Abstract
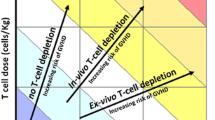
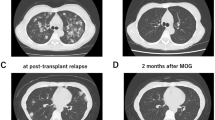
Mogamulizumab (Mog) is effective against adult T-cell leukemia–lymphoma (ATL), but as we reported previously, Mog increases the incidence of severe acute GVHD when administered before allogeneic hematopoietic cell transplantation (allo-HCT). Here, we report the cases of two ATL patients who did not develop acute GVHD despite receiving Mog before allo-HCT. Case 1: a 63-year-old female who underwent allo-HCT from an HLA-matched donor 2 months after the last dose of Mog. Case 2: a 47-year-old male with ATL that relapsed 3 months after first allo-HCT. He received eight doses of Mog and underwent a second allo-HCT from a haploidentical donor 4 months after the last dose of Mog. Mog blood levels were measured and lymphocytes analyzed by mass cytometry. Mog blood levels measured before starting the conditioning regimens were low. A small proportion of regulatory T cells (Tregs) was detected before and shortly after allo-HCT. When using Mog before allo-HCT, it is important to consider the number of Mog doses and the interval from the last dose of Mog to allo-HCT. Analyzing Mog blood levels and Treg counts before and after allo-HCT should also be useful.


Similar content being viewed by others
References
Cook LB, Fuji S, Hermine O, Bazarbachi A, Ramos JC, Ratner L, et al. Revised adult T-cell leukemia–lymphoma international consensus meeting report. J Clin Oncol. 2019;37(8):677–87.
Kanda J, Hishizawa M, Utsunomiya A, Taniguchi S, Eto T, Moriuchi Y, et al. Impact of graft-versus-host disease on outcomes after allogeneic hematopoietic cell transplantation for adult T-cell leukemia: a retrospective cohort study. Blood. 2012;119(9):2141–8.
Fuji S, Yamaguchi T, Inoue Y, Utsunomiya A, Moriuchi Y, Uchimaru K, et al. Development of a modified prognostic index for patients with aggressive adult T-cell leukemia–lymphoma aged 70 years or younger: possible risk-adapted management strategies including allogeneic transplantation. Haematologica. 2017;102(7):1258–65.
Fuji S, Kurosawa S, Inamoto Y, Murata T, Utsunomiya A, Uchimaru K, et al. Role of up-front allogeneic hematopoietic stem cell transplantation for patients with aggressive adult T-cell leukemia–lymphoma: a decision analysis. Bone Marrow Transplant. 2018;53(7):905–8.
Ishitsuka K, Yurimoto S, Tsuji Y, Iwabuchi M, Takahashi T, Tobinai K. Safety and effectiveness of mogamulizumab in relapsed or refractory adult T-cell leukemia–lymphoma. Eur J Haematol. 2019;102(5):407–15.
Michael M, Shimoni A, Nagler A. Regulatory T cells in allogeneic stem cell transplantation. Clin Dev Immunol. 2013;2013:608951.
Inoue Y, Fuji S, Tanosaki R, Fukuda T. Pretransplant mogamulizumab against ATLL might increase the risk of acute GVHD and non-relapse mortality. Bone Marrow Transplant. 2016;51(5):725–7.
Fuji S, Inoue Y, Utsunomiya A, Moriuchi Y, Uchimaru K, Choi I, et al. Pretransplantation Anti-CCR4 antibody mogamulizumab against adult T-cell leukemia/lymphoma is associated with significantly increased risks of severe and corticosteroid-refractory graft-versus-host disease, nonrelapse mortality, and overall mortality. J Clin Oncol. 2016;34(28):3426–33.
Katsuya H, Yamanaka T, Ishitsuka K, Utsunomiya A, Sasaki H, Hanada S, et al. Prognostic index for acute- and lymphoma-type adult T-cell leukemia/lymphoma. J Clin Oncol. 2012;30(14):1635–40.
Fuji S, Shindo T. Friend or foe? Mogamulizumab in allogeneic hematopoietic stem cell transplantation for adult T-cell leukemia/lymphoma. Stem Cell Investig. 2016;3:70.
Yamamoto K, Utsunomiya A, Tobinai K, Tsukasaki K, Uike N, Uozumi K, et al. Phase I study of KW-0761, a defucosylated humanized anti-CCR4 antibody, in relapsed patients with adult T-cell leukemia–lymphoma and peripheral T-cell lymphoma. J Clin Oncol. 2010;28(9):1591–8.
Inoue Y, Endo S, Matsuno N, Kikukawa Y, Shichijo T, Koga K, et al. Safety of mogamulizumab for relapsed ATL after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2019;54(2):338–42.
Stem Cell Trialists’ Collaborative G. Allogeneic peripheral blood stem-cell compared with bone marrow transplantation in the management of hematologic malignancies: an individual patient data meta-analysis of nine randomized trials. J Clin Oncol. 2005;23(22):5074–87.
Flowers ME, Inamoto Y, Carpenter PA, Lee SJ, Kiem HP, Petersdorf EW, et al. Comparative analysis of risk factors for acute graft-versus-host disease and for chronic graft-versus-host disease according to National Institutes of Health consensus criteria. Blood. 2011;117(11):3214–9.
Tanosaki R, Uike N, Utsunomiya A, Saburi Y, Masuda M, Tomonaga M, et al. Allogeneic hematopoietic stem cell transplantation using reduced-intensity conditioning for adult T cell leukemia/lymphoma: impact of antithymocyte globulin on clinical outcome. Biol Blood Marrow Transplant. 2008;14(6):702–8.
Acknowledgements
This research was partially supported by JSPS KAKENHI Grant Number 19K17861 and the Practical Research for Innovative Cancer Control Program of the Japan Agency for Medical Research and Development (20ck0106616h0001).
Author information
Authors and Affiliations
Contributions
YI, NN, MM, KI, KN, and MM participated in research design, data analysis, and writing of the paper. MM, MW, AY, and MI. participated in data analysis and writing of the paper. All authors approved the submission of this study.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Nosaka has received research funds from Kyowa Kirin, consulting fees from Kyowa Kirin, and honoraria from Kyowa Kirin and Celgene. Dr. Matsuoka has received honoraria from Kyowa Kirin Co. The other authors declare no conflicts of interest associated with this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Inoue, Y., Nishimura, N., Murai, M. et al. Prevention of acute graft-versus-host disease in adult T-cell leukemia–lymphoma patients who received mogamulizumab before allogeneic hematopoietic cell transplantation. Int J Hematol 115, 435–439 (2022). https://doi.org/10.1007/s12185-021-03250-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03250-3