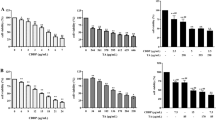
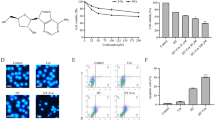
Abstract
KRAS mutant lung cancers have long been considered as untreatable with drugs. Transforming growth factor-β-activated kinase 1 (TAK1) appears to play an anti-apoptotic role in response to multiple stresses and has been reported to be a responsive kinase that regulates cell survival in KRAS-dependent cells. In this study, in order to find a useful approach to treat KRAS mutant lung cancer, we focused on the combined effects of 5Z-7-oxozeaenol, a TAK1 inhibitor, with hyperthermia (HT) in KRAS mutant lung cancer cell line A549. Annexin V-FITC/PI assay, cell cycle analysis, and colony formation assay revealed a significant enhancement in apoptosis induced by HT treatment, when the cells were pre-incubated with 5Z-7-oxozeaenol in a dose-dependent manner. The enhanced apoptosis by 5Z-7-oxozeaenol was accompanied by a significant increase in reactive oxygen species (ROS) generation and loss of mitochondrial membrane potential (MMP). In addition, western blot showed that 5Z-7-oxozeaenol enhanced HT-induced expressions of cleaved caspase-3, cleaved caspase-8, and HSP70 and decreased HT-induced expressions of Bcl-2, p-p38, p-JNK, and LC3. Moreover, 5Z-7-oxozeaenol pre-treatment resulted in a marked elevation of intracellular calcium level which might be associated with endoplasmic reticulum (ER) stress-related pathway. Taken together, our data provides further insights of the mechanism of action of 5Z-7-oxozeaenol and HT treatment, and their potential application as a novel approache to treat patients with KRAS mutant lung cancer.





Similar content being viewed by others
Abbreviations
- AIF:
-
Apoptosis-inducing factor
- DMSO:
-
Dimethyl sulfoxide
- DiOC6(3):
-
3,3′-dihexyloxacarbocyanine iodide
- FITC:
-
Fluorescein isothiocyanate
- HE:
-
Hydroethidine
- HPF:
-
Hydroxyphenyl fluorescein
- HT:
-
Hyperthermia
- IP3R:
-
Inositol 1,4,5-trisphosphate receptor
- MAP3K:
-
Mitogen-activated protein kinase kinase kinase
- MMP:
-
Mitochondrial membrane potential
- MKKs:
-
Mitogen-activated protein kinase kinases
- MPTP:
-
Mitochondria permeability transition pore
- NSCLC:
-
Non-small cell lung cancer
- PI:
-
Propidium iodide
- ROS:
-
Reactive oxygen species
- SCLC:
-
Small cell lung cancer
- TAK1:
-
Transforming growth factor-β-activated kinase 1
References
Adams JM, Cory S (2001) Life-or-death decisions by the Bcl-2 protein family. Trends Biochem Sci 26:61–66
Beere HM, Wolf BB, Cain K et al (2000) Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nat Cell Biol 2:469–475
Brookes PS, Yoon Y, Robotham JL, Anders MW, Sheu SS (2004) Calcium, ATP, and ROS: a mitochondrial love-hate triangle. Am J Physiol Cell Physiol 287:C817–833
Chen F, Wang CC, Kim E, Harrison LE (2008) Hyperthermia in combination with oxidative stress induces autophagic cell death in HT-29 colon cancer cells. Cell Biol Int 32:715–723
Chien CY, Chien CT, Wang SS (2014) Progressive thermopreconditioning attenuates rat cardiac ischemia/reperfusion injury by mitochondria-mediated antioxidant and antiapoptotic mechanisms. J Thorac Cardiovasc Surg 148:705–713
Choi AY, Choi JH, Lee JY, Yoon KS, Choe W, Ha J et al (2010) Apigenin protects HT22 murine hippocampal neuronal cells against endoplasmic reticulum stress-induced apoptosis. Neurochem Int 57:143–152
Cordero JB, Macagno JP, Stefanatos RK, Strathdee KE, Cagan RL, Vidal M (2010) Oncogenic Ras diverts a host TNF tumor suppressor activity into tumor promoter. Dev Cell 18:999–1011
Daniel S, Arvelo MB, Patel VI, Longo CR, Shrikhande G, Shukri T et al (2004) A20 protects endothelial cells from TNF-, Fas-, and NK-mediated cell death by inhibiting caspase 8 activation. Blood 104:2376–2384
Downward J (2003) Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer 3:11–22
Engelhardt R (1987) Rational for clinical application of hyperthermia and drugs. Rev Med Chir Soc Med Nat Iasi 91:347–352
Fan Y, Cheng J, Vasudevan SA, Patel RH, Liang L, Xu X, Zhao Y, Jia W, Lu F, Zhang H, Nuchtern JG, Kim ES, Yang J (2013) TAK1 inhibitor 5Z-7-oxozeaenol sensitizes neuroblastoma to chemotherapy. Apoptosis 18:1224–1234
Giorgi C, Baldassari F, Bononi A, Bonora M, De Marchi E, Marchi S, Missiroli S, Patergnani S, Rimessi A, Suski JM, Wieckowski MR, Pinton P (2012) Mitochondrial Ca(2+) and apoptosis. Cell Calcium 52:36–43
Gu ZT, Li L, Wu F, Zhao P, Yang H, Liu YS, Geng Y, Zhao M, Su L (2015) Heat stress induced apoptosis is triggered by transcription-independent p53, Ca(2+) dyshomeostasis and the subsequent Bax mitochondrial translocation. Sci Rep 5:11497
Hayden MS, Ghosh S (2008) Shared principles in NF-kB signaling. Cell 132:344–362
Hou CH, Lin FL, Hou SM, Liu JF (2014) Hyperthermia induces apoptosis through endoplasmic reticulum and reactive oxygen species in human osteosarcoma cells. Int J Mol Sci 15:17380–17395
Kim BJ, Ryu SW, Song BJ (2006) JNK- and p38 kinase-mediated phosphorylation of Bax leads to its activation and mitochondrial translocation and to apoptosis of human hepatoma HepG2 cells. J Biol Chem 281:21256–21265
Ku BM, Jho EH, Bae YH, Sun JM, Ahn JS, Park K, Ahn MJ (2015) BYL719, a selective inhibitor of phosphoinositide 3-kinase α, enhances the effect of selumetinib (AZD6244, ARRY-142886) in KRAS-mutant non-small cell lung cancer. Invest New Drugs 33:12–21
Lee CH, Jeon YT, Kim SH et al (2007) NF-kappaB as a potential molecular target for cancer therapy. BioFactors 29:19–35
Li P, Furusawa Y, Wei ZL, Sakurai H, Tabuchi Y, Zhao QL, Saiki I, Kondo T (2013) TAK1 promotes cell survival by TNFAIP3 and IL-8 dependent and NF-kB independent pathway in HeLa cells exposed to heat stress. Int J Hyperthermia 29:688–695
Li P, Zhao QL, Wu LH, Jawaid P, Jiao YF, Kadowaki M, Kondo T (2014) Isofraxidin, a potent reactive oxygen species (ROS) scavenger, protects human leukemia cells from radiation-induced apoptosis via ROS/mitochondria pathway in p53-independent manner. Apoptosis 19:1043–1053
McAnulty SR, McAnulty L, Pascoe DD, Gropper SS, Keith RE, Morrow JD, Gladden LB (2005) Hyperthermia increases exercise-induced oxidative stress. Int J Sports Med 26:188–192
McCullough KD, Martindale JL, Klotz LO, Aw TY, Holbrook NJ (2001) Gadd153 sensitizes cells to endoplasmic reticulum stress by down-regulating Bcl2 and perturbing the cellular redox state. Mol Cell Biol 21:1249–1259
Mihaly SR, Ninomiya-Tsuji J, Morioka S (2014) TAK1 control of cell death. Cell Death Differ 21:1667–1676
Miyazaki Y, Kaikita K, Endo M, Horio E, Miura M, Tsujita K et al (2011) C/EBP homologous protein deficiency attenuates myocardial reperfusion injury by inhibiting myocardial apoptosis and inflammation. Arterioscler Thromb Vasc Biol 31:1124–1132
Neubert M, Ridder DA, Bargiotas P, Akira S, Schwaninger M (2011) Acute inhibition of TAK1 protects against neuronal death in cerebral ischemia. Cell Death Differ 18:1521–1530
Ninomiya-Tsuji J, Kajino T, Ono K, Ohtomo T, Matsumoto M, Shiina M, Mihara M, Tsuchiya M, Matsumoto K (2003) A resorcylic acid lactone, 5Z-7-oxozeaenol, prevents inflammation by inhibiting the catalytic activity of TAK1 MAPK kinase kinase. J Biol Chem 278:18485–18490
Omori E, Morioka S, Matsumoto K, Ninomiya-Tsuji J (2008) TAK1 regulates reactive oxygen species and cell death in keratinocytes, which is essential for skin integrity. J Biol Chem 283:26161–26168
Orrenius S, Zhivotovsky B, Nicotera P (2003) Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol 4:552–565
Ott M, Gogvadze V, Orrenius S, Zhivotovsky B (2007) Mitochondria, oxidative stress and cell death. Apoptosis 12:913–922
Oyadomari S, Mori M (2004) Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ 11:381–389
Sakurai H (2012) Targeting of TAK1 in inflammatory disorders and cancer. Trends Pharmacol Sci 33:522–530
Schober A (2008) Chemokines in vascular dysfunction and remodeling. Arterioscler Thromb Vasc Biol 28:1950–1959
Siegel R, Ma J, Zou Z, Jemal A (2014) Cancer statistics. CA Cancer J Clin 64:9–29
Singh A, Sweeney MF, Yu M, Burger A, Greninger P, Benes C, Haber DA, Settleman J (2012) TAK1 inhibition promotes apoptosis in KRAS-dependent colon cancers. Cell 148:639–650
Song Z, Zhu X, Jin R, Wang C, Yan J, Zheng Q, Nanda A, Granger DN, Li G (2014) Roles of the kinase TAK1 in CD40-mediated effects on vascular oxidative stress and neointima formation after vascular injury. PLoS One 9:e101671
Speidel D (2010) Transcription-independent p53 apoptosis: an alternative route to death. Trends Cell Biol 20:14–24
Swanlund JM, Kregel KC, Oberley TD (2008) Autophagy following heat stress: the role of aging and protein nitration. Autophagy 4:936–939
Tripathi P, Hildeman D (2004) Sensitization of T cells to apoptosis—a role for ROS? Apoptosis 9:515–523
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 555:335–344
Wold LE, Ceylan-Isik AF, Fang CX, Yang X, Li SY, Sreejayan N, Privratsky JR, Ren J (2006) Metallothionein alleviates cardiac dysfunction in streptozotocin-induced diabetes: role of Ca2+ cycling proteins, NADPH oxidase, poly(ADP-Ribose) polymerase and myosin heavy chain isozyme. Free Radic Biol Med 40:1419–1429
Yamaguchi K, Shirakabe K, Shibuya H, Irie K, Oishi I, Ueno N et al (1995) Identification of a member of the MAPKKK family as a potential mediator of TGF-β signal transduction. Science 270:2008–2011
Yin XM (2000) Signal transduction mediated by Bid, a pro-death Bcl-2 family proteins, connects the death receptor and mitochondria apoptosis pathways. Cell Res 10:161–167
Yu T, Robotham JL, Yoon Y (2006) Increased production of reactive oxygen species in hyperglycemic conditions requires dynamic change of mitochondrial morphology. Proc Natl Acad Sci U S A 103:2653–2658
Zhang Y, Calderwood SK (2011) Autophagy, protein aggregation and hyperthermia: a mini-review. Int J Hyperthermia 27:409–414
Zhao QL, Fujiwara Y, Kondo T (2006) Mechanism of cell death induction by nitroxide and hyperthermia. Free Radic Biol Med 40:1131–1143
Acknowledgments
This work was supported in part by Grant-in-Aid for Challenging Exploratory Research (No. 26670551), Japan society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, P., Zhao, QL., Jawaid, P. et al. Enhancement of hyperthermia-induced apoptosis by 5Z-7-oxozeaenol, a TAK1 inhibitor, in A549 cells. Cell Stress and Chaperones 21, 873–881 (2016). https://doi.org/10.1007/s12192-016-0712-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-016-0712-6