Abstract
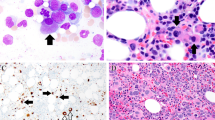
Few hematological complications have previously been reported in association with Cri du Chat syndrome (CdCS). A case of myelodysplastic syndromes (MDS) in a pediatric patient with CdCS is herein presented. A 17-year-old female with CdCS caused by ring chromosome 5 was admitted to the hospital for investigation of a 1-month history of anemia. Based on the morphological findings of bone marrow, the patient was diagnosed with refractory cytopenia with multilineage dysplasia. The risk group was classified as intermediate-1 in the International Prognostic Scoring System (IPSS), and low in the revised IPSS. Assessment by microarray comparative genomic hybridization (CGH) identified the breakpoints of ring chromosome 5 as 46,XX,r(5)(p14.3q35.3). This revealed that the 5q terminal deletion did not include the common deleted region of MDS with del(5q). Treatment with azacitidine was initiated to control disease progression and improve quality of life. At baseline, the patient had a mean transfusion requirement of 3 units/month, which decreased to 2 units/month after six cycles of azacitidine and to 1 unit/month after 10 cycles of azacitidine. Cytopenia observed in the presented case seemed irrelevant to ring chromosome 5 which is the causative cytogenetic abnormality of CdCS, and further analyses may be needed to clarify the pathogenesis.




Similar content being viewed by others
References
Locatelli F, Strahm B. How I treat myelodysplastic syndromes of childhood. Blood. 2018;131:1406–14.
Montalban-Bravo G, Garcia-Manero G. Myelodysplastic syndromes: 2018 update on diagnosis, risk-stratification and management. Am J Hematol. 2018;93:129–47.
Cseh AM, Niemeyer CM, Yoshimi A, et al. Therapy with low-dose azacitidine for MDS in children and young adults: a retrospective analysis of the EWOG-MDS study group. Br J Haematol. 2016;172:930–6.
Cerruti MP. Cri du Chat syndrome. Orphanet J Rare Dis. 2006;1:33.
Ai J, Visconte V, Rogers HJ, et al. Pure red cell aplasia in a patient with Cri-Du-Chat. Int Blood Res Rev. 2014;2:23–8.
Iwafuchi H, Ito M. Differences in the bone marrow histology between childhood myelodysplastic syndrome with multilineage dysplasia and refractory cytopenia of childhood without multilineage dysplasia. Histopathology. 2019;74:239–47.
Talati C, Sallman D, List A. Lenalidomide: myelodysplastic syndromes with del(5q) and beyond. Semin Hematol. 2017;54:159–66.
Sakaguchi H, Nishio N, Hama A, Kawashima N, et al., Japan Childhood Aplastic Anemia Study Group. Peripheral blood lymphocyte telomere length as a predictor of response to immunosuppressive therapy in childhood aplastic anemia. Haematologica. 2014;99:1312–6.
Colunga-Pedraza PR, Colunga-Pedraza JE, Garza-Ledezma MA, et al. Danazol as first-line therapy for myelodysplastic syndrome. Clin Lymphoma Myeloma Leuk. 2018;18:e109–e113113.
Haferlach T, Nagata Y, Grossmann V, et al. Landscape of genetic lesions in 944 patients with myelodysplastic syndromes. Leukemia. 2014;28:241–7.
Kunishima S, Okuno Y, Yoshida K, et al. ACTN1 mutations cause congenital macrothrombocytopenia. Am J Hum Genet. 2013;92:431–8.
Yamaguchi-Kabata Y, Nariai N, Kawai Y, et al. iJGVD: an integrative Japanese genome variation database based on whole-genome sequencing. Hum Genome Var. 2015;2:15050.
Ferner RE, Huson SM, Thomas N, et al. Guidelines for the diagnosis and management of individuals with neurofibromatosis 1. J Med Genet. 2007;44:81–8.
Invernizzi R, Quaglia F, Porta MG. Importance of classical morphology in the diagnosis of myelodysplastic syndrome. Mediterr J Hematol Infect Dis. 2015;7:e2015035.
Zhang A, Zheng C, Hou M, et al. Deletion of the telomerase reverse transcriptase gene and haploinsufficiency of telomere maintenance in Cri du chat syndrome. Am J Hum Genet. 2003;72:940–8.
Harrington L, Zhou W, McPhail T, et al. Human telomerase contains evolutionarily conserved catalytic and structural subunits. Genes Dev. 1997;11:3109–15.
Ropio J, Merlio JP, Soares P, Chevret E. Telomerase activation in hematological malignancies. Genes (Basel). 2016;7:61.
Hartwig FP, Collares T. Telomere dysfunction and tumor suppression responses in dyskeratosis congenita: balancing cancer and tissue renewal impairment. Ageing Res Rev. 2013;12:642–52.
Mainardi PC, Pastore G, Castronovo C, et al. The natural history of Cri du Chat Syndrome. A report from the Italian Register. Eur J Med Genet. 2006;49:363–83.
Perfumo C, Cerruti Mainardi P, Cali A, et al. The first three mosaic cri du chat syndrome patients with two rearranged cell lines. J Med Genet. 2000;37:967–72.
Acknowledgements
The authors thank Dr. Natsuko Suzui and Dr. Tatsuhiko Miyazaki of Gifu University Hospital for their helpful comments and staff of the Department of Pediatrics at Gifu University for their contributions. They thank Dr. Yusuke Okuno in Nagoya University for providing technical assistance of gene analysis. they thank Sarah Williams, PhD, and H. Nikki March, PhD, from Edanz Group (www.edanzediting.com) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest or funding to disclose.
Ethical approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional guidelines on human experimentation (Akifumi Nozawa, Michio Ozeki, Shiho Yasue, Saori Endo, Tomonori Kadowaki, Hidenori Ohnishi, Hideki Muramatsu, Asahito Hama, Yoshiyuki Takahashi, Seiji Kojima, and Toshiyuki Fukao) and with the Helsinki Declaration of 1975, as revised in 2008.
Informed consent
Written informed consent was obtained from the patient's parents for submission of this case report and any accompanying images.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Nozawa, A., Ozeki, M., Yasue, S. et al. Myelodysplastic syndromes in a pediatric patient with Cri du Chat syndrome with a ring chromosome 5. Int J Hematol 112, 728–733 (2020). https://doi.org/10.1007/s12185-020-02909-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-020-02909-7