Abstract
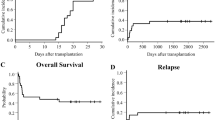
The optimal conditioning regimen for stem cell transplantation in elderly patients remains to be established. We developed a novel preparative regimen using fludarabine 180 mg/m2, intravenous busulfan 12.8 mg/m2, cytarabine 8 g/m2, and 4-Gy total body irradiation before cord blood transplantation (CBT) in patients older than 55 years with various hematological malignancies. All but one patient received graft-versus-host disease (GVHD) prophylaxis consisting of cyclosporine (CsA) and short-term methotrexate (sMTX). Thirty-three patients were included in this study, with a median age of 64 years (range 56–70). The disease risk index was high or very high in 67% of patients, and 73% had a disease status other than complete remission. The probabilities of overall survival and disease-free survival at 3 years were 60 and 57%, respectively. The cumulative incidences of relapse and non-relapse mortality at 3 years were 18 and 25%, respectively. Regimen-related toxicities were generally tolerable. Disease-free survivors (n = 20) stopped immunosuppressants at a median of 7.4 months (range 2.6–25.0), in all cases by the time of the last follow-up. In conclusion, this highly myeloablative conditioning regimen resulted in a high probability of disease-free, GVHD-free, immunosuppressant-free survival after single CBT.
(190 words)




Similar content being viewed by others
References
Muffly L, Pasquini MC, Martens M, Brazauskas R, Zhu X, Adekola K, et al. Increasing use of allogeneic hematopoietic cell transplantation in patients aged 70 years and older in the United States. Blood. 2017;130:1156–64.
He F, Cao Q, Lazaryan A, Brunstein C, Holtan S, Warlick E, et al. Allogeneic hematopoietic cell transplantation for older patients: prognosis determined by disease risk index. Biol Blood Marrow Transplant. 2017;23:1485–90.
Klepin HD, Geiger AM, Tooze JA, Kritchevsky SB, Williamson JD, Pardee TS, et al. Geriatric assessment predicts survival for older adults receiving induction chemotherapy for acute myelogenous leukemia. Blood. 2013;121:4287–94.
Wheatley K, Brookes CL, Howman AJ, Goldstone AH, Milligan DW, Prentice AG, et al. Prognostic factor analysis of the survival of elderly patients with AML in the MRC AML11 and LRF AML14 trials. Brit J Haematol. 2009;145:598–605.
Piccirillo JF, Vlahiotis A, Barrett LB, Flood KL, Spitznagel EL, Steyerberg EW. The changing prevalence of comorbidity across the age spectrum. Crit Rev Oncol Hemat. 2008;67:124–32.
Yamamoto H. Single cord blood transplantation in Japan; expanding the possibilities of CBT. Int J Hematol. 2019;110:39–49.
Horwitz ME, Morris A, Gasparetto C, Sullivan K, Long G, Chute J, et al. Myeloablative intravenous busulfan/fludarabine conditioning does not facilitate reliable engraftment of dual umbilical cord blood grafts in adult recipients. Biol Blood Marrow Transplant. 2008;14:591–4.
Arai Y, Kondo T, Shigematsu A, Takahashi S, Kobayashi T, Uchida N, et al. High-dose cytarabine added to CY/TBI improves the prognosis of cord blood transplantation for acute lymphoblastic leukemia in adults: a retrospective cohort study. Bone Marrow Transpl. 2016;51:1636–9.
Arai Y, Takeda J, Aoki K, Kondo T, Takahashi S, Onishi Y, et al. Efficiency of high-dose cytarabine added to CY/TBI in cord blood transplantation for myeloid malignancy. Blood. 2015;126:415–22.
Inamoto Y, Kimura F, Kanda J, Sugita J, Ikegame K, Nakasone H, et al. Comparison of graft-versus-host disease-free, relapse-free survival according to a variety of graft sources: antithymocyte globulin and single cord blood provide favorable outcomes in some subgroups. Haematologica. 2016;101:1592–602.
Robin M, Ruggeri A, Labopin M, Niederwieser D, Tabrizi R, S Guillermo et al. Comparison of Unrelated Cord Blood and Peripheral Blood Stem Cell Transplantation in Adults with Myelodysplastic Syndrome after Reduced-Intensity Conditioning Regimen: A Collaborative Study from Eurocord (Cord blood Committee of Cellular Therapy & Immunobiology Working Party of EBMT) and Chronic Malignancies Working Party. Biol Blood Marrow Transplant. 2015;21:489–95.
Kurosawa S, Yamaguchi T, Oshima K, Yoshimura K, Misaki Y, Gomyo A, et al. Resolved versus active chronic graft-versus-host disease: Impact on post-transplant quality of life. Biol Blood Marrow Transplant. 2019;25:1851–8.
Pidala J, Kurland B, Chai X, Majhail N, Weisdorf DJ, Pavletic S, et al. Patient-reported quality of life is associated with severity of chronic graft-versus-host disease as measured by NIH criteria: report on baseline data from the Chronic GVHD Consortium. Blood. 2011;117(17):4651–7.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V, et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant. 2009;15:1628–33.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 Consensus conference on acute GVHD grading. Bone Marrow Transpl. 1995;15:825–8.
Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: I. The 2014 Diagnosis and Staging Working Group Report. Biol Blood Marrow Transplant. 2015;21:389–401.
Kawamura K, Nakasone H, Kurosawa S, Yoshimura K, Misaki Y, Gomyo A, et al. Refractory graft-versus-host disease-free, relapse-free survival as an accurate and easy-to-calculate endpoint to assess the long-term transplant success. Biol Blood Marrow Transplant. 2018;24:1521–6.
Mohty M, Malard F, Abecassis M, Aerts E, Alaskar AS, Aljurf M, et al. Revised diagnosis and severity criteria for sinusoidal obstruction syndrome/veno-occlusive disease in adult patients: a new classification from the European Society for Blood and Marrow Transplantation. Bone Marrow Transpl. 2016;51:906–12.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier FB, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9.
Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123(23):3664–71.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transpl. 2013;48:452–8.
Ringdén O, Boumendil A, Labopin M, Canaani J, Beelen D, Ehninger G, et al. Outcome of allogeneic hematopoietic stem cell transplantation in patients over 69 years of age with acute myeloid leukemia: on behalf of the acute leukemia working party of the EBMT. Biol Blood Marrow Transplant. 2019;25:1975–83.
Konuma T, Tsukada N, Kanda J, Uchida N, Ohno Y, Miyakoshi S, et al. Comparison of transplant outcomes from matched sibling bone marrow or peripheral blood stem cell and unrelated cord blood in patients 50 years or older. Am J Hematol. 2016;91:E284–92.
Takahashi S, Ooi J, Tomonari A, Konuma T, Tsukada N, Oiwa-Monna M, et al. Comparative single-institute analysis of cord blood transplantation from unrelated donors with bone marrow or peripheral blood stem-cell transplants from related donors in adult patients with hematologic malignancies after myeloablative conditioning regimen. Blood. 2007;109:1322–30.
Konuma T, Ooi J, Uchida N, Ogawa H, Ohashi K, Kanamori H, et al. Granulocyte colony-stimulating factor combined regimen in cord blood transplantation for acute myeloid leukemia: a nationwide retrospective analysis in Japan. Haematologica. 2014;99:e264–8.
Konuma T, Kato S, Ooi J, Oiwa-Monna M, Ebihara Y, Mochizuki S, et al. Single-unit cord blood transplantation after granulocyte colony-stimulating factor-combined myeloablative conditioning for myeloid malignancies not in remission. Biol Blood Marrow Transplant. 2014;20:396–401.
Bredeson C, LeRademacher J, Kato K, Dipersio JF, Agura E, Devine SM, et al. Prospective cohort study comparing intravenous busulfan to total body irradiation in hematopoietic cell transplantation. Blood. 2013;122:3871–8.
Copelan EA, Hamilton BK, Avalos B, Ahn KW, Bolwell BJ, Zhu X, et al. Better leukemia-free and overall survival in AML in first remission following cyclophosphamide in combination with busulfan compared with TBI. Blood. 2013;122:3863–70.
Fujiwara S, Kanda J, Tatara R, Ogawa H, Fukuda T, Okumura H, et al. Clinical significance of low-dose total body irradiation in HLA-mismatched reduced-intensity stem cell transplantation. Bone Marrow Transpl. 2019;54:1327–36.
Itonaga H, Ishiyama K, Aoki K, Aoki J, Ishikawa T, Uchida N, et al. Increased opportunity for prolonged survival after allogeneic hematopoietic stem cell transplantation in patients aged 60–69 years with myelodysplastic syndrome. Ann Hematol. 2019;98:1367–81.
Konuma T, Kato S, Isobe M, Mizusawa M, Oiwa-Monna M, Takahashi S, et al. Reduced-toxicity myeloablative conditioning consisting of fludarabine/busulfan/low-dose TBI/G-CSF-combined cytarabine in single cord blood transplantation for elderly patients with non-remission myeloid malignancies. Biol Blood Marrow Transplant. 2019;25:764–70.
Yamamoto H, Uchida N, Yuasa M, Kageyama K, Ota H, Kaji D, et al. A novel reduced-toxicity myeloablative conditioning regimen using full-dose busulfan, fludarabine, and melphalan for single cord blood transplantation provides durable engraftment and remission in nonremission myeloid malignancies. Biol Blood Marrow Transplant. 2016;22:1844–50.
Nakasone H, Fukuda T, Kanda J, Mori T, Yano S, Kobayashi T, et al. Impact of conditioning intensity and TBI on acute GVHD after hematopoietic cell transplantation. Bone Marrow Transpl. 2015;50:559–65.
Flowers ME, Inamoto Y, Carpenter PA, Lee SJ, Kiem HP, Petersdorf EW, et al. Comparative analysis of risk factors for acute graft-versus-host disease and for chronic graft-versus-host disease according to National Institutes of Health consensus criteria. Blood. 2011;117:3214–9.
Terakura S, Kuwatsuka Y, Yamasaki S, Wake A, Kanda J, Inamoto Y, et al. GvHD prophylaxis after single-unit reduced intensity conditioning cord blood transplantation in adults with acute leukemia. Bone Marrow Transpl. 2017;52:1261–7.
Holtan SG, DeFor TE, Lazaryan A, Bejanyan N, Arora M, Brunstein CG, et al. Composite end point of graft-versus-host disease-free, relapse-free survival after allogeneic hematopoietic cell transplantation. Blood. 2015;125:1333–8.
Solomon SR, Sizemore C, Zhang X, Ridgeway M, Solh M, Morris LE, et al. Current graft-versus-host disease-free, relapse-free survival: a dynamic endpoint to better define efficacy after allogenic transplant. Biol Blood Marrow Transplant. 2017;23:1208–14.
Imus PH, Tsai HL, Luznik L, Fuchs EJ, Huff CA, Gladstone DE, et al. Haploidentical transplantation using posttransplant cyclophosphamide as GVHD prophylaxis in patients over age 70. Blood Adv. 2019;3:2608–16.
Arai I, Aoki K, Takeda J, Kondo T, Eto T, Ota S, et al. Clinical significance of high-dose cytarabine added to cyclophosphamide/total-body irradiation in bone marrow or peripheral blood stem cell transplantation for myeloid malignancy. J Hematol Oncol. 2015;8:102.
Acknowledgments
I would like to express my sincere gratitude to Dr. Hiroaki Mitsuya for providing me this study opportunity in his laboratory. The authors thank all of the physicians, nurses, pharmacists, and other staff at the hospital for taking care of patients. The authors also thank the cord blood bank in Japan.
Author information
Authors and Affiliations
Contributions
TK designed the study, took care of patients, analyzed extracted data, and contributed to writing the paper; MH took care of patients, reviewed the study design, and contributed to writing the paper. YI and MM reviewed the study design and contributed to writing the paper; HS, MY, AK, TH, NM, SY, YI, TS, and NH took care of patients and contributed to writing the paper, and YI contributed to data coordination.
Corresponding author
Ethics declarations
Conflict of Interest
Toshiro Kawakita reports personal fees from Otsuka Pharmaceutical, personal fees from Sanofi, outside the submitted work; Masao Matsuoka reports personal fees from Otsuka Pharmaceutical, personal fees from Sanofi, outside the submitted work. The other authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kawakita, T., Hirano, T., Inoue, Y. et al. Feasibility of an intensified myeloablative conditioning regimen consisting of busulfan, fludarabine, cytarabine, and total body irradiation before single cord blood transplantation in elderly patients. Int J Hematol 114, 85–93 (2021). https://doi.org/10.1007/s12185-021-03131-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03131-9