Abstract
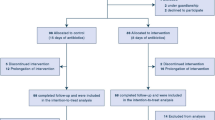
Although survival of children with hematological diseases and cancer has increased dramatically, febrile neutropenia (FN) is a frequently observed complication and is sometimes life-threatening in pediatric cancer patients. A prospective, randomized study was performed to clarify the usefulness of meropenem (MEPM) and piperacillin/tazobactam (PIPC/TAZ) for pediatric patients with FN. Ninety-nine patients with 394 episodes were randomly assigned to receive MEPM or PIPC/TAZ. MEPM was administered at 120 mg/kg/day as a 1-h drip infusion 3 times a day. On the other hand, PIPC/TAZ was administered at 360 mg/kg/day as a 1-h drip infusion 4 times a day. MEPM was effective in 69.5% of the 200 episodes, and PIPC/TAZ was effective in 77.2% of the 193 episodes. Compared with our previous study of MEPM 120 mg/kg/day as a 1-h drip infusion 3 times a day versus PIPC/TAZ 337.5 mg/kg/day as a 1-h drip infusion 3 times a day, the success rate of the MEPM group was not different. However, the success rate of the PIPC/TAZ group was higher than in the previous study (p = 0.001). In particular, the success rate in patients ≥ 15 years of age was improved in the PIPC/TAZ group of the present study compared with the previous study (p = 0.005).

Similar content being viewed by others
References
Hann I, Viscoli C, Paesmans M, Gaya H, Glauser M. A comparison of outcome from febrile neutropenic episodes in children compared with adults: results from four EORTC studies. International Antimicrobial Therapy Cooperative Group (IATCG) of the European Organization for Research and Treatment of Cancer (EORTC). Br J Haematol. 1997;99:580–8.
Viscoli C, Moroni C, Boni L, Bruzzi P, Comelli A, Dini G, et al. Ceftazidime plus amikacin versus ceftazidime plus vancomycin as empiric therapy in febrile neutropenic children with cancer. Rev Infect Dis. 1991;13:397–404.
Lehrnbecher T, Robinson P, Fisher B, Alexander S, Ammann RA, Beauchemin M, et al. Guideline for the management of fever and neutropenia in children with cancer and hematopoietic stem-cell transplantation recipients: 2017 update. J Clin Oncol. 2017;35:2082–94.
Freifeld AG, Bow EJ, Sepkowitz KA, Boeckh MJ, Ito JI, Mullen CA, Infectious Diseases Society of America, et al. Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the infectious diseases society of America. Clin Infect Dis. 2011;52:e56-93.
Sezgin G, Acipayam C, Ozkan A, Bayram I, Tanyeli A. Meropenem versus piperacillin-tazobactam as empiric therapy for febrile neutropenia in pediatric oncology patients. Asian Pac J Cancer Prev. 2014;15:4549–53.
Sato T, Kobayashi R, Yasuda K, Kaneda M, Iguchi A, Kobayashi K. A prospective, randomized study comparing cefozopran with piperacillin-tazobactam plus ceftazidime as empirical therapy for febrile neutropenia in children with hematological disorders. Pediatr Blood Cancer. 2008;51:774–7.
Kobayashi R, Sato T, Nakajima M, Kaneda M, Iguchi A. Piperacillin/tazobactam plus ceftazidime versus sulbactam/ampicillin plus aztreonam as empirical therapy for fever in severely neutropenic pediatric patients. J Pediatr Hematol Oncol. 2009;31:270–3.
Sarashina T, Kobayashi R, Yoshida M, Toriumi N, Suzuki D, Sano H, et al. A randomized trial of cefozopran versus cefepime as empirical antibiotic treatment of febrile neutropenia in pediatric cancer patients. Pediatr Blood Cancer. 2014;61:1992–5.
Sano H, Kobayashi R, Suzuki D, Kishimoo K, Yasuda K, Kobayashi K. Comparison of Piperacillin Tazobactam and cefepime monotherapy as an empiric therapy for febrile neutropenia in children with hematological and malignant disorders: a prospective randomized study. Pediatr Blood Cancer. 2015;62:356–8.
Sano H, Kobayashi R, Suzuki D, Hori D, Kishimoto K, Kobayashi K. A prospective randomized trial comparing piperacillin/tazobactam with meropenem as empiric antibiotic treatment of febrile neutropenic children and adolescents with hematologic and malignant disorders. Pediatr Blood Cancer. 2017;64:e26360.
Kanda Y. Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Viscoli C, Moroni C, Boni L, Bruzzi P, Comelli A, Dini G, et al. Rational dosing of antimicrobial agents: pharmacokinetic and pharmacodynamic strategies. Am J Health Syst Pharm. 2009;66:S23-30.
Solórzano-Santos F, Quezada-Herrera A, Fuentes-Pacheco Y, Rodríguez-Coello G, Aguirre-Morales CE, Izelo-Flores D, et al. Piperacillin/tazobactam in continuous infusion versus intermittent infusion in children with febrile neutropenia. Rev Invest Clin. 2019;71:283–90.
Benech N, Dumitrescu O, Conrad A, Balsat M, Paubelle E, Ducastelle-Lepretre S, Lyon HEMINF Study Group, et al. Parameters influencing the pharmacokinetics/pharmacodynamics of piperacillin/tazobactam in patients with febrile neutropenia and haematological malignancy: a prospective study. J Antimicrob Chemother. 2019;74:2676–80.
Sano H, Kobayashi R, Suzuki D, Kishimoto K, Hori D, Matsushima S, et al. Differential efficacy of empirical antibiotic therapy for febrile neutropenia in adolescent/young adult (AYA) and child patients. Int J Hematol. 2018;108:543–9.
Klastersky J, Paesmans M, Rubenstein EB, Boyer M, Elting L, Feld R, et al. The multinational association for supportive care in cancer risk index: a multinational scoring system for identifying low-risk febrile neutropenic cancer patients. J Clin Oncol. 2000;18:3038–51.
Acknowledgements
The authors would like to thank Ms. Yukiko Shiota for data management.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that there are no conflicts of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Kobayashi, R., Sano, H., Matsushima, S. et al. Meropenem versus piperacillin/tazobactam for febrile neutropenia in pediatric patients: efficacy of piperacillin/tazobactam as a 1-h drip infusion four times a day. Int J Hematol 113, 430–435 (2021). https://doi.org/10.1007/s12185-020-03031-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-020-03031-4