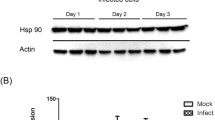
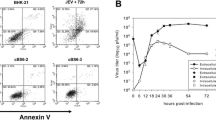
Abstract
Theiler’s murine encephalomyelitis virus (TMEV) is a positive-sense RNA virus belonging to the Cardiovirus genus in the family Picornaviridae. In addition to other host cellular factors and pathways, picornaviruses utilise heat shock proteins (Hsps) to facilitate their propagation in cells. This study investigated the localisation of Hsps 70 and 90 in TMEV-infected BHK-21 cells by indirect immunofluorescence and confocal microscopy. The effect of Hsp90 inhibitors novobiocin (Nov) and geldanamycin (GA) on the development of cytopathic effect (CPE) induced by infection was also examined. Hsp90 staining was uniformly distributed in the cytoplasm of uninfected cells but was found concentrated in the perinuclear region during late infection where it overlapped with the signal for non-structural protein 2C within the viral replication complex. Hsp70 redistributed into the vicinity of the viral replication complex during late infection, but its distribution did not overlap with that of 2C. Inhibition of Hsp90 by GA and Nov had a negative effect on virus growth over a 48-h period as indicated by no observable CPE in treated compared to untreated cells. 2C was detected by Western analysis of GA-treated infected cell lysates at doses between 0.01 and 0.125 μM, suggesting that processing of viral precursors was not affected in the presence of this drug. In contrast, 2C was absent in cell lysates of Nov-treated cells at doses above 10 μM, although CPE was evident 48 hpi. This is the first study describing the dynamic behaviour of Hsps 70 and 90 in TMEV-infected cells and to identify Hsp90 as an important host factor in the life cycle of this virus.






Similar content being viewed by others
Abbreviations
- ADP:
-
Adenosine diphosphate
- ATP:
-
Adenosine triphosphate
- CPE:
-
Cytopathic effect
- DAPI:
-
4’,6-Diamino-2-phenylindole dihydrochloride
- DMEM:
-
Dulbecco’s modified Eagle Medium
- DMSO:
-
Dimethyl sulphoxide
- DTT:
-
Dithiothreitol
- EDTA:
-
Ethylene diamine tetra-acetic acid
- EMCV:
-
Encephalomyelitis virus
- FMDV:
-
Foot-and-mouth disease virus
- GA:
-
Geldanamycin
- Hop:
-
Hsp70/Hsp90 organising protein
- hpi:
-
Hours post-infection
- Hsp:
-
Heat shock protein
- MOI:
-
Multiplicity of infection
- Nov:
-
Novobiocin
- PMSF:
-
Phenyl methyl sulphonyl fluoride
- PV:
-
Poliovirus
- TMEV:
-
Theiler’s murine encephalomyelitis virus
References
Abed Y, Boivin G (2008) New Saffold cardioviruses in 3 children, Canada. Emerg Infect Dis 14:834–836
Beck R, Nassal M (2003) Efficient Hsp-90-independent in vitro activation by Hsc70 and Hsp40 of duck hepatitis B virus reverse transcriptase, an assumed Hsp90 client protein. J Biol Chem 278:36128–36138
Blinkova O, Kapoor A, Victoria J, Wolfe N, Naeem A, Shaukat S, Sharif S, Alam MM, Angez M, Zaidi S, Delwart EL (2009) Cardioviruses are genetically diverse and common enteric infections in South Asian children. J Virol 83:4631–4641
Brahic M, Bureau J, Michiels T (2005) The genetics of the persistent infection caused by Theiler’s virus. Ann Rev Microbiol 59:279–298
Bukau B, Horwich AL (1998) The Hsp70 and Hsp60 chaperone machines. Cell 92:351–366
Burch AD, Weller SK (2005) Herpes simplex virus type 1 DNA polymerase requires the mammalian chaperone hsp90 for proper localization to the nucleus. J Virol 79(16):10740–10749
Castorena KM, Weeks SA, Stapleford KA, Cadwallader AM, Miller DJ (2007) A functional heat shock protein 90 chaperone is essential for efficient flock house virus RNA polymerase synthesis in Drosophila cells. J Virol 81(16):8412–8420
Chase G, Deng T, Fodor E, Wah Leung B, Mayer D, Schwemmle M, Brownlee G (2008) Hsp90 inhibitors reduce influenza virus replication in cell culture. Virology 377:431–439
Chiu ChY, Greninger AL, Kanada K, Kwok T, Fischer KF, Runkel C, Louie JK, Glaser CA, Yagi S, Schnuur DP, Haggerty TD, Parsonnet J, Ganem D, DeRisi JL (2008) Identification of cardioviruses related to Theiler’s murine encephalomyelitis virus in human infections. PNAS 105:14124–14129
Collier NC, Schlesinger MJ (1986) The dynamic state of heat shock proteins in chicken embryo fibroblasts. J Cell Biol 103:1495–1507
Connor JH, McKenzie MO, Parks GD, Lyles DS (2007) Antiviral activity and RNA polymerase degradation following Hsp90 inhibition in a range of negative strand viruses. Virology 362:109–119
Donnelly A, Blagg BSJ (2008) Novobiocin and additional inhibitors of the Hsp90 C-terminal nucleotide-binding pocket. Curr Med Chem 15:2702–2717
Drexler JF, de Souza Luna LK, Stocker A, Almeida PS, Ribeiro TCM, Petersen N, Herzog P, Pedroso C, Huppertz HI, da Costra RH, Baumgarte S, Drosten C (2008) Circulation of three lineages of a novel Saffold cardiovirus in humans. Emerg Infect Dis 14:1398–1405
Franke B, Margolin J (1981) Effect of novobiocin and other gyrase inhibitors on virus replication and DNA synthesis in Herpes Simplex virus type 1-infected BHK-21 cells. J Gen Virol 52:401–404
Frydman J (2001) Folding of newly translated proteins in vivo: the role of molecular chaperones. Annu Rev Biochem 70:603–647
Frydman J, Nimmesgern E, Ohtsuka K, Hartl FU (1994) Folding of nascent polypeptide chains in a high molecular mass assembly with molecular chaperones. Nature 370:111–117
Geller R, Vignuzzi M, Adino P, Freedman J (2007) Evolutionary constraints on chaperone-mediated folding provide an antiviral approach refractory to development of drug resistance. Genes Dev 21:195–205
Gething MJ, Sambrook J (1992) Protein folding in the cell. Nature 355:33–45
Grenert JP, Sullival WP, Faddan P, Haystead TAJ, Clark J, Mimnaugh E, Krutzsch H, Ochel H-J, Schultz TW, Sausville E, Neckers LM, Toft DO (1997) The amino-terminal domain of heat shock protein 90 (hsp90) that binds geldanamycin is an ATP/ADP switch domain that regulates hsp90 conformation. J Biol Chem 272:23843–23850
Guerrero CA, Bouyssounade D, Zarate S, Isa P, Lǒpez T, Espinosa R, Romero P, Mĕndez E, Lǒpez S, Aras CF (2002) Heat shock cognate protein 70 is involved in rotavirus cell entry. J Virol 76:4096–4102
Hartl F (1996) Molecular chaperones in cellular folding. Nature 381:571–579
Hu J, Toft DO, Seeger C (1997) Hepadnavirus assembly and reverse transcription require a multi-component chaperone complex which is incorporated into nucleocapsids. EMBO J 16(1):59–68
Hung JJ, Chung CS, Chang W (2002) Molecular chaperone Hsp90 is important for vaccinia virus growth in cells. J Virol 76(3):1379–1390
Jauka TI, Mutsvuguma L, Boshoff A, Edkins AL, Knox C (2010) Localisation of Theiler’s Murine Encephalomyelitis virus protein 2C to the Golgi apparatus using antibodies generated against a peptide region. J Virol Methods 168:162–169
Jones MS, Lukashov VV, Ganac RD, Schnurr DP (2007) Discovery of a novel human picornavirus in a stool sample from a pediatric patient presenting with fever of unknown origin. J Clin Microbiol 45:2144–2150
Kampmueller KM, Miller DJ (2005) The cellular chaperone heat shock protein 90 facilitates flock house virus RNA replication in Drosophila cells. J Virol 79:6827–6837
Knox C, Moffat K, Ali S, Ryan MD, Wileman T (2005) Foot and Mouth Disease Virus replication sites form next to the nucleus and close to the Golgi apparatus but exclude marker proteins associated with host membrane compartments. J Gen Virol 86:687–696
Kumar M, Mitra D (2005) Heat shock protein 40 is necessary for human immunodeficiency virus-1 Nef-mediated enhancement of viral gene expression and replication. J Biol Chem 280:40041–40050
Landini MP, Baldassarri B (1982) Early inhibition of cytomegalovirus replication by novobiocin. J Antimicrob Chemother 10:533–537
Larsen AK, Escargueil AE, Skladanowski A (2003) Catalytic topoisomerase II inhibitors in cancer therapy. Pharmacol Ther 99:167–181
Liang Z, Manoj Kumar AS, Jones MS, Knowles NJ, Lipton HL (2008) Phylogenetic analysis of the species Theilovirus: emerging murine and human pathogens. J Virol 82:11545–11554
Lührmann A, Thölke J, Behn I, Schumann J, Tiegs G, Hauschildt S (1998) Immunomodulating properties of the antibiotic novobiocin in human monocytes. Antimicrob Agents Chemother 42:1911–1916
Macejak DG, Sarnow P (1992) Association of heat shock protein 70 with enterovirus capsid precursor P1 in infected human cells. J Virol 66:1520–1527
Marcu MG, Chadli A, Bouhouche I, Catelli M, Neckers LM (2000a) The heat shock protein 90 antagonist novobiocin interacts with a previously unrecognised ATP-binding domain in the carboxyl terminus of the chaperone. J Biol Chem 275:37181–37186
Marcu MG, Schulte TW, Neckers L (2000b) Novobiocin and related coumarins and depletion of heat shock protein 90-dependent signalling proteins. J Natl Cancer Inst 92:242–248
Momose F, Naito T, Yano K, Sugimoto S, Morikawa Y, Nagata K (2002) Identification of Hsp90 as a stimulatory host factor involved in influenza virus RNA synthesis. J Biol Chem 277(47):45306–45314
Murphy PJM, Kanelakis KC, Galigniana MD, Morishima Y, Pratt WB (2001) Stoichiometry, abundance, and functional significance of the hsp90/hsp70-based multiprotein chaperone machinery in reticulocyte lysate. J Biol Chem 276:30092–30098
Murray L, Luke GA, Ryan MD, Wileman T, Knox C (2009) Amino acid substitutions in the 2C coding sequence of Theiler’s murine encephalomyelitis virus affect virus growth and alter protein distribution. Virus Res 144:74–82
Naito T, Momose F, Kawaguchi A, Nagata K (2007) Involvement of Hsp90 in assembly and nuclear import of influenza virus RNA polymerase subunits. J Virol 81:1339–1349
Oleszak EL, Chang JR, Friedman H, Katsetos CD, Platsoucas CD (2004) Theiler’s virus infection: a model for multiple sclerosis. Clin Microbiol Rev 17:174–207
Pelham HRB (1984) Hsp70 accelerates the recovery of nucleolar morphology after heat shock. EMBO J 3:3095–3100
Picard D (2002) Heat-shock protein 90, a chaperone for folding and regulation. Cell Mol Life Sci 59:1640–1648
Pratt WB, Toft DO (2003) Regulation of signaling protein function and trafficking by the hsp90/hsp70-based chaperone machinery. Exp Biol Med 228:111–133
Racaniello VR (2001) Picornaviridae: the viruses and their replication. In: Knipe DM, Howley PM, Griffin DE, Lamb RA, Martin MA, Roizman B, Straus SE (eds) Fields virology, 4th edn. Lippincott Williams and Wilkins Publishers, Philadelphia, pp 685–722
Reece RJ, Maxwell A (1991) DNA gyrase: structure and function. Crit Rev Biochem Mol Biol 26:335–375
Robinson MJ, Corbett AH, Osheroff N (1993) Effects of topoisomerase II-targeted drugs on enzyme-mediated DNA cleavage and ATP hydrolysis: evidence for distinct drug interaction domains on topoisomerase II. Biochemistry 32:3638–3643
Schneider C, Sepp-Lorenzino L, Nimmesgern E, Ouerfelli O, Danishefsky S, Rosen N, Hartl FU (1996) Pharmacologic shifting of a balance between protein refolding and degradation mediated by Hsp90. Proc Natl Acad Sci USA 93(25):14536–14540
Singh GK, Jayanarayan G, Dey CS (2005) Novobiocin induces apoptosis-like cell death in topoisomerase II over-expressing arsenite resistant Leishmania donovani. Mol Biochem Parasitol 141:57–69
Smith DR, McCarthya S, Chroviana A, Olingera G, Stossela A, Geisbertb TW, Hensleya LE, Connor JH (2010) Inhibition of heat-shock protein 90 reduces Ebola virus replication. Antivir Res 87:187–194
Snoeckx L, Cornelussen RN, Van Nieuwenhoven FA, Reneman RS, Van der Vusse GJ (2001) Heat shock proteins and cardiovascular pathophysiology. Physiol Rev 81:1461–1497
Stahl M, Retziaff M, Nassal M, Beck J (2007) Chaperone activation of the hepadnaviral reverse transcriptase for template RNA binding is established by the Hsp70 and stimulated by the Hsp90 system. Nucleic Acids Res 35(18):6124–6136
Stebbins CE, Russo AA, Schneider C, Rosen N, Hartl FU, Pavletich NP (1997) Crystal structure of an Hsp90–geldanamycin complex: targeting of a protein chaperone by an antitumor agent. Cell 89:239–250
Sullivan CS, Pipas JM (2001) The virus-chaperone connection. Virology 287:1–8
Sullivan W, Stensgard B, Caucutt G, Bartha B, McMahon N, Alnemri ES, Litwack G, Toft D (1997) Nucleotides and two functional states of hsp90. J Biol Chem 272:8007–8012
Sumiyoshi Y, Takuto N, Watanabe T, Kano K (1983) Inhibition of retrovirus RNA-dependent DNA polymerase by novobiocin and nalidixic acid. J Gen Virol 64:2329–2333
Terasawa K, Minami M, Minami Y (2005) Constantly updated knowledge of Hsp90. J Biochem 137(4):443–447
Tomita Y, Mizuno T, Diez J, Naito S, Ahlquist P, Ishikawa M (2003) Mutation of host DnaJ homolog inhibits brome mosaic virus negative-strand RNA synthesis. J Virol 77:2990–2997
Ujino S, Yamaguchi S, Shimotohno K, Takaku H (2009) Heat-shock protein 90 is essential for stabilization of the hepatitis C virus non-structural protein NS3. J Biol Chem 284(11):6841–6846
Vance LM, Moscufo N, Chow M, Heinz BA (1997) Poliovirus 2C region functions during encapsidation of viral RNA. J Virol 71:8759–8765
Vasconcelos DY, Cai XH, Oglesbee MJ (1998) Constitutive overexpression of the major inducible 70kDa heat shock protein mediates large plaque formation by measles virus. J Gen Virol 79:2239–2247
Xiao A, Wong J, Luo H (2010) Viral interaction with molecular chaperones: role in regulating viral infection. Arch Virol 155:1021–1031
Young JC, Agashe VR, Siegers K, Hartl FU (2004) Pathways of chaperone-mediated protein folding in the cytosol. Nat Rev Mol Cell Biol 5:781–791
Yun B, Huang W, Leach N, Hartson SD, Matts RL (2004) Novobiocin induces a distinct conformation of Hsp90 and alters Hsp90-co-chaperone-client interactions. Biochemistry 43:8217–8229
Zoll J, Hulshof SE, Lamke K, Lunel FV, Melchers JG, Schoondermark-van de Ven E, Roivainen M, Galama JMD, van Kuppeveld FJM (2009) Saffold virus, a human Theiler’s-like cardiovirus, is ubiquitous and causes infection early in life. PLoS Pathog 5:e1000416
Acknowledgements
We gratefully acknowledge T. Michiels for providing TMEV GDVII cDNA used for preparation of virus. This work was supported by a National Research Foundation grant (NRF, South Africa) and Joint Research Council funding (Rhodes University, South Africa).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mutsvunguma, L.Z., Moetlhoa, B., Edkins, A.L. et al. Theiler’s murine encephalomyelitis virus infection induces a redistribution of heat shock proteins 70 and 90 in BHK-21 cells, and is inhibited by novobiocin and geldanamycin. Cell Stress and Chaperones 16, 505–515 (2011). https://doi.org/10.1007/s12192-011-0262-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12192-011-0262-x