Abstract
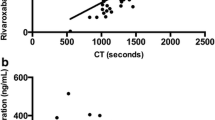
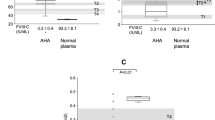
Patients with severe congenital protein (P)C deficiency require long-term anticoagulant management. Recombinant PC concentrates for prophylactic use are not available in Japan; prothrombin complex concentrates (PCC), containing factors (F)II, VII, IX, X, and PC (PPSB-HT®), have been used ‘off-label’ in a few patients. We investigated the combined use of prophylactic PCC and Warfarin (VKA; PT-INR 2.0–2.5) in a severely PC-deficient patient in whom VKA alone did not prevent recurrent purpura. Plasma VKA-dependent factor levels and global PC function (Thrombopath®) were assessed. Plasma activity levels of FII/FVII/FIX/FX post-infusion of PCC (6.3 unit/kg) increased 35/27/27/35 (initial level) to 59/60/38/83 IU/dl, respectively. FVII:C and FIX:C rapidly returned to baseline levels 12–24 h post-infusion, but FII:C and FX:C returned more slowly. PC antigen (< 5%) increased to ~ 15%, followed by return to baseline levels 24 h post-infusion. Global PC function was very low (%PiCi 24%), but improved post-PCC infusion. This potential was slightly detectable even at an undetectable PC level. At day 3, high levels of d-dimer and FDP were observed without thrombotic event, but these improved post-infusion. Although PCC restored VKA-dependent coagulation factors, PC contained in PCC significantly improved global anticoagulation, and was clinically beneficial in this severely deficient patient.




Similar content being viewed by others
References
Ohga S, Kang D, Kinjo T, Ochiai M, Doi T, Ishimura M, et al. Paediatric presentation and outcome of congenital protein C deficiency in Japan. Haemophilia. 2013;19:378–84.
Goldenberg NA, Manco-Johnson MJ. Protein C deficiency. Haemophilia. 2008;14:1214–21.
Ishiguro A, Ezinne CC, Michihata N, Nakadate H, Manabe A, Taki M, et al. Pediatric thromboembolism: a national survey in Japan. Int J Hematol. 2017;105:52–8.
Monagle P, Chan AKC, Goldenberg NA, Ichord RN, Journeycake JM, Nowak-Göttl U, et al. Antithrombotic therapy in neonates and children: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e737S–801S.
Oka T, Suzuki Y, Hiramoto A, Azuma H, Okuno A, Yoshioka H, et al. Severe heterozygous protein C deficiency with relapsing purpura fulminans from the newborn period. Rinsho Ketsueki. 1986;27:1971–6 (Article in Japanese).
Yasaka M, Sakata T, Minematsu K, Naritomi H. Correction of INR by prothrombin complex concentrate and vitamin K in patients with warfarin related hemorrhagic complication. Thromb Res. 2002;108:25–30.
Lisman T, Porte RJ. Rebalanced hemostasis in patients with liver disease: evidence and clinical consequences. Blood. 2010;116:878–85.
Toulon P, Smirnov M, Triscott M, Safa O, Biguzzi E, Bouziane K, et al. A new chromogenic assay (HemosIL ThromboPath) is sensitive to major prothrombotic risk factors affecting the protein C pathway. Results of a multicenter study. Thromb Res. 2009;124:137–43.
Shen L, Dahlbäck B. Factor V and protein S as synergistic cofactors to activated protein C in degradation of factor VIIIa. J Biol Chem. 1994;269:18735–8.
Dahlbäck B. Pro- and anticoagulant properties of factor V in pathogenesis of thrombosis and bleeding disorders. Int J Lab Hematol. 2016;38(Suppl 1):4–11.
Koenen RR, Tans G, van Oerle R, Hamulyak K, Roseng J, Hackeng TM. The APC-independent anticoagulant activity of protein S in plasma is decreased by elevated prothrombin levels due to the prothrombin G20210A mutation. Blood. 2003;102:1686–92.
Acknowledgements
We thank Dr. Satoshi Mushiake for providing patient’s lab data, and thank patient’s home doctor for keeping the patient’s condition stable for long term. This work was partly supported by a Grant-in-Aid for Scientific Research (KAKENHI) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) to KN (Grant No. 18K07885) and by a Japan Agency for Medical Research and Development (AMED) to KN (18ek0109210h0002).
Author information
Authors and Affiliations
Contributions
KO; performed experiments, interpreted the data, made the figures, and wrote the paper, KN; designed the research, interpreted the data, wrote the paper, edited the manuscript, and approved the final version to be published, KM, TN; performed experiments, NN, SO, performed the genetic analyses and interpreted the data, MS; supervised the studies.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ogiwara, K., Nogami, K., Mizumachi, K. et al. Hemostatic assessment of combined anticoagulant therapy using warfarin and prothrombin complex concentrates in a case of severe protein C deficiency. Int J Hematol 109, 650–656 (2019). https://doi.org/10.1007/s12185-019-02645-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-019-02645-7