropical house sparrows, Passer domesticus. Gen Comp Endocrinol 140:126–135. https://doi.org/10.1016/j.ygcen.2004.10.010
Martin LB, Weil ZM, Nelson RJ (2008) Seasonal changes in vertebrate immune activity: mediation by physiological trade-offs. Philos Trans R Soc Lond Ser B-Biol Sci 363:321–339. https://doi.org/10.1098/rstb.2007.2142
Mashaly MM, Trout JM, Hendricks G III, Al-Dokhi LM, Gehad A (1998) The role of neuroendocrine immune interactions in the initiation of humoral immunity in chickens. Domest Anim Endocrinol 15:409–422. https://doi.org/10.1016/s0739-7240(98)00023-x
McDowell LR (2003) Minerals in animal and human nutrition (Second Edition). Elsevier, Amsterdam. https://doi.org/10.1016/B978-0-444-51367-0.X5001-6
Meng T, Liu YL, Xie CY, Zhang B, Huang YQ, Zhang YW, Yao Y, Huang R, Wu X (2019) Effects of different selenium sources on laying performance, egg selenium concentration, and antioxidant capacity in laying hens. Biol Trace Elem Res 189:548–555. https://doi.org/10.1007/s12011-018-1490-z
Merrill L, Angelier F, O’Loghlen AL, Rothstein SI, Wingfield JC (2012) Sex-specific variation in brown-headed cowbird immunity following acute stress: a mechanistic approach. Oecologia 170:25–38. https://doi.org/10.1007/s00442-012-2281-4
Mohamed RA, Eltholth MM, El-Saidy NR (2014) Rearing broiler chickens under monochromatic blue light improve performance and reduce fear and stress during pre-slaughter handling and transportation. Biotechnol Anim Husb 30:457–471. https://doi.org/10.2298/BAH1403457M
Monson MS, Van Goor AG, Ashwell CM, Persia ME, Rothschild MF, Schmidt CJ, Lamont SJ (2018) Immunomodulatory effects of heat stress and lipopolysaccharide on the bursal transcriptome in two distinct chicken lines. BMC Genomics 19:643. https://doi.org/10.1186/s12864-018-5033-y
Monson MS, Van Goor AG, Persia ME, Rothschild MF, Schmidt CJ, Lamont SJ (2019) Genetic lines respond uniquely within the chicken thymic transcriptome to acute heat stress and low dose lipopolysaccharide. Sci Rep 9:13649. https://doi.org/10.1038/s41598-019-50051-0
Morimoto RI, Kroeger PE, Cotto JJ (1996) The transcriptional regulation of heat shock genes: a plethora of heat shock factors and regulatory conditions. In: Feige U, Yahara I, Morimoto RI, Polla BS (eds) Stress-Inducible Cellular Responses. Birkhäuser Basel, Basel, pp 139–163. https://doi.org/10.1007/978-3-0348-9088-5_10
Muhammad I, Wang X, Li S, Li R, Zhang X (2018) Curcumin confers hepatoprotection against AFB1-induced toxicity via activating autophagy and ameliorating inflammation involving Nrf2/HO-1 signaling pathway. Mol Biol Rep 45:1775–1785. https://doi.org/10.1007/s11033-018-4323-4
Mujahid A (2010) Acute cold-induced thermogenesis in neonatal chicks (Gallus gallus). Comp Biochem Physiol A-Mol Integr Physiol 156:34–41. https://doi.org/10.1016/j.cbpa.2009.12.004
Mumma JO, Thaxton JP, Vizzier-Thaxton Y, Dodson WL (2006) Physiological stress in laying hens. Poult Sci 85:761–769. https://doi.org/10.1093/ps/85.4.761
Najafi P, Zulkifli I, Amat Jajuli N, Farjam AS, Ramiah SK, Amir AA, O’Reily E, Eckersall D (2015) Environmental temperature and stocking density effects on acute phase proteins, heat shock protein 70, circulating corticosterone and performance in broiler chickens. Int J Biometeorol 59:1577–1583. https://doi.org/10.1007/s00484-015-0964-3
Nguyen T, Nioi P, Pickett CB (2009) The Nrf2-antioxidant response element signaling pathway and its activation by oxidative stress. J Biol Chem 284:13291–13295. https://doi.org/10.1074/jbc.R900010200
O’Neal DM, Ketterson ED (2012) Life-history evolution, hormones, and avian immune function. In: Demas GE, Nelson RJ (eds) Ecoimmunology. Oxford University Press, Inc., New York, NY, USA
Ognik K, Sembratowicz I, Cholewińska E, Jankowski J, Kozłowski K, Juśkiewicz J, Zduńczyk Z (2018) The effect of administration of copper nanoparticles to chickens in their drinking water on the immune and antioxidant status of the blood. Anim Sci J 89:579–588. https://doi.org/10.1111/asj.12956
Orhan C, Tuzcu M, Gencoglu H, Sahin N, Hayirli A, Sahin K (2013) Epigallocatechin-3-gallate exerts protective effects against heat stress through modulating stress-responsive transcription factors in poultry. Br Poult Sci 54:447–453. https://doi.org/10.1080/00071668.2013.806787
Osorio D, Vorobyev M, Jones C (1999) Colour vision of domestic chicks. J Exp Biol 202:2951–2959
Pamplona R, Costantini D (2011) Molecular and structural antioxidant defenses against oxidative stress in animals. Am J Physiol Regul Integr Comp Physiol 301:R843–R863. https://doi.org/10.1152/ajpregu.00034.2011
Pan JM, Yang YF, Yang B, Yu YH (2014) Artificial polychromatic light affects growth and physiology in chicks. Plos One 9:e113595. https://doi.org/10.1371/journal.pone.0113595
Paul S, Ghosh S, Mandal S, Sau S, Pal M (2018) NRF2 transcriptionally activates the heat shock factor 1 promoter under oxidative stress and affects survival and migration potential of MCF7 cells. J Biol Chem 293:19303–19316. https://doi.org/10.1074/jbc.RA118.003376
Pechova A, Pavlata L (2007) Chromium as an essential nutrient: a review. Vet Med 52:1–18. https://doi.org/10.17221/2010-VETMED
Peng L, Du W, Balhuizen MD, Haagsman HP, de Haan CAM, Veldhuizen EJA (2020a) Antiviral activity of chicken cathelicidin b1 against influenza A virus. Front Microbiol 11. https://doi.org/10.3389/fmicb.2020.00426
Peng L, Scheenstra MR, van Harten RM, Haagsman HP, Veldhuizen EJA (2020b) The immunomodulatory effect of cathelicidin-B1 on chicken macrophages. Vet Res 51:122. https://doi.org/10.1186/s13567-020-00849-y
Pirkkala L, Nykänen P, Sistonen L (2001) Roles of the heat shock transcription factors in regulation of the heat shock response and beyond. FASEB J 15:1118–1131. https://doi.org/10.1096/fj00-0294rev
Prescott N, Wathes CM, Jarvis J (2003) Light, vision and the welfare of poultry. Anim Welf 12:269–288
Quinteiro-Filho WM, Gomes AVS, Pinheiro ML, Ribeiro A, Ferraz-de-Paula V, Astolfi-Ferreira CS, Ferreira AJP, Palermo-Neto J (2012a) Heat stress impairs performance and induces intestinal inflammation in broiler chickens infected with Salmonella Enteritidis. Avian Pathol 41:421–427. https://doi.org/10.1080/03079457.2012.709315
Quinteiro-Filho WM, Rodrigues MV, Ribeiro A, Ferraz-de-Paula V, Pinheiro ML, Sá LRM, Ferreira AJP, Palermo-Neto J (2012b) Acute heat stress impairs performance parameters and induces mild intestinal enteritis in broiler chickens: role of acute hypothalamic-pituitary-adrenal axis activation. J Anim Sci 90:1986–1994. https://doi.org/10.2527/jas.2011-3949
Rahimi G, Rezaei M, Hafezian H, Saiyahzadeh H (2005) The effect of intermittent lighting schedule on broiler performance. Int J Poult Sci 4:396–398. https://doi.org/10.3923/ijps.2005.396.398
Ramiah SK, Awad EA, Mookiah S, Idrus Z (2019) Effects of zinc oxide nanoparticles on growth performance and concentrations of malondialdehyde, zinc in tissues, and corticosterone in broiler chickens under heat stress conditions. Poult Sci 98:3828–3838. https://doi.org/10.3382/ps/pez093
Rault J-L, Clark K, Groves PJ, Cronin GM (2017) Light intensity of 5 or 20 lux on broiler behavior, welfare and productivity. Poult Sci 96:779–787. https://doi.org/10.3382/ps/pew423
Regnier JA, Kelley KW (1981) Heat-and cold-stress suppresses in vivo and in vitro cellular immune responses of chickens. Am J Vet Res 42:294–299
Rimoldi S, Lasagna E, Sarti FM, Marelli SP, Cozzi MC, Bernardini G, Terova G (2015) Expression profile of six stress-related genes and productive performances of fast and slow growing broiler strains reared under heat stress conditions. Meta Gene 6:17–25. https://doi.org/10.1016/j.mgene.2015.08.003
Rizk YS, Ibrahim AF, Mansour MK, Mohamed HS, El-Slamony AE, Soliman AAM (2017) Effect of dietary source of selenium on productive and reproductive performance of Sinai laying hens under heat stress conditions. Egypt Poult Sci 37:461–489
Roushdy EM, Zaglool AW, Hassan FAM (2020) Thermal stress consequences on growth performance, immunological response, antioxidant status, and profitability of finishing broilers: transcriptomic profile change of stress-related genes. Trop Anim Health Prod 52:3685–3696. https://doi.org/10.1007/s11250-020-02405-4
Saelao P et al (2018) Integrated proteomic and transcriptomic analysis of differential expression of chicken lung tissue in response to NDV infection during heat stress. Genes 9:579. https://doi.org/10.3390/genes9120579
Safdari-Rostamabad M, Hosseini-Vashan SJ, Perai AH, Sarir H (2017) Nanoselenium supplementation of heat-stressed broilers: effects on performance, carcass characteristics, blood metabolites, immune response, antioxidant status, and jejunal morphology. Biol Trace Elem Res 178:105–116. https://doi.org/10.1007/s12011-016-0899-5
Sahin K, Orhan C, Tuzcu M, Ali S, Sahin N, Hayirli A (2010) Epigallocatechin-3-gallate prevents lipid peroxidation and enhances antioxidant defense system via modulating hepatic nuclear transcription factors in heat-stressed quails. Poult Sci 89:2251–2258. https://doi.org/10.3382/ps.2010-00749
Sahin K, Orhan C, Tuzcu M, Sahin N, Hayirli A, Bilgili S, Kucuk O (2016) Lycopene activates antioxidant enzymes and nuclear transcription factor systems in heat-stressed broilers. Poult Sci 95:1088–1095. https://doi.org/10.3382/ps/pew012
Sato T, Tezuka K, Shibuya H, Watanabe T, Kamata H, Shirai W (2002) Cold-induced ascites in broiler chickens and its improvement by temperature-controlled rearing. Avian Dis 46:989–996. https://doi.org/10.1637/0005-2086(2002)046[0989:CIAIBC]2.0.CO;2
Scanes CG (2016) Biology of stress in poultry with emphasis on glucocorticoids and the heterophil to lymphocyte ratio. Poult Sci 95:2208–2215. https://doi.org/10.3382/ps/pew137
Scott NR (2005) Nanotechnology and animal health. Rev Sci Tech 24:425–432. https://doi.org/10.20506/rst.24.1.1579
Scott A, Vadalasetty KP, Łukasiewicz M, Jaworski S, Wierzbicki M, Chwalibog A (2018) Effect of different levels of copper nanoparticles and copper sulphate on performance, metabolism and blood biochemical profiles in broiler chicken. J Anim Physiol Anim Nutr 102:e364–e373. https://doi.org/10.1111/jpn.12754
Senaratna D, Samarakone TS, Gunawardena WWDA (2016) Red color light at different intensities affects the performance, behavioral activities and welfare of broilers. Asian-Australas J Anim Sci 29:1052–1059. https://doi.org/10.5713/ajas.15.0757
Sesti-Costa R, Ignacchiti M, Chedraoui-Silva S, Marchi L, Mantovani B (2012) Chronic cold stress in mice induces a regulatory phenotype in macrophages: correlation with increased 11β-hydroxysteroid dehydrogenase expression. Brain Behav Immun 26:50–60. https://doi.org/10.1016/j.bbi.2011.07.234
Sharideh H, Zaghari M (2021) Effect of dietary L-tryptophan supplementation and light-emitting diodes on growth and immune response of broilers. Vet Res Forum 12:63–67. https://doi.org/10.30466/vrf.2019.96558.2310
Sharma MC, Joshi C, Pathak NN, Kaur H (2005) Copper status and enzyme, hormone, vitamin and immune function in heifers. Res Vet Sci 79:113–123. https://doi.org/10.1016/j.rvsc.2004.11.015
Shini S, Kaiser P (2009) Effects of stress, mimicked by administration of corticosterone in drinking water, on the expression of chicken cytokine and chemokine genes in lymphocytes. Stress 12:388–399. https://doi.org/10.1080/10253890802526894
Shini S, Kaiser P, Shini A, Bryden WL (2008a) Biological response of chickens (Gallus gallus domesticus) induced by corticosterone and a bacterial endotoxin. Comp Biochem Physiol B-Biochem Mol Biol 149:324–333. https://doi.org/10.1016/j.cbpb.2007.10.003
Shini S, Kaiser P, Shini A, Bryden WL (2008b) Differential alterations in ultrastructural morphology of chicken heterophils and lymphocytes induced by corticosterone and lipopolysaccharide. Vet Immunol Immunopathol 122:83–93. https://doi.org/10.1016/j.vetimm.2007.10.009
Shini S, Huff GR, Shini A, Kaiser P (2010a) Understanding stress-induced immunosuppression: exploration of cytokine and chemokine gene profiles in chicken peripheral leukocytes. Poult Sci 89:841–851. https://doi.org/10.3382/ps.2009-00483
Shini S, Shini A, Kaiser P (2010b) Cytokine and chemokine gene expression profiles in heterophils from chickens treated with corticosterone. Stress 13:185–194. https://doi.org/10.3109/10253890903144639
Siegel HS (1985) Immunological responses as indicators of stress. Worlds Poult Sci J 41:36–44. https://doi.org/10.1079/WPS19850003
Siegel HS (1987) Effects of behavioural and physical stressors on immune responses. In: Wiepkema PR, Van Adrichem PWM (eds) Biology of Stress in Farm Animals: An Integrative Approach. Springer Netherlands, Dordrecht, The Netherlands, pp 39–54. https://doi.org/10.1007/978-94-009-3339-2_4
Slawinska A, Hsieh JC, Schmidt CJ, Lamont SJ (2016) Heat stress and lipopolysaccharide stimulation of chicken macrophage-like cell line activates expression of distinct sets of genes. Plos One 11:e0164575. https://doi.org/10.1371/journal.pone.0164575
Soliman A, Safwat AM (2020) Climate change impact on immune status and productivity of poultry as well as the quality of meat and egg products. In: Ewis Omran E-S, Negm AM (eds) Climate Change Impacts on Agriculture and Food Security in Egypt: Land and Water Resources—Smart Farming—Livestock, Fishery, and Aquaculture. Springer International Publishing, Cham, pp 481–498. https://doi.org/10.1007/978-3-030-41629-4_20
Staal FJT, Luis TC, Tiemessen MM (2008) WNT signalling in the immune system: WNT is spreading its wings. Nat Rev Immunol 8:581–593. https://doi.org/10.1038/nri2360
Stier KS, Almasi B, Gasparini J, Piault R, Roulin A, Jenni L (2009) Effects of corticosterone on innate and humoral immune functions and oxidative stress in barn owl nestlings. J Exp Biol 212:2085–2091. https://doi.org/10.1242/jeb.024406
Surai PF, Fisinin VI (2016a) Vitagenes in poultry production: part 1. Technological and environmental stresses. Worlds Poult Sci J 72:721–734. https://doi.org/10.1017/S0043933916000714
Surai PF, Fisinin VI (2016b) Vitagenes in poultry production: part 2. Nutritional and internal stresses. Worlds Poult Sci J 72:761–772. https://doi.org/10.1017/S0043933916000726
Surai PF, Kochish II, Fisinin VI, Kidd MT (2019) Antioxidant defence systems and oxidative stress in poultry biology: an update. Antioxidants 8:235. https://doi.org/10.3390/antiox8070235
Suttle NF (2010) The mineral nutrition of livestock (4th Edition) CABI Publishing. Oxfordshire, UK
Tella JL, Forero MG, Bertellotti M, Donázar JA, Blanco G, Ceballos O (2001) Offspring body condition and immunocompetence are negatively affected by high breeding densities in a colonial seabird: a multiscale approach. Proc R Soc Lond Ser B-Biol Sci 268:1455–1461. https://doi.org/10.1098/rspb.2001.1688
Trachootham D, Lu W, Ogasawara MA, Valle NR-D, Huang P (2008) Redox regulation of cell survival. Antioxid Redox Signal 10:1343–1374. https://doi.org/10.1089/ars.2007.1957
Tsiouris V, Georgopoulou I, Batzios C, Pappaioannou N, Ducatelle R, Fortomaris P (2015a) The effect of cold stress on the pathogenesis of necrotic enteritis in broiler chicks. Avian Pathol 44:430–435. https://doi.org/10.1080/03079457.2015.1083094
Tsiouris V, Georgopoulou I, Batzios C, Pappaioannou N, Ducatelle R, Fortomaris P (2015b) High stocking density as a predisposing factor for necrotic enteritis in broiler chicks. Avian Pathol 44:59–66. https://doi.org/10.1080/03079457.2014.1000820
Tuerkyilmaz MK (2008) The effect of stocking density on stress reaction in broiler chickens during summer. Turk J Vet Anim Sci 32:31–36
Wang Y (2009) Differential effects of sodium selenite and nano-Se on growth performance, tissue Se distribution, and glutathione peroxidase activity of avian broiler. Biol Trace Elem Res 128:184–190. https://doi.org/10.1007/s12011-008-8264-y
Wang L-C, Liao LX, Lv HN, Liu D, Dong W, Zhu J, Chen JF, Shi ML, Fu G, Song XM, Jiang Y, Zeng KW, Tu PF (2017) Highly selective activation of heat shock protein 70 by allosteric regulation provides an insight into efficient neuroinflammation inhibition. EBioMedicine 23:160–172. https://doi.org/10.1016/j.ebiom.2017.08.011
Wang Y, Saelao P, Chanthavixay K, Gallardo R, Bunn D, Lamont SJ, Dekkers JM, Kelly T, Zhou H (2018) Physiological responses to heat stress in two genetically distinct chicken inbred lines. Poult Sci 97:770–780. https://doi.org/10.3382/ps/pex363
Xie D, Wang ZX, Dong YL, Cao J, Wang JF, Chen JL, Chen YX (2008) Effects of monochromatic light on immune response of broilers. Poult Sci 87:1535–1539. https://doi.org/10.3382/ps.2007-00317
Xie D, Li J, Wang ZX, Cao J, Li TT, Chen JL, Chen YX (2011) Effects of monochromatic light on mucosal mechanical and immunological barriers in the small intestine of broilers. Poult Sci 90:2697–2704. https://doi.org/10.3382/ps.2011-01416
Yalcin S, Özkan S, Türkmut L, Siegel PB (2001) Responses to heat stress in commercial and local broiler stocks. 1. Performance traits. Br Poult Sci 42:149–152. https://doi.org/10.1080/00071660120048375
Yang YF, Jin SF, Zhong ZT, Yu YH, Yang B, Yuan HB, Pan JM (2015) Growth responses of broiler chickens to different periods of artificial light. J Anim Sci 93:767–775. https://doi.org/10.2527/jas2014-8096
Yang Y, Yu Y, Pan J, Ying Y, Zhou H (2016) A new method to manipulate broiler chicken growth and metabolism: response to mixed LED light system. Sci Rep 6:25972. https://doi.org/10.1038/srep25972
Yasuda M, Kajiwara E, Ekino S, Taura Y, Hirota Y, Horiuchi H, Matsuda H, Furusawa S (2003) Immunobiology of chicken germinal center: I. Changes in surface Ig class expression in the chicken splenic germinal center after antigenic stimulation. Dev Comp Immunol 27:159–166. https://doi.org/10.1016/s0145-305x(02)00066-6
Yenari MA, Liu J, Zheng Z, Vexler ZS, Lee JE, Giffard RG (2005) Antiapoptotic and anti-inflammatory mechanisms of heat-shock protein protection. Ann N Y Acad Sci 1053:74–83. https://doi.org/10.1196/annals.1344.007
Zha L, Zeng J, Chu X, Mao L, Luo H (2009) Efficacy of trivalent chromium on growth performance, carcass characteristics and tissue chromium in heat-stressed broiler chicks. J Sci Food Agric 89:1782–1786. https://doi.org/10.1002/jsfa.3656
Zhang JS, Gao XY, Zhang LD, Bao YP (2001) Biological effects of a nano red elemental selenium. BioFactors 15:27–38. https://doi.org/10.1002/biof.5520150103
Zhang L, Zhang HJ, Qiao X, Yue HY, Wu SG, Yao JH, Qi GH (2012) Effect of monochromatic light stimuli during embryogenesis on muscular growth, chemical composition, and meat quality of breast muscle in male broilers. Poult Sci 91:1026–1031. https://doi.org/10.3382/ps.2011-01899
Zhang Y, Zhou Y, Sun G, Li K, Li Z, Su A, Liu X, Li G, Jiang R, Han R, Tian Y, Kang X, Yan F (2018) Transcriptome profile in bursa of Fabricius reveals potential mode for stress-influenced immune function in chicken stress model. BMC Genomics 19:918. https://doi.org/10.1186/s12864-018-5333-2
Zhao C-Y, Tan S-X, Xiao X-Y, Qiu X-S, Pan J-Q, Tang Z-X (2014a) Effects of dietary zinc oxide nanoparticles on growth performance and antioxidative status in broilers. Biol Trace Elem Res 160:361–367. https://doi.org/10.1007/s12011-014-0052-2
Zhao FQ, Zhang ZW, Qu JP, Yao HD, Li M, Li S, Xu SW (2014b) Cold stress induces antioxidants and Hsps in chicken immune organs. Cell Stress Chaperones 19:635–648. https://doi.org/10.1007/s12192-013-0489-9
Zhou X, Wang Y (2011) Influence of dietary nano elemental selenium on growth performance, tissue selenium distribution, meat quality, and glutathione peroxidase activity in Guangxi Yellow chicken. Poult Sci 90:680–686. https://doi.org/10.3382/ps.2010-00977
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abo-Al-Ela, H.G., El-Kassas, S., El-Naggar, K. et al. Stress and immunity in poultry: light management and nanotechnology as effective immune enhancers to fight stress. Cell Stress and Chaperones 26, 457–472 (2021). https://doi.org/10.1007/s12192-021-01204-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-021-01204-6