Abstract
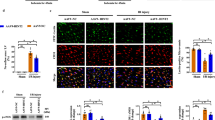
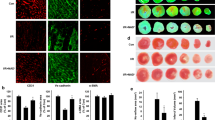
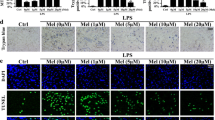
The cardiac microvascular reperfusion injury is characterized by the microvascular endothelial cells (CMECs) oxidative damage which is responsible for the progression of cardiac dysfunction. However, few strategies are available to reverse such pathologies. This study aimed to explore the mechanism by which oxidative stress induced CMECs death and the beneficial actions of melatonin on CMECs survival, with a special focused on IP3R-[Ca2+]c/VDAC-[Ca2+]m damage axis and the MAPK/ERK survival signaling. We found that oxidative stress induced by H2O2 significantly activated cAMP response element binding protein (CREB) that enhanced IP3R and VDAC transcription and expression, leading to [Ca2+]c and [Ca2+]m overload. High concentration of [Ca2+]m suppressed ΔΨm, opened mPTP, and released cyt-c into cytoplasm where it activated mitochondria-dependent death pathway. However, melatonin could protect CMECs against oxidative stress injury via stimulation of MAPK/ERK that inactivated CREB and therefore blocked IP3R/VDAC upregulation and [Ca2+]c/[Ca2+]m overload, sustaining mitochondrial structural and function integrity and ultimately blockading mitochondrial-mediated cellular death. In summary, these findings confirmed the mechanisms by which oxidative injury induced CMECs mitochondrial-involved death and provided an attractive and effective way to enhance CMECs survival.







Similar content being viewed by others
References
Alonso-Alconada D, Alvarez A, Arteaga O, Martinez-Ibarguen A, Hilario E (2013) Neuroprotective effect of melatonin: a novel therapy against perinatal hypoxia-ischemia. Int J Mol Sci 14:9379–9395. doi:10.3390/ijms14059379
Back K, Tan DX, Reiter RJ (2016) Melatonin biosynthesis in plants: multiple pathways catalyze tryptophan to melatonin in the cytoplasm or chloroplasts. J Pineal Res 61:426–437. doi:10.1111/jpi.12364
Chang L, Karin M (2001) Mammalian MAP kinase signalling cascades. Nature 410:37–40. doi:10.1038/35065000
Chapple SJ, Cheng X, Mann GE (2013) Effects of 4-hydroxynonenal on vascular endothelial and smooth muscle cell redox signaling and function in health and disease. Redox Biol 1:319–331. doi:10.1016/j.redox.2013.04.001
Chen HH et al (2016) Melatonin pretreatment enhances the therapeutic effects of exogenous mitochondria against hepatic ischemia-reperfusion injury in rats through suppression of mitochondrial permeability transition. J Pineal Res 61:52–68. doi:10.1111/jpi.12326
Choi YH (2016) The cytoprotective effect of isorhamnetin against oxidative stress is mediated by the upregulation of the Nrf2-dependent HO-1 expression in C2C12 myoblasts through scavenging reactive oxygen species and ERK inactivation. Gen Physiol Biophys 35:145–154. doi:10.4149/gpb_2015034
Cosentino K, Garcia-Saez AJ (2014) Mitochondrial alterations in apoptosis. Chem Phys Lipids 181:62–75. doi:10.1016/j.chemphyslip.2014.04.001
Dominguez-Rodriguez A et al (2017) Effect of intravenous and intracoronary melatonin as an adjunct to primary percutaneous coronary intervention for acute ST-elevation myocardial infarction: results of the melatonin adjunct in the acute myocardial infarction treated with angioplasty trial. J Pineal Res:62. doi:10.1111/jpi.12374
Fernandez-Sanz C et al (2014) Defective sarcoplasmic reticulum-mitochondria calcium exchange in aged mouse myocardium. Cell Death Dis 5:e1573. doi:10.1038/cddis.2014.526
Gaspar R et al (2016) The cytoprotective effect of biglycan core protein involves toll-like receptor 4 signaling in cardiomyocytes. J Mol Cell Cardiol. doi:10.1016/j.yjmcc.2016.08.006
Givvimani S, Pushpakumar SB, Metreveli N, Veeranki S, Kundu S, Tyagi SC (2015) Role of mitochondrial fission and fusion in cardiomyocyte contractility. Int J Cardiol 187:325–333. doi:10.1016/j.ijcard.2015.03.352
Grimm S (2012) The ER-mitochondria interface: the social network of cell death. Biochim Biophys Acta 1823:327–334. doi:10.1016/j.bbamcr.2011.11.018
Han D et al (2016) Melatonin facilitates adipose-derived mesenchymal stem cells to repair the murine infarcted heart via the SIRT1 signaling pathway. J Pineal Res 60:178–192. doi:10.1111/jpi.12299
Haorah J, Knipe B, Gorantla S, Zheng J, Persidsky Y (2007) Alcohol-induced blood-brain barrier dysfunction is mediated via inositol 1,4,5-triphosphate receptor (IP3R)-gated intracellular calcium release. J Neurochem 100:324–336. doi:10.1111/j.1471-4159.2006.04245.x
Herrera EA, Farias JG, Ebensperger G, Reyes RV, Llanos AJ, Castillo RL (2015) Pharmacological approaches in either intermittent or permanent hypoxia: a tale of two exposures. Pharmacol Res 101:94–101. doi:10.1016/j.phrs.2015.07.011
Hill SM, Cheng C, Yuan L, Mao L, Jockers R, Dauchy B, Blask DE (2013) Age-related decline in melatonin and its MT1 receptor are associated with decreased sensitivity to melatonin and enhanced mammary tumor growth. Curr Aging Sci 6:125–133
Hough MA, Silkstone G, Worrall JA, Wilson MT (2014) NO binding to the proapoptotic cytochrome c-cardiolipin complex. Vitam Horm 96:193–209. doi:10.1016/B978-0-12-800254-4.00008-8
Hu W et al (2016) Melatonin: the dawning of a treatment for fibrosis? J Pineal Res 60:121–131. doi:10.1111/jpi.12302
Hurst S, Hoek J, Sheu SS (2016) Mitochondrial Ca2+ and regulation of the permeability transition pore. J Bioenerg Biomembr. doi:10.1007/s10863-016-9672-x
Ivanova H, Vervliet T, Missiaen L, Parys JB, De Smedt H, Bultynck G (2014) Inositol 1,4,5-trisphosphate receptor-isoform diversity in cell death and survival. Biochim Biophys Acta 1843:2164–2183. doi:10.1016/j.bbamcr.2014.03.007
Kagan VE et al (2004) Oxidative lipidomics of apoptosis: redox catalytic interactions of cytochrome c with cardiolipin and phosphatidylserine. Free Radic Biol Med 37:1963–1985. doi:10.1016/j.freeradbiomed.2004.08.016
Kim EK, Choi EJ (2015) Compromised MAPK signaling in human diseases: an update. Arch Toxicol 89:867–882. doi:10.1007/s00204-015-1472-2
Laudon M, Frydman-Marom A (2014) Therapeutic effects of melatonin receptor agonists on sleep and comorbid disorders. Int J Mol Sci 15:15924–15950. doi:10.3390/ijms150915924
Lin YW et al (2016) Melatonin inhibits MMP-9 transactivation and renal cell carcinoma metastasis by suppressing Akt-MAPKs pathway and NF-kappaB DNA-binding activity. J Pineal Res 60:277–290. doi:10.1111/jpi.12308
Majidinia M, Sadeghpour A, Mehrzadi S, Reiter RJ, Khatami N, Yousefi B (2017) Melatonin: a pleiotropic molecule that modulates DNA damage response and repair pathways. J Pineal Res. doi:10.1111/jpi.12416
Mayo JC, Sainz RM, Gonzalez Menendez P, Cepas V, Tan DX, Reiter RJ (2017) Melatonin and sirtuins: a “not-so unexpected” relationship. J Pineal Res 62. doi:10.1111/jpi.12391
Monaco G et al (2015) The BH4 domain of anti-apoptotic Bcl-XL, but not that of the related Bcl-2, limits the voltage-dependent anion channel 1 (VDAC1)-mediated transfer of pro-apoptotic Ca2+ signals to mitochondria. J Biol Chem 290:9150–9161. doi:10.1074/jbc.M114.622514
Moosavi F, Hosseini R, Saso L, Firuzi O (2016) Modulation of neurotrophic signaling pathways by polyphenols. Drug Des Devel Ther 10:23–42. doi:10.2147/DDDT.S96936
Orlova DD, Tribulovich VG, Garabadzhiu AV, Barlev NA, Martin S (2015) The role of mitochondrial dynamics in cell death. Tsitologiia 57:184–191
Park BK, Gonzales EL, Yang SM, Bang M, Choi CS, Shin CY (2016) Effects of triclosan on neural stem cell viability and survival. Biomol Ther (Seoul) 24:99–107. doi:10.4062/biomolther.2015.164
Pei HF et al (2017) Melatonin attenuates postmyocardial infarction injury via increasing Tom70 expression. J Pineal Res 62. doi:10.1111/jpi.12371
Peters SC, Piper HM (2007) Reoxygenation-induced Ca2+ rise is mediated via Ca2+ influx and Ca2+ release from the endoplasmic reticulum in cardiac endothelial cells. Cardiovasc Res 73:164–171. doi:10.1016/j.cardiores.2006.09.015
Potier E, Ferreira E, Meunier A, Sedel L, Logeart-Avramoglou D, Petite H (2007) Prolonged hypoxia concomitant with serum deprivation induces massive human mesenchymal stem cell death. Tissue Eng 13:1325–1331. doi:10.1089/ten.2006.0325
Reiter RJ, Mayo JC, Tan DX, Sainz RM, Alatorre-Jimenez M, Qin L (2016a) Melatonin as an antioxidant: under promises but over delivers. J Pineal Res. doi:10.1111/jpi.12360
Reiter RJ, Mayo JC, Tan DX, Sainz RM, Alatorre-Jimenez M, Qin L (2016b) Melatonin as an antioxidant: under promises but over delivers. J Pineal Res 61:253–278. doi:10.1111/jpi.12360
Saggioro D (2011) Anti-apoptotic effect of tax: an NF-kappaB path or a CREB way? Viruses 3:1001–1014. doi:10.3390/v3071001
Shah SZ, Zhao D, Khan SH, Yang L (2015) Regulatory mechanisms of endoplasmic reticulum resident IP3 receptors. J Mol Neurosci 56:938–948. doi:10.1007/s12031-015-0551-4
Shore GC, Papa FR, Oakes SA (2011) Signaling cell death from the endoplasmic reticulum stress response. Curr Opin Cell Biol 23:143–149. doi:10.1016/j.ceb.2010.11.003
Sirtori CR, Arnoldi A, Cicero AF (2015) Nutraceuticals for blood pressure control. Ann Med 47:447–456. doi:10.3109/07853890.2015.1078905
Sun K, Fan J, Han J (2015a) Ameliorating effects of traditional Chinese medicine preparation, Chinese materia medica and active compounds on ischemia/reperfusion-induced cerebral microcirculatory disturbances and neuron damage. Acta Pharm Sin B 5:8–24. doi:10.1016/j.apsb.2014.11.002
Sun Y, Liu WZ, Liu T, Feng X, Yang N, Zhou HF (2015b) Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J Recept Signal Transduct Res 35:600–604. doi:10.3109/10799893.2015.1030412
Thorburn A (2004) Death receptor-induced cell killing. Cell Signal 16:139–144
Vervloessem T, Yule DI, Bultynck G, Parys JB (2015) The type 2 inositol 1,4,5-trisphosphate receptor, emerging functions for an intriguing Ca(2)(+)-release channel. Biochim Biophys Acta 1853:1992–2005. doi:10.1016/j.bbamcr.2014.12.006
Xu P et al (2017) Melatonin prevents obesity through modulation of gut microbiota in mice. J Pineal Res 62. doi:10.1111/jpi.12399
Yiallourou SR, Wallace EM, Miller SL, Horne RS (2016) Effects of intrauterine growth restriction on sleep and the cardiovascular system: the use of melatonin as a potential therapy? Sleep Med Rev 26:64–73. doi:10.1016/j.smrv.2015.04.001
Yu L et al (2017a) Melatonin rescues cardiac thioredoxin system during ischemia-reperfusion injury in acute hyperglycemic state by restoring Notch1/Hes1/Akt signaling in a membrane receptor-dependent manner. J Pineal Res 62. doi:10.1111/jpi.12375
Yu S et al (2017b) Melatonin regulates PARP1 to control the senescence-associated secretory phenotype (SASP) in human fetal lung fibroblast cells. J Pineal Res. doi:10.1111/jpi.12405
Zhang Y et al (2016) Liraglutide protects cardiac microvascular endothelial cells against hypoxia/reoxygenation injury through the suppression of the SR-Ca(2+)-XO-ROS axis via activation of the GLP-1R/PI3K/Akt/survivin pathways. Free Radic Biol Med 95:278–292. doi:10.1016/j.freeradbiomed.2016.03.035
Zhou H et al. (2017a) Mff-dependent mitochondrial fission contributes to the pathogenesis of cardiac microvasculature ischemia/reperfusion injury via induction of mROS-mediated cardiolipin oxidation and HK2/VDAC1 disassociation-involved mPTP opening J Am Heart Assoc 6. doi:10.1161/JAHA.116.005328
Zhou H et al (2015) Effects of Exendin-4 on bone marrow mesenchymal stem cell proliferation, migration and apoptosis in vitro. Sci Rep 5:12898. doi:10.1038/srep12898
Zhou H et al (2014) Exendin-4 protects adipose-derived mesenchymal stem cells from apoptosis induced by hydrogen peroxide through the PI3K/Akt-Sfrp2 pathways. Free Radic Biol Med 77:363–375. doi:10.1016/j.freeradbiomed.2014.09.033
Zhou H et al (2017b) Melatonin protects cardiac microvasculature against ischemia/reperfusion injury via suppression of mitochondrial fission-VDAC1-HK2-mPTP-mitophagy axis. J Pineal Res. doi:10.1111/jpi.12413
Acknowledgments
This study was supported by grants from the National 863 high technology R&D Program (No. 2011AA020101), National Natural Science Foundation of China (No: 81270186, 81102079), and Key Project of Natural Science Foundation of China (No: 81030002). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhu, H., Jin, Q., Li, Y. et al. Melatonin protected cardiac microvascular endothelial cells against oxidative stress injury via suppression of IP3R-[Ca2+]c/VDAC-[Ca2+]m axis by activation of MAPK/ERK signaling pathway. Cell Stress and Chaperones 23, 101–113 (2018). https://doi.org/10.1007/s12192-017-0827-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-017-0827-4