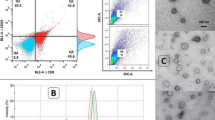
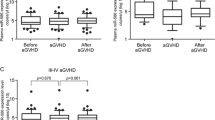
Abstract
The involvement of microRNAs in the regulation of hematopoietic stem cells paves the way for their use in the management of autologous HSC transplantation (AHSCT). We aimed to evaluate the predictive value of circulatory microRNAs in extracellular vesicles (EVs) and plasma in platelet and neutrophil engraftment. Circulatory miR-125b, mir-126, miR-150, and miR-155 expression was assessed in isolated EVs and plasma in samples collected from AHSCT candidates. Multivariate analysis, COX models, and ROC assessment were performed to evaluate the predictive values of these microRNAs in platelet and neutrophil engraftment. miR-155 expression following conditioning with other clinical factors such as chemotherapy courses after diagnosis was the most significant predictors of platelet/neutrophil engraftment. A CD34+ cell count ≥ 3.5 × 106/kg combined with miR-155 could be used as an engraftment predictor; however, in cases where the CD34+ cell count was < 3.5 × 106/kg, this parameter lost its predictive value for engraftment and could be replaced by miR-155. The correlation between miR-155 and platelet/neutrophil engraftment even with lower numbers of CD34+ cells suggests the importance of this microRNA in the prediction of AHSCT outcome. Moreover, miR-155 could be utilized in therapeutic approaches to provide a better outcome for patients undergoing AHSCT.





Similar content being viewed by others
References
Hübel K, Ostermann H, Glaß B, Noppeney R, Kron F, Kron A, et al. Plerixafor in non-Hodgkin’s lymphoma patients: a German analysis of time, effort and costs. Bone Marrow Transpl. 2019;54(1):123–9.
Snowden JA, Sharrack B, Akil M, Kiely DG, Lobo A, Kazmi M, et al. Autologous haematopoietic stem cell transplantation (aHSCT) for severe resistant autoimmune and inflammatory diseases–a guide for the generalist. Clin Med. 2018;18(4):329.
Giralt S, Costa L, Schriber J, DiPersio J, Maziarz R, McCarty J, et al. Optimizing autologous stem cell mobilization strategies to improve patient outcomes: consensus guidelines and recommendations. Biol Blood Marrow Tr. 2014;20(3):295–308.
Kim JS. Hematopoietic stem cell mobilization: current status and future perspective. Blood Res. 2017;52(2):79–81.
Calvi LM, Link DC. The hematopoietic stem cell niche in homeostasis and disease. Blood. 2015;126(22):2443–51.
Roden C, Lu J. MicroRNAs in control of stem cells in normal and malignant hematopoiesis. Curr Stem Cell Rep. 2016;2(3):183–96.
Nowicki M, Szemraj J, Wierzbowska A, Misiewicz M, Małachowski R, Pluta A, et al. Slower engraftment in patients with high expression of miRNA-15a, miRNA-16, miRNA-126, miRNA-146a, miRNA-223 prior to autologous stem cell transplantation and at early time after transplantation. Blood. 2016;128(22):5717. https://doi.org/10.1182/blood.V128.22.5717.5717.
Qiu G, Zheng G, Ge M, Wang J, Huang R, Shu Q, et al. Mesenchymal stem cell-derived extracellular vesicles affect disease outcomes via transfer of microRNAs. Stem Cell Res Ther. 2018;9(1):320.
Wang S-Y, Hong Q, Zhang C-Y, Yang Y-J, Cai G-Y, Chen X-M. miRNAs in stem cell-derived extracellular vesicles for acute kidney injury treatment: comprehensive review of preclinical studies. Stem Cell Res Ther. 2019;10(1):1–7.
Bejanyan N, Brunstein CG, Cao Q, Lazaryan A, Luo X, Curtsinger J, et al. Delayed immune reconstitution after allogeneic transplantation increases the risks of mortality and chronic GVHD. Blood Adv. 2018;2(8):909–22.
Song G-Y, Jung S-H, Ahn S-Y, Jung S-Y, Yang D-H, Ahn J-S, et al. Optimal chemo-mobilization for the collection of peripheral blood stem cells in patients with multiple myeloma. BMC Cancer. 2019;19(1):59.
Wang S, Min J, Yu Y, Yin L, Wang Q, Shen H, et al. Differentially expressed miRNAs in circulating exosomes between atrial fibrillation and sinus rhythm. J Thorac Dis. 2019;11(10):4337.
Zhang L, Li H, Yuan M, Li D, Sun C, Wang G. Serum exosomal microRNAs as potential circulating biomarkers for endometriosis. Dis Markers. 2020;2020:2456340.
Tian F, Shen Y, Chen Z, Li R, Ge Q. No significant difference between plasma miRNAs and plasma-derived exosomal miRNAs from healthy people. Biomed Res Int. 2017;2017:1304816.
Montagnana M, Benati M, Tagetti A, Raffaelli R, Danese E, Zatti N, et al. Evaluation of circ_100219 and miR-135b in serum and exosomes of healthy pregnant women. J Matern Fetal Neonatal Med. 2019;. https://doi.org/10.1080/14767058.2019.1689556.
Salvucci O, Jiang K, Gasperini P, Maric D, Zhu J, Sakakibara S, et al. MicroRNA126 contributes to granulocyte colony-stimulating factor-induced hematopoietic progenitor cell mobilization by reducing the expression of vascular cell adhesion molecule 1. Haematologica. 2012;97(6):818–26.
Itkin T, Kumari A, Schneider E, Gur-Cohen S, Ludwig C, Brooks R, et al. MicroRNA-155 promotes G-CSF-induced mobilization of murine hematopoietic stem and progenitor cells via propagation of CXCL12 signaling. Leukemia. 2017;31(5):1247–50.
Tano N, Kim HW, Ashraf M. microRNA-150 regulates mobilization and migration of bone marrow-derived mononuclear cells by targeting Cxcr4. PLoS ONE. 2011;6(10):e23114.
Ooi AL, Sahoo D, Adorno M, Wang Y, Weissman IL, Park CY. MicroRNA-125b expands hematopoietic stem cells and enriches for the lymphoid-balanced and lymphoid-biased subsets. Proc Natl Acad Sci USA. 2010;107(50):21505–10.
Adams BD, Guo S, Bai H, Guo Y, Megyola CM, Cheng J, et al. An in vivo functional screen uncovers miR-150-mediated regulation of hematopoietic injury response. Cell Rep. 2012;2(4):1048–60. https://doi.org/10.1016/j.celrep.2012.09.014.
Itkin T, Ludin A, Gur-Cohen S, Ludwig C, Brooks R, Golan K, et al. Microrna-155 promotes hematopoietic stem and progenitor cell mobilization and proliferation. Blood. 2012;120(21):214. https://doi.org/10.1182/blood.V120.21.214.214.
Yamaguchi J, Chinen Y, Takimoto-Shimomura T, Nagata H, Muramatsu A, Kuriyama K, et al. Prediction of delayed platelet engraftment after autologous stem cell transplantation for B-cell non-Hodgkin lymphoma. Leuk Lymphoma. 2019;60(14):3434–41.
Weaver CH, Hazelton B, Birch R, Palmer P, Allen C, Schwartzberg L, et al. An analysis of engraftment kinetics as a function of the CD34 content of peripheral blood progenitor cell collections in 692 patients after the administration of myeloablative chemotherapy. Blood. 1995;86(10):3961–9.
Takizawa H, Kubo-Akashi C, Nobuhisa I, Kwon S-M, Iseki M, Taga T, et al. Enhanced engraftment of hematopoietic stem/progenitor cells by the transient inhibition of an adaptor protein. Lnk Blood. 2006;107(7):2968–75.
Zhang Y, Roos M, Himburg H, Termini CM, Quarmyne M, Li M, et al. PTPσ inhibitors promote hematopoietic stem cell regeneration. Nat Commun. 2019;10(1):1–15.
Noach EJ, Ausema A, van Os R, Akkerman I, Koopal S, Weersing E, et al. Chemotherapy prior to autologous bone marrow transplantation impairs long-term engraftment in mice. Exp Hematol. 2003;31(6):528–34.
Lucas D, Scheiermann C, Chow A, Kunisaki Y, Bruns I, Barrick C, et al. Chemotherapy-induced bone marrow nerve injury impairs hematopoietic regeneration. Nat Med. 2013;19(6):695.
Kenyon M, Babic A. Engraftment, graft failure, and rejection—the european blood and marrow transplantation textbook for nurses: under the auspices of EBMT. 2018. Available from: https://www.ncbi.nlm.nih.gov/books/NBK543668/doi:10.1007/978-3-319-50026-3.
Lee K, Jung S, Kim S, Jang J, Kim K, Kim W, et al. Incidence and risk factors of poor mobilization in adult autologous peripheral blood stem cell transplantation: a single-centre experience. Vox Sang. 2014;107(4):407–15.
Weaver C, Birch R, Greco F, Schwartzberg L, McAneny B, Moore M, et al. Mobilization and harvesting of peripheral blood stem cells: randomized evaluations of different doses of filgrastim. Br J Haematol. 1998;100(2):338–47.
Wolf JTD, Imhoff GV, Huls GA, Vellenga E. Influence of different dosages of cyclophosphamide on stem cell mobilization and engraftment in newly diagnosed multiple myeloma patients treated with a thalidomide containing regimen. Blood. 2010;116(21):3494. https://doi.org/10.1182/blood.V116.21.3494.3494.
Xu Y, Yao Y, Yao W, Jin S, Yan L, Shang J, et al. Application of the conditioning regimen with busulfan and cyclophosphamide in autologous hematopoietic stem cell transplantation in multiple myeloma. Blood. 2017;130(Supplement 1):5517.
Reshef R, Porter D. Reduced-intensity conditioned allogeneic SCT in adults with AML. Bone Marrow Transpl. 2015;50(6):759–69.
Acknowledgements
The authors would like to thank staff of BMT department of Taleghani Hospital, Tehran for their general support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Rafiee, M., Farsani, M.A., Amiri, V. et al. Circulatory miR-155 correlation with platelet and neutrophil recovery after autologous hematopoietic stem cell transplantation, a multivariate analysis. Int J Hematol 114, 235–245 (2021). https://doi.org/10.1007/s12185-021-03154-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03154-2