Abstract
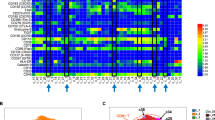
A 37-year-old man with refractory classical Hodgkin lymphoma (cHL) underwent PD-1 blockade therapy with nivolumab, which resulted in a partial response. However, treatment was discontinued due to immune-related adverse events (irAEs), including myasthenia gravis and myositis. Retreatment with nivolumab resulted in a complete metabolic response and hepatic irAE. Subsequently, nivolumab was administered at extended dosing intervals. Intermittent infusion of ten doses of nivolumab for a total dose of 2400 mg/body helped control the relapsed/refractory cHL over three years. During nivolumab treatment, disease progression and emergence of irAEs were associated with the proportion of CD8 + T cells expressing nivolumab-free PD-1 relative to the total number of CD8 + T cells. The findings in this nivolumab-sensitive patient highlight the clinical utility of monitoring immune cells expressing nivolumab-free PD-1 in patients with cHL who have been treated with nivolumab and have experienced irAEs.



Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Younes A, Santoro A, Shipp M, Zinzani PL, Timmerman JM, Ansell S, et al. Nivolumab for classical Hodgkin’s lymphoma after failure of both autologous stem-cell transplantation and Brentuximab vedotin: a multicentre, multicohort, single-arm phase 2 trial. Lancet Oncol. 2016;17:1283–94.
Ramos-Casals M, Brahmer JR, Callahan MK, Flores-Chávez A, Keegan N, Khamashta MA, et al. Immune-related adverse events of checkpoint inhibitors. Nat Rev Dis Primers. 2020;6:38.
Haverkos BM, Abbott D, Hamadani M, Armand P, Flowers ME, Merryman R, et al. PD-1 blockade for relapsed lymphoma post-allogeneic hematopoietic cell transplant: high response rate but frequent GVHD. Blood. 2017;130:221–8.
Brahmer JR, Drake CG, Wollner I, Powderly JD, Picus J, Sharfman WH, et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J Clin Oncol. 2010;28:3167–75.
Esfahani K, Elkrief A, Calabrese C, Lapointe R, Hudson M, Routy B, et al. Moving towards personalized treatments of immune-related adverse events. Nat Rev Clin Oncol. 2020;17:504–15.
Osa A, Uenami T, Koyama S, Fujimoto K, Okuzaki D, Takimoto T, et al. Clinical implications of monitoring nivolumab immunokinetics in non-small cell lung cancer patients. JCI Insight. 2018;3: e59125.
Naito Y, Osa A, Masuhiro K, Hirai T, Koyama S, Kumanogoh A. Monitoring PD-1-blocking antibodies bound to T cells derived from a drop of peripheral blood. J Vis Exp. 2020. https://doi.org/10.3791/60608.
Kameoka Y, Akagi T, Murai K, Noji H, Kato Y, Sasaki O, et al. Safety and efficacy of high-dose ranimustine (MCNU) containing regimen followed by autologous stem cell transplantation for diffuse large B-cell lymphoma. Int J Hematol. 2018;108:510–5.
Allouchery M, Lombard T, Martin M, Rouby F, Sassier M, Bertin C, et al. Safety of immune checkpoint inhibitor rechallenge after discontinuation for grade ≥ 2 immune-related adverse events in patients with cancer. J Immunother Cancer. 2020;8: e001622.
Allouchery M, Beuvon C, Pérault-Pochat MC, Roblot P, Puyade M, Martin M. Safety of immune checkpoint inhibitor resumption after interruption for immune-related adverse events, a narrative review. Cancers (Basel). 2022;14:955.
Araujo DV, Uchoa B, Soto-Castillo JJ, Furlan LL, Oliva M. When less may be enough: dose selection strategies for immune checkpoint inhibitors focusing on AntiPD-(L)1 agents. Target Oncol. 2022;17:253–70.
Jiang M, Hu Y, Lin G, Chen C. Dosing regimens of immune checkpoint inhibitors: attempts at lower dose, less frequency. Shorter Course Front Oncol. 2022;12: 906251.
Lepik KV, Fedorova LV, Kondakova EV, Zalyalov YR, Babenko EV, Lepik EE, et al. A Phase 2 study of nivolumab using a fixed dose of 40 mg (Nivo40) in patients with relapsed/refractory Hodgkin lymphoma. Hemasphere. 2020;4: e480.
Armand P, Engert A, Younes A, Fanale M, Santoro A, Zinzani PL, et al. Nivolumab for relapsed/refractory classic hodgkin lymphoma after failure of autologous hematopoietic cell transplantation: extended follow-up of the multicohort single-arm phase II CheckMate 205 trial. J Clin Oncol. 2018;36:1428–39.
Damoiseaux J. The IL-2 - IL-2 receptor pathway in health and disease: The role of the soluble IL-2 receptor. Clin Immunol. 2020;218: 108515.
Takai R, Funakoshi Y, Suto H, Nagatani Y, Imamura Y, Toyoda M, et al. Serum soluble interleukin-2 receptor as a potential biomarker for immune-related adverse events. Anticancer Res. 2021;41:1021–6.
Kamphorst AO, Wieland A, Nasti T, Yang S, Zhang R, Barber DL, et al. Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science. 2017;355:1423–7.
Green MR, Monti S, Rodig SJ, Juszczynski P, Currie T, O’Donnell E, et al. Integrative analysis reveals selective 9p24.1 amplification, increased PD-1 ligand expression, and further induction via JAK2 in nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Blood. 2010;116:3268–77.
Yamamoto R, Nishikori M, Kitawaki T, Sakai T, Hishizawa M, Tashima M, et al. PD-1-PD-1 ligand interaction contributes to immunosuppressive microenvironment of Hodgkin lymphoma. Blood. 2008;111:3220–4.
Veatch JR, Singhi N, Jesernig B, Paulson KG, Zalevsky J, Iaccucci E, et al. Mobilization of pre-existing polyclonal T cells specific to neoantigens but not self-antigens during treatment of a patient with melanoma with bempegaldesleukin and nivolumab. J Immunother Cancer. 2020;8: e001591.
Acharya N, Madi A, Zhang H, Klapholz M, Escobar G, Dulberg S, et al. Endogenous glucocorticoid signaling regulates CD8+ T cell differentiation and development of dysfunction in the tumor microenvironment. Immunity. 2020;53:658-71.e6.
Taves MD, Ashwell JD. Glucocorticoids in T cell development, differentiation and function. Nat Rev Immunol. 2021;21:233–43.
Ogasawara R, Hashimoto D, Sugita J, Yamawaki F, Naka T, Mitsuhashi T, et al. Loss of nivolumab binding to T cell PD-1 predicts relapse of Hodgkin lymphoma. Int J Hematol. 2020;111:475–9.
Acknowledgements
We would like to thank Dr. Haruna Miyazawa for the neurological examinations and Editage (www.editage.com) for English language editing.
Funding
This study was financially supported by the JSPS KAKENHI (grant number: JP19K08873).
Author information
Authors and Affiliations
Contributions
SK conducted the study, analyzed the data, and wrote the manuscript. YS performed flow cytometry. YT, TS, KA, TM, AYA, YO, RS, TO, and SI conducted the study and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Under an intermittent infusion of nivolumab, disease progression and the emergence of irAEs were associated with the proportion of CD8+ T cells expressing nivolumab-free PD-1.
About this article
Cite this article
Kowata, S., Seki, Y., Tsukushi, Y. et al. Association of CD8 + T cells expressing nivolumab-free PD-1 with clinical status in a patient with relapsed refractory classical Hodgkin lymphoma. Int J Hematol 118, 751–757 (2023). https://doi.org/10.1007/s12185-023-03644-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-023-03644-5