Abstract
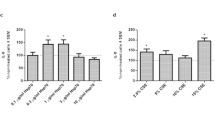
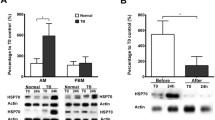
Extracellular Hsp70 (eHsp70) can act as damage-associated molecular pattern (DAMP) via Toll-like receptors TLR2 and TLR4, and stimulate immune and inflammatory responses leading to sterile inflammation and propagation of already existing inflammation. It was found elevated in the blood of patients with chronic obstructive pulmonary disease (COPD), who might suffer occasional bacterial colonizations and infections. We used a monocytic THP-1 cell line as a cellular model of systemic compartment of COPD to assess inflammatory effects of eHsp70 when present alone or together with bacterial products lypopolysaccharide (LPS) and lypoteichoic acid (LTA). THP-1 cells were differentiated into macrophage-like cells and treated with various concentrations of recombinant human Hsp70 protein (rhHsp70), LPS (TLR4 agonist), LTA (TLR2 agonist), and their combinations for 4, 12, 24, and 48 h. Concentrations of IL-1α, IL-6, IL-8, and TNF-α were determined by ELISA. Cell viability was assessed by MTS assay, and mode of cell death by luminometric measurements of caspases-3/7, -8, and -9 activities. rhHsp70 showed cell protecting effect by suppressing caspases-3/7 activation, while LPS provoked cytotoxicity through caspases-8 and -3/7 pathway. Regarding inflammatory processes, rhHsp70 alone induced secretion of IL-1α and IL-8, but had modulatory effects on release of all four cytokines when applied together with LPS or LTA. Combined effect with LPS was mainly synergistic, and with LTA mainly antagonistic, although it was cytokine- and time-dependent. Our results confirmed pro-inflammatory function of extracellular Hsp70, and suggest its possible implication in COPD exacerbations caused by bacterial infection through desensitization or inappropriate activation of TLR2 and TLR4 receptors.










Similar content being viewed by others
References
Abboud PA, Lahni PM, Page K et al (2008) The role of endogenously produced extracellular HSP72 in mononuclear cell reprogramming. Shock 30:285–292. https://doi.org/10.1097/SHK.0b013e318164e2c3
Agusti AGN, Noguera A, Sauleda J et al (2003) Systemic effects of chronic obstructive pulmonary disease. Eur Respir J 21:347–360. https://doi.org/10.1183/09031936.03.00405703
Aneja R, Odoms K, Dunsmore K et al (2006) Extracellular heat shock protein-70 induces endotoxin tolerance in THP-1 cells. J Immunol 177:7184–7192. https://doi.org/10.4049/jimmunol.177.10.7184
Asea A, Rehli M, Kabingu E et al (2002) Novel signal transduction pathway utilized by extracellular HSP70. Role of toll-like receptor (TLR) 2 and TLR4. J Biol Chem 277:15028–15034. https://doi.org/10.1074/jbc.M200497200
Bangen JM, Schade FU, Flohé SB (2007) Diverse regulatory activity of human heat shock proteins 60 and 70 on endotoxin-induced inflammation. Biochem Biophys Res Commun 359:709–715. https://doi.org/10.1016/j.bbrc.2007.05.167
Barnes PJ, Celli BR (2009) Systemic manifestations and comorbidities of COPD. Eur Respir J 33:1165–1185
Chase MA, Wheeler DS, Lierl KM et al (2007) Hsp72 induces inflammation and regulates cytokine production in airway epithelium through a TLR4- and NF-kappaB-dependent mechanism. J Immunol 179:6318–6324
Choi JY (2010) Anti-inflammatory activity of Houttuynia cordata against lipoteichoic acid-induced inflammation in human dermal fibroblasts. Chonnam Med J 46:140–147. https://doi.org/10.4068/cmj.2010.46.3.140
De Maio A, Vazquez D (2013) Extracellular heat shock proteins: a new location, a new function. Shock 40:239–246. https://doi.org/10.1097/SHK.0b013e3182a185ab
Dokladny K, Lobb R, Wharton W et al (2010) LPS-induced cytokine levels are repressed by elevated expression of HSP70 in rats: possible role of NF-κB. Cell Stress Chaperones 15:153–163. https://doi.org/10.1007/s12192-009-0129-6
Dong J, Guo L, Liao Z et al (2013) Increased expression of heat shock protein 70 in chronic obstructive pulmonary disease. Int Immunopharmacol 17:885–893. https://doi.org/10.1016/j.intimp.2013.09.003
Duprez L, Wirawan E, Vanden BT, Vandenabeele P (2009) Major cell death pathways at a glance. Microbes Infect 11:1050–1062. https://doi.org/10.1016/j.micinf.2009.08.013
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35:495–516. https://doi.org/10.1080/01926230701320337
Ferat-Osorio E, Sánchez-Anaya A, Gutiérrez-Mendoza M et al (2014) Heat shock protein 70 down-regulates the production of toll-like receptor-induced pro-inflammatory cytokines by a heat shock factor-1/constitutive heat shock element-binding factor-dependent mechanism. J Inflamm (Lond) 11:19. https://doi.org/10.1186/1476-9255-11-19
Franco L, Terrinca J, Rodríguez AB et al (2016) Extracellular heat shock proteins protect U937 cells from H2O2-induced apoptotic cell death. Mol Cell Biochem 412:19–26. https://doi.org/10.1007/s11010-015-2604-y
Garcia JJ, Martin-Cordero L, Hinchado MD et al (2013) Effects of habitual exercise on the eHsp72-induced release of inflammatory cytokines by macrophages from obese Zucker rats. Int J Sports Med 34:559–564. https://doi.org/10.1055/s-0032-1327650
Giuliano JS, Lahni PM, Wong HR, Wheeler DS (2011) Pediatric sepsis—part V: extracellular heat shock proteins: alarmins for the host immune system. Open Inflamm J 4:49–60. https://doi.org/10.2174/1875041901104010049
Global Initiative for Chronic Obstructive Lung Disease (GOLD) (2017) Global strategy for the diagnosis, management and prevention of COPD. In: www.goldcopd.org. www.goldcopd.org
Gupta A, Cooper ZA, Tulapurkar ME et al (2013) Toll-like receptor agonists and febrile range hyperthermia synergize to induce heat shock protein 70 expression and extracellular release. J Biol Chem 288:2756–2766. https://doi.org/10.1074/jbc.M112.427336
Hacker S, Lambers C, Hoetzenecker K et al (2009) Elevated HSP27, HSP70 and HSP90α in chronic obstructive pulmonary disease: markers for immune activation and tissue destruction. Clin Lab 55:31–40
Hsu J-H, Yang R-C, Lin S-J et al (2014) Exogenous heat shock cognate protein 70 pretreatment attenuates cardiac and hepatic dysfunction with associated anti-inflammatory responses in experimental septic shock. Shock 42:540–547
Huang M-Y, Liao M-H, Wang Y-K et al (2012) Effect of lavender essential oil on LPS-stimulated inflammation. Am J Chin Med 40:845–859. https://doi.org/10.1142/S0192415X12500632
Hutchison ML, Bonell EC, Poxton IR, Govan JRW (2000) Endotoxic activity of lipopolysaccharides isolated from emergent potential cystic fibrosis pathogens. FEMS Immunol Med Microbiol 27:73–77. https://doi.org/10.1016/S0928-8244(99)00165-0
Jean-Pierre C, Perni SC, Bongiovanni AM, et al (2006) Extracellular 70-kd heat shock protein in mid-trimester amniotic fluid and its effect on cytokine production by ex vivo-cultured amniotic fluid cells. In: American Journal of Obstetrics and Gynecology. pp 694–698
Johnson JD, Fleshner M (2006) Releasing signals, secretory pathways, and immune function of endogenous extracellular heat shock protein 72. J Leukoc Biol 79:425–434. https://doi.org/10.1189/jlb.0905523
Joly A-L, Wettstein G, Mignot G et al (2010) Dual role of heat shock proteins as regulators of apoptosis and innate immunity. J Innate Immun 2:238–247. https://doi.org/10.1159/000296508
Liu F-L, Chuang C-Y, Tai Y-T et al (2012) Lipoteichoic acid induces surfactant protein-A biosynthesis in human alveolar type II epithelial cells through activating the MEK1/2-ERK1/2-NF-κB pathway. Respir Res 13:88. https://doi.org/10.1186/1465-9921-13-88
Luo X, Tao L, Lin P et al (2012) Extracellular heat shock protein 72 protects Schwann cells from hydrogen peroxide-induced apoptosis. J Neurosci Res 90:1261–1269. https://doi.org/10.1002/jnr.22810
Mathur S, Walley KR, Wang Y et al (2011) Extracellular heat shock protein 70 induces cardiomyocyte inflammation and contractile dysfunction via TLR2. Circ J 75:2445–2452. https://doi.org/10.1253/circj.CJ-11-0194
Midwood KS, Piccinini AM (2010) DAMPening inflammation by modulating TLR signalling. Mediat Inflamm. 2010
Munshi N, Fernandis AZ, Cherla RP et al (2002) Lipopolysaccharide-induced apoptosis of endothelial cells and its inhibition by vascular endothelial growth factor. J Immunol 168:5860–5866. https://doi.org/10.4049/jimmunol.168.11.5860
Ofengeim D, Yuan J (2013) Regulation of RIP1 kinase signalling at the crossroads of inflammation and cell death. Nat Rev Mol Cell Biol 14:727–736. https://doi.org/10.1038/nrm3683
Pockley AG (2003) Heat shock proteins as regulators of the immune response. Lancet 362:469–476. https://doi.org/10.1016/S0140-6736(03)14075-5
Pockley AG, Muthana M, Calderwood SK (2008) The dual immunoregulatory roles of stress proteins. Trends Biochem Sci 33:71–79
Pouwels SD, Heijink IH, ten Hacken NHT et al (2014) DAMPs activating innate and adaptive immune responses in COPD. Mucosal Immunol 7:215–226. https://doi.org/10.1038/mi.2013.77
Quintana FJ, Cohen IR (2005) Heat shock proteins as endogenous adjuvants in sterile and septic inflammation. J Immunol 175:2777–2782. https://doi.org/10.4049/jimmunol.175.5.2777
Rovina N, Koutsoukou A, Koulouris NG (2013) Inflammation and immune response in COPD: where do we stand? Mediat Inflamm. 2013
Šegvić Klarić M, Medić N, Hulina A et al (2014) Disturbed Hsp70 and Hsp27 expression and thiol redox status in porcine kidney PK15 cells provoked by individual and combined ochratoxin A and citrinin treatments. Food Chem Toxicol 71:97–105. https://doi.org/10.1016/j.fct.2014.06.002
Sethi S, Mahler DA, Marcus P et al (2012) Inflammation in COPD: implications for management. Am J Med 125:1162–1170
Thériault JR, Mambula SS, Sawamura T et al (2005) Extracellular HSP70 binding to surface receptors present on antigen presenting cells and endothelial/epithelial cells. FEBS Lett 579:1951–1960. https://doi.org/10.1016/j.febslet.2005.02.046
Tsan M-F, Gao B (2004) Endogenous ligands of toll-like receptors. J Leukoc Biol 76:514–519. https://doi.org/10.1189/jlb.0304127
Tulapurkar ME, Ramarathnam A, Hasday JD, Singh IS (2015) Bacterial lipopolysaccharide augments febrile-range hyperthermia-induced heat shock protein 70 expression and extracellular release in human THP1 cells. PLoS One. https://doi.org/10.1371/journal.pone.0118010
Vabulas RM, Wagner H, Schild H (2002) Heat shock proteins as ligands of toll-like receptors. Curr Top Microbiol Immunol 270:169–184
Vanden Berghe T, Linkermann A, Jouan-Lanhouet S et al (2014) Regulated necrosis: the expanding network of non-apoptotic cell death pathways. Nat Rev Mol Cell Biol 15:135–147. https://doi.org/10.1038/nrm3737
Vinokurov M, Ostrov V, Yurinskaya M et al (2012) Recombinant human Hsp70 protects against lipoteichoic acid-induced inflammation manifestations at the cellular and organismal levels. Cell Stress Chaperones 17:89–101. https://doi.org/10.1007/s12192-011-0288-0
Wheeler DS, Chase MA, Senft AP et al (2009) Extracellular Hsp72, an endogenous DAMP, is released by virally infected airway epithelial cells and activates neutrophils via Toll-like receptor (TLR)-4. Respir Res 10:31. https://doi.org/10.1186/1465-9921-10-31
Wouters EFM, Celis MPM, Breyer MK et al (2007) Co-morbid manifestations in COPD. Respir Med COPD Update 3:135–151. https://doi.org/10.1016/j.rmedu.2007.08.003
Xaus J, Comalada M, Valledor AF et al (2000) LPS induces apoptosis in macrophages mostly through the autocrine production of TNF-alpha. Blood 95:3823–3831. https://doi.org/10.1016/0167-5699(94)90150-3
Zhang D, Lin J, Han J (2010) Receptor-interacting protein (RIP) kinase family. Cell Mol Immunol 7:243–249. https://doi.org/10.1038/cmi.2010.10
Acknowledgements
This work has been fully supported by the Croatian Science Foundation under the project number IP-2014-09-1247.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hulina, A., Grdić Rajković, M., Jakšić Despot, D. et al. Extracellular Hsp70 induces inflammation and modulates LPS/LTA-stimulated inflammatory response in THP-1 cells. Cell Stress and Chaperones 23, 373–384 (2018). https://doi.org/10.1007/s12192-017-0847-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-017-0847-0