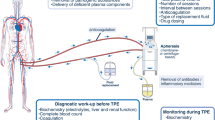
Abstract
Hereditary TTP (hTTP), termed Upshaw–Schulman syndrome, is an ultra-rare disorder caused by a severe deficiency of plasma ADAMTS13 activity that allows circulation of ultra-large von Willebrand factor (UL-VWF) multimers. The greatest risk for hTTP is in their first days after birth, when 35–50% of patients will have severe hemolysis, jaundice, and thrombocytopenia. It is often fatal without effective treatment. In utero, fetal blood flowing from the pulmonary artery through the ductus arteriosus (DA) to the aorta is under low-shear-force. At birth, blood flow through the DA reverses to a left-to-right shunt, and the diameter of the DA begins to decrease due to hyper-oxygenated blood and decreased plasma prostaglandin E2. This causes turbulent circulation that unfolds UL-VWF, allowing platelet aggregation. If the DA closes promptly, hTTP newborns survive, but if it remains patent, turbulent circulation persists, triggering microvascular thrombosis. hTTP is commonly diagnosed as hemolytic disease of the fetus and newborn (HDFN) caused by anti-red cell antibodies and treated with exchange blood transfusion, which prevents kernicterus even when the diagnosis of hTTP is missed. The diagnosis of newborn-onset hTTP should be considered because HDFN does not cause severe thrombocytopenia, which might be effectively treated with recombinant ADAMTS13.


Similar content being viewed by others
Data availability
No data was used for the research described in the article.
References
Moises KJ. Hemolytic disease of the fetus and newborn. In: Creasy RK, Iams JD, Lockwood CJ, Moore TR, editors. Creasy & Resnik’s maternal-fetal medicine; principles and practice. 6th ed. Philadephia, PA: Elsevier; 2009. p. 477–503.
De Haas M, Finning K, Massey E, Roberts DJ. Anti-D prophylaxis: past, present and future. Transfus Med. 2014;24(1):1–7.
Grundbacher FJ. The etiology of ABO hemolytic disease of the newborn. Transfusion. 1980;20(5):563–8.
Rath MEA, Smits-Wintjnes VEHJ, Oepkes D, van Zwet EW, van Kamp IL, Brand A, et al. Thrombocytopenia at birth in neonates with red cell alloimmune haemolytic disease. Vox Sang. 2012;102(3):228–33.
Matsumoto M, Miyakawa Y, Kokame K, Ueda Y, Wada H, Higasa S, et al. Diagnostic and treatment guidelines for thrombotic thrombocytopenic purpura (TTP) in Japan. Int J Hematol. 2023. https://doi.org/10.1007/s12185-023-03657-0.
Kokame K, Kokubo Y, Miyata T. Polymorphisms and mutations of ADAMTS13 in the Japanese population and estimation of the number of patients with Upshaw-Schulman syndrome. J Thromb Haemost. 2011;9(8):1654–6.
Zhao T, Fan S, Sun L. The global carrier frequency and genetic prevalence of Upshaw-Schulman syndrome. BMC Genomic Data. 2021;22:22–50.
Kremer-Hovinga JA, George JN. Hereditary thrombotic thrombocytopenic purpura. New Eng J Med. 2019;381(17):1653–62.
Stubbs MJ, Kendall G, Scully M. Recombinant ADAMTS13 in severe neonatal thrombotic thrombocytopenic purpura. New Engl J Med. 2022;387(25):2391–2.
Monnens LA, Retera RJ. Thrombotic thrombocytopenic purpura in a neonatal infant. J Pediatrics. 1967;71(1):118–23.
Kinoshita S, Yoshioka A, Park Y-D, Ishizashi H, Konno M, Funato M, et al. Upshaw-Schulman syndrome revisited: a concept of congenital thrombotic thrombocytopenic purpura. Int J Hematol. 2001;74:101–8.
Levy GG, Nichols WC, Lian EC, Foroud F, McClintick JN, McGee BM, et al. Mutations in a member of the ADAMTS gene family cause thrombotic thrombocytopenic purpura. Nature. 2001;413(6855):488–94.
Fujimura Y, Matsumoto M, Isonishi A, Yagi H, Kokame K, Soejima K, et al. Natural history of Upshaw-Schulman syndrome based on ADAMTS13 gene analysis in Japan. J Thromb Haemost. 2011;9(Suppl 1):283–301.
Fujimura Y, Kokame K, Yagi H, Isonishi A, Matsumtoto M, Miyata T. Hereditary deficiency of ADAMTS13 activity: Upshaw-Schulman syndrome. In: Rodgers GM, editor. ADAMTS13 biology and disease. Cham: Springer; 2015. p. 73–90.
Fujimura Y, Lämmle B, Tanabe S, Sakai K, Kimura T, Kokame K, et al. Patent ductus arteriosus generates neonatal hemolytic jaundice with thrombocytopenia in Upshaw-Schulman syndrome. Blood Adv. 2019;3(21):3191–5.
Sakai K, Hamada E, Kokame K, Matsumoto M. Congenital thrombotic thrombocytopenic purpura: genetics and emerging therapies. Ann Blood. 2023;8:24.
von Krogh AS, Quist-Paulsen P, Waage A, Langseth OO, Thorstensen K, Brudevold R, et al. High prevalence of hereditary thrombotic thrombocytopenic purpura in central Norway: from clinical observations to evidence. J Thromb Haemost. 2016;14:73–82.
Tarasco E, Butikofer L, Friedman KD, George JN, Hrachovinova I, Knöbl PN, et al. Annual incidence and severity of acute episodes in hereditary thrombotic thrombocytopenic purpura. Blood. 2021;137(25):3563–75.
Joly BS, Boisseau P, Roose E, Stepanian A, Biebuyck N, Hogan J, et al. ADAMTS13 gene mutations influence ADAMTS13 conformation and disease age-onset in the French cohort of Upshaw-Schulman syndrome. Thromb Haemost. 2018;118(11):1902–17.
Alwan F, Vendramin C, Liesner R, Clark A, Lester W, Dutt T, et al. Characterization and treatment of congenital thrombotic thrombocytopenic purpura. Blood. 2019;133(15):1644–51.
Liu J, Zhang Y, Li Z, Zhang L, Jian S, Wang C, et al. Early indicators of neonatal onset of hereditary thrombotic thrombocytopenic purpura. Res Pract Thromb Haemost. 2022;6(7): e12820. https://doi.org/10.1002/rth2.12820.eCollection.
Furlan M, Lämmle B. Aetiology and pathogenesis of thrombotic thrombocytopenic purpura and haemolytic uraemic syndrome: the role of von Willebrand factor-cleaving protease. Best Pract Res Clin Haemat. 2001;14(2):437–54.
George JN. Hereditary thrombotic thrombocytopenic purpura: the risk for death at birth. Res Pract Thromb Haemost. 2022;6: e12840. https://doi.org/10.1002/rth2.40.
Gao Y, Usha-Raj J. Regulation of the pulmonary circulation in the fetus and newborn. Physiol Rev. 2010;90(4):1291–335.
Jain A, Mohamed A, Kavanaugh B, Shah PS, Kuipers BCW, El-Khuiffash A, et al. Cardiopulmonary adaptation during the first day of life in human neonates. J Pediat. 2018;200(1):50–7.
Parkerson S, Philip R, Talati A, Sathanandam S. Management of patent ductus arteriosus in premature infants in 2020. Front Pediatr. 2021;8: 590578. https://doi.org/10.3389/fped.2020.590578.eCollection.
Schneider DJ, Moore JW. Patent ductus arteriosus. Circulation. 2006;114(17):1873–82.
Hung Y-C, Yeh J-L, Hsu J-H. Molecular mechanisms for regulating postnatal ductus arteriosus closure. Int J Mol Sci. 2018;19(7):1861. https://doi.org/10.3390/ijms19071861.
Echtler K, Stark K, Lorenz M, Kerstan S, Walch A, Jennen L, et al. Platelets contribute to postnatal occlusion of the ductus arteriosus. Nature Med. 2010;16(1):75–82. https://doi.org/10.1038/nm.2060.
Yokoyama U, Minamisawa S, Quan H, Ghatak S, Akaike T, Segi-Nisbhida E, et al. Chronic activation of the prostaglandin receptor EP4 promotes hyaluronan-mediated neointimal formation in the ductus arteriosus. J Clin Invest. 2006;116(11):3026–34. https://doi.org/10.1172/JCI28639.
Moake JL, Rudy CK, Troll JH, Weinstein MJ, Colannino NM, Azocar J, et al. Unusually large plasma factor VIII: von Willebrand factor multimers in chronic relapsing thrombotic thrombocytopenic purpura. New Eng J Med. 1982;307(23):1432–5.
Byrnes JJ, Khurana M. Treatment of thrombotic thrombocytopenic purpura with plasma. New Eng J Med. 1977;297:1386–9.
Schulman I, Pierce M, Lukens A, Currimbhoy Z. Studies on thrombopoiesis: I. A factor in normal plasma required for platelet production: chronic thrombocytopenia due to its deficiency. Blood. 1960;16(1):943–57.
Lian ECY, Harkness DR, Byrnes JJ, Wallach H, Nunez R. Presence of a platelet aggregating factor in the plasma of patients with thrombotic thrombocytopenic purpura (TTP) and its inhibition by normal plasma. Blood. 1979;53:333–8.
Upshaw JDJ. Congenital deficiency of a factor in normal plasma that reverses microangiopathic hemolysis and thrombocytopenia. New Eng J Med. 1978;298(24):1350–2.
Kalagara T, Moutsis T, Yang Y, Pappelbaum KI, Farken A, Cladder-Micus L, et al. The endothelial glycocalyx anchors von Willebrand factor fibers to the vascular endothelium. Blood adv. 2018;18:2347–57.
Dong J-F, Moake JL, Nolasco L, Bernardo A, Arceneaux W, Shrimpton CN, et al. ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood. 2002;100(12):4033–9.
Shao B, Nusrat S, George JN, Xia L. Aspirin prophylaxis for hereditary and acquired thrombotic thrombocytopenic purpura. Amer J Hematolo. 2022;97:E304–6.
Johnson SS, Montgomery RR, Hathaway W. Newborn factor VIII complex: elevated activities in term infants and alterations in electrophoretic mobility related to illness and activated coagulation. Brit J Haematol. 1981;47(4):597–660.
Andrew M, Paes B, Milner R, Johnston M, Mitchell L, Tollefsen DM, et al. Development of the human coagulation system in the full-term infant. Blood. 1987;70:165–72.
Thomas KB, Sutor AH, Altinkaya N, Grohmann A, Zehenter A, Leititis JU. von Willebrand factor-collagen binding activity is increased in newborns and infants. Acta Paediatr. 1995;84:697–9.
Tsai HM, Sarode R, Downes KA. Ultralarge von Willebrand factor multimers and normal ADAMTS13 activity in the umbilical cord blood. Thromb Res. 2002;108(2–3):121–5.
Schmugge M, Dunn MS, Amankwah KS, Blanchette VS, Freedman J, Rand ML. The activity of the von Willebrand factor-cleaving protease ADAMTS-13 in newborn infants. J Thromb Haemost. 2004;2:228–33.
Mannucci PM, Canciani MT, Forza I, Lussana F, Lattuada A, Rossi E. Changes in health and disease of the metalloprotease that cleaves von Willebrand factor. Blood. 2001;98(9):2730–5.
Hellstrom-Westas L, Ley D, Berg A-C, Kristoffersson A-C, Holmberg L. VWF-cleaving protease (ADAMTS13) in premature infants. Acta Paediatr. 2005;94(2):205–10.
Feys HB, Canciani MT, Peyvandi F, Deckmyn H, Vanhoorelbeke K, Mannucci PM. ADAMTS13 activity to antigen ratio in physiological and pathological conditions associated with an increased risk of thrombosis. Brit J Haematol. 2007;138(4):534–40.
Katz JA, Moake JL, McPherson PD, Weinstein MJ, Moise KJ, Carpenter RJ, et al. Relationship between human development and disappearance of unusually large von Willebrand factor multimers from plasma. Blood. 1989;73(7):1851–8.
Weinstein MJ, Blanchard R, Moake JL, Vosburgh E, Moise K. Fetal and neonatal von Willebrand factor (vWF) is unusually large and similar to the vWF in patients with thrombotic thrombocytopenic purpura. Br J Haematol. 1989;72(1):68–72.
Ferrer-Marin F, Sola-Visner M. Neonatal platelet physiology and implications for transfusion. Platelets. 2022;33(1):14–22.
Corby DG, O’Barr TP. Decreased alpha-adrenergic receptors in newborn platelets: cause of abnormal response to epinephrine. Dev Pharmacol Ther. 1981;2(4):215–25.
Schlagenhauf A, Schweintzger S, Birner-Gruenberger R, Leschnik B, Muntean W. Newborn platelets: lower levels of protease-activated receptors cause hypoaggregability to thrombin. Platelets. 2010;21(8):641–7. https://doi.org/10.3109/09537104.2010.504869.
Caparros-Perez E, Teruel-Montoya R, Lopez-Andreo MJ, Llanos MC, Rivera J, Palma-Barqueros V, et al. Comprehensive comparison of neonate and adult human platelet transcriptomes. PLoS ONE. 2017;12(8): e0183042. https://doi.org/10.1371/journal.pone.0183042.
Gross GP, Hathaway WE, McGaughey HR. Hyperviscosity in the neonate. J Pediatr. 1973;82(6):1004–12. https://doi.org/10.1016/s0022-3476(73)80433-0.
Kajino H, Chen Y-Q, Seidner SR, Waleh N, Mauray F, Roman C, et al. Factors that increase the contractile tone of the ductus arteriosus also regulate its anatomic remodeling. Am J Physiol Regul Integr Comp Physiol. 2001;281(1):R291–301. https://doi.org/10.1152/ajpregu.2001.281.1.R291.
Mydam J, Alok Rastogi A, Naheed ZJ. Base excess and hematocrit predict response to indomethacin in very low birth weight infants with patent ductus arteriosus. Ital J Pediatr. 2019;45(1):107. https://doi.org/10.1186/s13052-019-0706-y.
Tsujii N, Shiraishi I, Kokame K, Shima M, Fujimura Y, Takahashi Y, et al. Severe hemolysis and pulmonary hypertension in a neonate with Upshaw-Schulman syndrome. Pediatrics. 2016;138(6): e20161565. https://doi.org/10.1542/peds.2016-1565.
Reiter CD, Wang X, Tanus-Santos JE, Hogg N, Cannon RO 3rd, Schechter AN, et al. Cell-free hemoglobin limits nitric oxide bioavailability in sickle-cell disease. Nat Med. 2002;8(12):1383–9.
Amorosi EL, Ultmann JE. Thrombotic thrombocytopenic purpura: report of 16 cases and review of the literature. Medicine. 1966;45:136–60.
Wilkie ME, Stevens CR, Cunningham J, Blake D. Hypoxia-induced von Willebrand factor release is blocked by verapamil. Miner Electrolyte Metab. 1992;18(2–5):141–4.
Pinsky DJ, Naka Y, Liao H, Oz MC, Wagner DD, Mayadas TN, et al. Hypoxia-induced exocytosis of endothelial cell Weibel-Palade bodies. A mechanism for rapid neutrophil recruitment after cardiac preservation. J Clin Invest. 1996;97(2):493–500. https://doi.org/10.1172/JCI118440.
Fujimura Y, Holland LZ. COVID-19 microthrombosis: unusually large VWF multimers are a platform for activation of the alternative complement pathway under cytokine storm. Int J Hematol. 2022;115(4):457–69. https://doi.org/10.1007/s12185-022-03324-w.
Kato S, Matsumoto M, Matsuyama T, Isonishi A, Hiura H, Fujimura Y. Novel monoclonal antibody-based enzyme immunoassay for determining plasma levels of ADAMTS13 activity. Transfusion. 2006;46(8):1444–52. https://doi.org/10.1111/j.1537-2995.2006.00914.x.
Kokame K, Nobe Y, Kokubo Y, Okayama A, Miyata T. FRETS-VWF73, a first fluorogenic assay for ADAMTS13 activity. Br J Haematol. 2005;129(1):93–100. https://doi.org/10.1111/j.1365-2141.2005.05420.x.
Acknowledgements
The author acknowledges to Prof. James N. George of University of Oklahoma Health Science Center and Prof. Bernhard Lämmle of University Bern for their critical reading of this manuscript. The author also thanks to Dr. Saori Tanabe of Nihonkai General Hospital, Drs. Yoshihiko Sakurai and Tomoya Hayashi of Japanese Red Cross Kinki Block Blood Center, and Drs. Ayami Isonishi and Kazuya Sakai, and Prof. Masanori Matsumoto of Nara Medical University for collecting the data for this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
This project had no outside support. YF receives a patent royalty for ADAMTS13-act-ELISA from Alfresa Pham. (Japan).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Fujimura, Y. Hereditary TTP/Upshaw–Schulman syndrome: the ductus arteriosus controls newborn survival. Int J Hematol (2024). https://doi.org/10.1007/s12185-024-03731-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12185-024-03731-1