Abstract
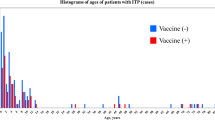
Childhood vaccine-associated immune thrombocytopenia (ITP) has a mostly favorable prognosis. To identify factors associated with prognosis, a retrospective survey was conducted with children with ITP who were registered in the Japanese Society of Pediatric Hematology/Oncology registry from 2008 to 2011. A total of 477 patients were categorized into four groups by event preceding ITP onset: vaccine-precedence (VP; n = 43), vaccine/infection-precedence (VIP; n = 34), infection-precedence (IP; n = 162), and no vaccine/infection-precedence (NVI; n = 238). Compared to IP and NVI, VP and VIP were significantly younger at diagnosis, with the age distribution peaking at infancy, and more frequently had favorable prognosis. Time to platelet recovery to 100 × 103/µL was significantly faster for VP and VIP than NVI. Multivariate Cox regression analysis with sex, age at diagnosis, infection-precedence, and vaccine-precedence as variables revealed age < 36 months (HR 0.992, 95% CI 0.989–0.995; p < 0.001) and male sex (HR 0.770, 95% CI 0.623–0.952; p = 0.015) as associated factors, but not infection-precedence (p = 0.149) or vaccine-precedence (p = 0.650). In subgroup analysis in patients < 36 months, age at diagnosis (p < 0.001) was the only associated factor. Favorable prognosis of childhood vaccine-associated ITP is correlated with young age at vaccination, but not with vaccination itself.




Similar content being viewed by others
References
Cines DB, Bussel JB, Liebman HA, Luning Prak ET. The ITP syndrome: pathogenic and clinical diversity. Blood. 2009;113:6511–21.
Li J, Sullivan JA, Ni H. Pathophysiology of immune thrombocytopenia. Curr Opin Hematol. 2018;25:373–81.
Kashiwagi H, Tomiyama Y. Pathophysiology and management of primary immune thrombocytopenia. Int J Hematol. 2013;98:24–33.
Marini I, Bakchoul T. Pathophysiology of autoimmune thrombocytopenia: current insight with a specifically examine thrombopoiesis. Haemostaseologie. 2019;39:227–37.
Wraith DC, Goldman M, Lambert PH. Vaccination and autoimmune disease: what is the evidence? Lancet. 2003;362:1659–66.
Toussirot É, Bereau M. Vaccination and induction of autoimmune diseases. Inflamm Allergy Drug Targets. 2015;14:94–8.
Perricone C, Ceccarelli F, Nesher G, Borella E, Odeh Q, Conti F, et al. Immune thrombocytopenic purpura (ITP) associated with vaccinations: a review of reported cases. Immunol Res. 2014;60:226–35.
Saitoh A, Okabe N. Progress and challenges for the Japanese immunization program: beyond the “vaccine gap.” Vaccine. 2018;36:4582–8.
Changes in the immunization schedule recommended by the Japan Pediatric Society October 1, 2020. Japan Pediatric Society. http://www.jpeds.or.jp/uploads/files/2020%20English%20JPS%20Immunization%20Schedule.pdf
France EK, Glanz J, Xu S, Hambidge S, Yamasaki K, Black SB, et al. Risk of immune thrombocytopenic purpura after measles–mumps–rubella immunization in children. Pediatrics. 2008;121:e687–92.
Mantadakis E, Farmaki E, Buchanan GR. Thrombocytopenic purpura after measles–mumps–rubella vaccination: a systematic review of the literature and guidance for management. J Pediatr. 2010;156:623–8.
Nieminen U, Peltola H, Syrjälä MT, Mäkipernaa A, Kekomäki R. Acute thrombocytopenic purpura following measles, mumps and rubella vaccination. A report on 23 patients. Acta Paediatr. 1993;82:267–70.
O’Leary ST, Glanz JM, McClure DL, Akhtar A, Daley MF, Nakasato C, et al. The risk of immune thrombocytopenia purpura after vaccination in children and adolescents. Pediatrics. 2012;129:248–55.
Grimaldi-Bensouda L, Michel M, Aubrun E, Leighton P, Viallard JF, Adoue D, et al. A case-control study to assess the risk of immune thrombocytopenia associated with vaccines. Blood. 2012;120:4938–44.
Rodeghiero F, Stasi R, Gernsheimer T, Michel M, Provan D, Arnold DM, et al. Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group. Blood. 2009;113:2386–93.
Provan D, Stasi R, Newland AC, Blanchette VS, Bolton-Maggs P, Bussel JB, et al. International consensus report on the investigation and management of primary immune thrombocytopenia. Blood. 2010;115:168–86.
Shirahata A, Fujisawa K, Ishii E, Ohta S, Sako M, Takahashi Y, et al. A nationwide survey of newly diagnosed childhood idiopathic thrombocytopenic purpura in Japan. J Pediatr Hematol Oncol. 2009;3:27–32.
Schattner A. Consequence or coincidence? The occurrence, pathogenesis and significance of autoimmune manifestations after viral vaccines. Vaccine. 2005;23:3876–86.
Kitazawa J, Ohtani K, Shiotani M, Satoh A, Sugimoto K, Nomura Y, et al. Idiopathic thrombocytopenic purpura following measles–rubella-conjugated vaccine. Pediatr Int. 2007;49:688–9.
Sadarangani M, Scheifele DW, Halperin SA, Vaudry W, Le Saux N, Tsang R, et al. The impact of the meningococcal serogroup C conjugate vaccine in Canada between 2002 and 2012. Clin Infect Dis. 2014;59:1208–15.
Yokomichi H, Tanaka-Taya K, Koshida R, Nakano T, Yasui Y, Mori M, et al. Immune thrombocytopenic purpura risk by live, inactivated and simultaneous vaccinations among Japanese adults, children and infants: a matched case-control study. Int J Hematol. 2020;112:105–14.
Kubota M, Adachi S, Usami I, Okada M, Kitoh T, Shiota M, et al. Characterization of chronic idiopathic thrombocytopenic purpura in Japanese children: a retrospective multi-center study. Int J Hematol. 2010;91:252–7.
Tamminga R, Berchtold W, Bruin M, Buchanan GR, Kühne T. Possible lower rate of chronic ITP after IVIG for acute childhood ITP an analysis from registry I of the Intercontinental Cooperative ITP Study Group (ICIS). Br J Haematol. 2009;146:180–4.
Grimaldi-Bensouda L, Nordon C, Leblanc T, Abenhaim L, Allali S, Armari-Alla C, et al. Childhood immune thrombocytopenia: a nationwide cohort study on condition management and outcomes. Pediatr Blood Cancer. 2017. https://doi.org/10.1002/pbc.26389.
Johnsen J. Pathogenesis in immune thrombocytopenia: new insights. Hematol Am Soc Hematol Educ Progr. 2012;2012:306–12.
Kucuksezer UC, Ozdemir C, Akdis M, Akdis CA. Influence of innate immunity on immune tolerance. Acta Med Acad. 2020;49:164–80.
Soyer OU, Akdis M, Ring J, Behrendt H, Crameri R, Lauener R, et al. Mechanisms of peripheral tolerance to allergens. Allergy. 2013;68:161–70.
Durand M, Segura E. The known unknowns of the human dendritic cell network. Front Immunol. 2015;6:129.
Sakakura M, Wada H, Tawara I, Nobori T, Sugiyama T, Sagawa N, et al. Reduced CD4+CD25+ T cells in patients with idiopathic thrombocytopenic purpura. Thromb Res. 2007;120:187–93.
Wang Q, Li J, Yu TS, Liu Y, Li K, Liu S, et al. Disrupted balance of CD4+ T-cell subsets in bone marrow of patients with primary immune thrombocytopenia. Int J Biol Sci. 2019;15:2798–814.
Nishimoto T, Kuwana M. CD4+CD25+Foxp3+ regulatory T cells in the pathophysiology of immune thrombocytopenia. Semin Hematol. 2013;50(Suppl 1):S43–9.
Tulic MK, Andrews D, Crook ML, Charles A, Tourigny MR, Moqbel R, et al. Changes in thymic regulatory T-cell maturation from birth to puberty: differences in atopic children. J Allergy Clin Immunol. 2012;129:199–206 ((e1–4)).
Lluis A, Depner M, Gaugler B, Saas P, Casaca VI, Raedler D, et al. Increased regulatory T-cell numbers are associated with farm milk exposure and lower atopic sensitization and asthma in childhood. J Allergy Clin Immunol. 2014;133:551–9.
Acknowledgements
We extend our sincere condolences to family, friends, and colleagues of Dr. Shigeru Ota, who passed away on February 20, 2019, at the age of 69, without seeing the completion of this dissertation. We extend our sincere thanks to Ms. Michiyo Kanda for her efforts in data processing.
Author information
Authors and Affiliations
Consortia
Contributions
JK conceived the study, analyzed the data, and helped write the manuscript. HN carried out data collection and data cleaning. KM, YT, AI, YS, KF, NM, TO, and EI wrote the manuscript. EI analyzed the platelet recovery data statistically. MI conceived and designed the study, analyzed the data further, and mainly wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Kitazawa, J., Nakadate, H., Matsubara, K. et al. Favorable prognosis of vaccine-associated immune thrombocytopenia in children is correlated with young age at vaccination: Retrospective survey of a nationwide disease registry. Int J Hematol 115, 114–122 (2022). https://doi.org/10.1007/s12185-021-03230-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03230-7