Abstract
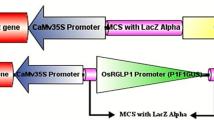
Rice (Oryza sativa) ClpB-C (OsClpB-C) protein is expressed upon heat stress in vegetative tissues and constitutively in seeds. We produced stably transformed Arabidopsis plants carrying β-glucuronidase (Gus) reporter gene downstream to 1-kb OsClpB-C promoter (1kbPro plants). In the 1kbPro plants, expression of Gus transcript and protein followed the expression pattern of OsClpB-C gene in rice plants, i.e., heat induced in vegetative tissues and constitutive in seeds. Next, we produced transgenic Arabidopsis plants containing Gus downstream to 862-bp fragment of OsClpB-C promoter [lacking 138 nucleotides from 3′ end of the 5′untranslated region (5′UTR); ∆UTR plants). In ∆UTR plants, Gus transcript was expressed in heat-inducible manner, but strikingly, Gus protein levels were negligible after heat treatment. However, Gus protein was expressed in ∆UTR seedlings at levels comparable to 1kbPro seedlings when recovery treatment of 22 °C/2 h was given post heat stress (38 °C/15 min). This suggests that 5′UTR of OsClpB-C gene is involved in its post-transcriptional regulation and is an obligate requirement for protein expression during persistent heat stress. Furthermore, the Gus transcript levels were higher in the polysomal RNA fraction in heat-stressed seedlings of 1kbPro plants as compared to ∆UTR plants, indicating that 5′UTR aids in assembly of ribosomes onto the Gus transcript during heat stress. Unlike the case of seedlings, Gus protein was formed constitutively in ∆UTR seeds at levels comparable to 1kbPro seeds. Hence, the function of 5′UTR of OsClpB-C is dispensable for expression in seeds.









Similar content being viewed by others
References
Bate N, Spurr C, Foster GD, Twell D (1996) Maturation-specific translational enhancement mediated by the 5′-UTR of a late pollen transcript. Plant J 10:613–623
Caspar T, Quail PH (1993) Promoter and leader regions involved in the expression of the Arabidopsis ferredoxin A gene. Plant J 3:161–174
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159. doi:10.1016/0003-2697(87)90021-2
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Crone D, Rueda J, Martin KL, Hamilton§ DA, Mascarenhas JP (2001) The differential expression of a heat shock promoter in floral and reproductive tissues. Plant Cell Environ 24:869–874
de Nadal E, Ammerer G, Posas F (2011) Controlling gene expression in response to stress. Nat Rev Genet 12:833–845. doi:10.1038/nrg3055
DeGracia DJ, Jamison JT, Szymanski JJ, Lewis MK (2008) Translation arrest and ribonomics in post-ischemic brain: layers and layers of players. J Neurochem 106:2288–2301. doi:10.1111/j.1471-4159.2008.05561.x
Dinkova TD, Zepeda H, Martinez-Salas E, Martinez LM, Nieto-Sotelo J, de Jimenez ES (2005) Cap-independent translation of maize Hsp101. Plant J 41:722–731. doi:10.1111/j.1365-313X.2005.02333.x
Echevarria-Zomeno S, Yanguez E, Fernandez-Bautista N, Castro-Sanz AB, Ferrando A, Castellano MM (2013) Regulation of translation initiation under biotic and abiotic stresses. Int J Mol Sci 14:4670–4683. doi:10.3390/ijms14034670
Fragkostefanakis S, Roth S, Schleiff E, Scharf KD (2014) Prospects of engineering thermotolerance in crops through modulation of heat stress transcription factor and heat shock protein networks. Plant Cell Environ. doi:10.1111/pce.12396
Freeman J, Sparks CA, West J, Shewry PR, Jones HD (2011) Temporal and spatial control of transgene expression using a heat-inducible promoter in transgenic wheat. Plant Biotechnol J 9:788–796. doi:10.1111/j.1467-7652.2011.00588.x
Gallie DR (2002) The 5′-leader of tobacco mosaic virus promotes translation through enhanced recruitment of eIF4F. Nucleic Acids Res 30:3401–3411
Holcik M, Pestova TV (2007) Translation mechanism and regulation: old players, new concepts. Meeting on translational control and non-coding RNA. EMBO Rep 8:639–643. doi:10.1038/sj.embor.7400988
Hong SW, Vierling E (2000) Mutants of Arabidopsis thaliana defective in the acquisition of tolerance to high temperature stress. Proc Natl Acad Sci U S A 97:4392–4397
Hong SW, Vierling E (2001) Hsp101 is necessary for heat tolerance but dispensable for development and germination in the absence of stress. Plant J 27:25–35
Hu WW, Gong H, Pua EC (2005) The pivotal roles of the plant S-adenosylmethionine decarboxylase 5′ untranslated leader sequence in regulation of gene expression at the transcriptional and posttranscriptional levels. Plant Physiol 138:276–286. doi:10.1104/pp. 104.056770
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: beta-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Katiyar-Agarwal S, Agarwal M, Grover A (2003) Heat-tolerant basmati rice engineered by over-expression of hsp101. Plant Mol Biol 51:677–686
Kawaguchi R, Bailey-Serres J (2005) mRNA sequence features that contribute to translational regulation in Arabidopsis. Nucleic Acids Res 33:955–965. doi:10.1093/nar/gki240
Khurana N, Chauhan H, Khurana P (2013) Wheat chloroplast targeted sHSP26 promoter confers heat and abiotic stress inducible expression in transgenic Arabidopsis plants. PLoS One 8:e54418. doi:10.1371/journal.pone.0054418
Kobayashi N, Horikoshi T, Katsuyama H, Handa T, Takayanagi K (1998) A simple and efficient DNA extraction method for plants, especially woody plants. Plant Tissue Cult Biotech 4:76–80
Lawless C et al (2009) Upstream sequence elements direct post-transcriptional regulation of gene expression under stress conditions in yeast. BMC Genomics 10:7. doi:10.1186/1471-2164-10-7
Lee KT, Chen SC, Chiang BL, Yamakawa T (2007a) Heat-inducible production of beta-glucuronidase in tobacco hairy root cultures. Appl Microbiol Biotechnol 73:1047–1053. doi:10.1007/s00253-006-0576-2
Lee U, Rioflorido I, Hong SW, Larkindale J, Waters ER, Vierling E (2007b) The Arabidopsis ClpB/Hsp100 family of proteins: chaperones for stress and chloroplast development. Plant J 49:115–127. doi:10.1111/j.1365-313X.2006.02940.x
Lee LY, Fang MJ, Kuang LY, Gelvin SB (2008) Vectors for multi-color bimolecular fluorescence complementation to investigate protein-protein interactions in living plant cells. Plant Methods 4:24. doi:10.1186/1746-4811-4-24
Liu H et al (2012) Characterization and evolution of 5′ and 3′ untranslated regions in eukaryotes. Gene 507:106–111. doi:10.1016/j.gene.2012.07.034
Martin F, Barends S, Jaeger S, Schaeffer L, Prongidi-Fix L, Eriani G (2011) Cap-assisted internal initiation of translation of histone H4. Mol Cell 41:197–209. doi:10.1016/j.molcel.2010.12.019
Martinez-Salas E, Pineiro D, Fernandez N (2012) Alternative mechanisms to initiate translation in eukaryotic mRNAs. Comp Funct Genom 2012:391546. doi:10.1155/2012/391546
Mishra RC, Grover A (2014) Intergenic sequence between Arabidopsis ClpB-C/Hsp100 and choline kinase genes functions as a heat inducible bidirectional promoter. Plant Physiol. doi:10.1104/pp. 114.250787
Mishra RC, Grover A (2015) ClpB/Hsp100 proteins and heat stress tolerance in plants. Crit Rev Biotechnol 3:1–13. doi:10.3109/07388551.2015.1051942
Mittal D, Enoki Y, Lavania D, Singh A, Sakurai H, Grover A (2011) Binding affinities and interactions among different heat shock element types and heat shock factors in rice (Oryza sativa L.). FEBS J 278:3076–3085. doi:10.1111/j.1742-4658.2011.08229.x
Pareek A, Singla SL, Grover A (1995) Immunological evidence for accumulation of two high-molecular-weight (104 and 90 kDa) HSPs in response to different stresses in rice and in response to high temperature stress in diverse plant genera. Plant Mol Biol 29:293–301
Patel M, Corey AC, Yin LP, Ali S, Taylor WC, Berry JO (2004) Untranslated regions from C4 amaranth AhRbcS1 mRNAs confer translational enhancement and preferential bundle sheath cell expression in transgenic C4 Flaveria bidentis. Plant Physiol 136:3550–3561. doi:10.1104/pp. 104.051508
Qu AL, Ding YF, Jiang Q, Zhu C (2013) Molecular mechanisms of the plant heat stress response. Biochem Biophys Res Commun 432:203–207. doi:10.1016/j.bbrc.2013.01.104
Rubtsova MP, Sizova DV, Dmitriev SE, Ivanov DS, Prassolov VS, Shatsky IN (2003) Distinctive properties of the 5′-untranslated region of human hsp70 mRNA. J Biol Chem 278:22350–22356. doi:10.1074/jbc.M303213200
Saidi Y, Finka A, Chakhporanian M, Zryd JP, Schaefer DG, Goloubinoff P (2005) Controlled expression of recombinant proteins in Physcomitrella patens by a conditional heat-shock promoter: a tool for plant research and biotechnology. Plant Mol Biol 59:697–711. doi:10.1007/s11103-005-0889-z
Sarkar NK, Kim YK, Grover A (2009) Rice sHsp genes: genomic organization and expression profiling under stress and development. BMC Genomics 10:393. doi:10.1186/1471-2164-10-393
Sherameti I, Nakamura M, Yamamoto YY, Pfannschmidt T, Obokata J, Oelmuller R (2002) Polyribosome loading of spinach mRNAs for photosystem I subunits is controlled by photosynthetic electron transport. Plant J 32:631–639
Singh A, Grover A (2010) Plant Hsp100/ClpB-like proteins: poorly-analyzed cousins of yeast ClpB machine. Plant Mol Biol 74:395–404. doi:10.1007/s11103-010-9682-8
Singh A, Singh U, Mittal D, Grover A (2010) Genome-wide analysis of rice ClpB/HSP100, ClpC and ClpD genes. BMC Genomics 11:95. doi:10.1186/1471-2164-11-95
Singh A, Mittal D, Lavania D, Agarwal M, Mishra RC, Grover A (2012) OsHsfA2c and OsHsfB4b are involved in the transcriptional regulation of cytoplasmic OsClpB (Hsp100) gene in rice (Oryza sativa L.). Cell Stress Chaperones 17:243–254. doi:10.1007/s12192-011-0303-5
Singla SL, Pareek A, Kush AK, Grover A (1998) Distribution patterns of 104 kDa stress-associated protein in rice. Plant Mol Biol 37:911–919
Taliercio E, Ray JD (2001) Identification of transcripts translated on free or membrane-bound polyribosomes by differential display. Plant Mol Biol Report 19:269a–269f
Vivinus S et al (2001) An element within the 5′ untranslated region of human Hsp70 mRNA which acts as a general enhancer of mRNA translation. Eur J Biochem 268:1908–1917
Yamamoto YY, Tsuji H, Obokata J (1995) 5′-leader of a photosystem I gene in Nicotiana sylvestris, psaDb, contains a translational enhancer. J Biol Chem 270:12466–12470
Yanguez E, Castro-Sanz AB, Fernandez-Bautista N, Oliveros JC, Castellano MM (2013) Analysis of genome-wide changes in the translatome of Arabidopsis seedlings subjected to heat stress. PLoS One 8:e71425. doi:10.1371/journal.pone.0071425
Young TE, Ling J, Geisler-Lee CJ, Tanguay RL, Caldwell C, Gallie DR (2001) Developmental and thermal regulation of the maize heat shock protein, HSP101. Plant Physiol 127:777–791
Acknowledgments
RCM, AS, and LDT acknowledge the Council of Scientific and Industrial Research (CSIR), the Government of India, New Delhi, for the Fellowship awards. Richa acknowledges the Department of Science and Technology, the Government of India, for the Inspire Fellowship Award. AG acknowledges the J.C. Bose Fellowship award grant from the Department of Science and Technology (DST), the Government of India, and financial support from the Center for Advanced Research and Innovation on Plant Stress and Developmental Biology, Department of Biotechnology (DBT), Government of India.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mishra, R.C., Richa, Singh, A. et al. Characterization of 5′UTR of rice ClpB-C/Hsp100 gene: evidence of its involvement in post-transcriptional regulation. Cell Stress and Chaperones 21, 271–283 (2016). https://doi.org/10.1007/s12192-015-0657-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-015-0657-1