Abstract
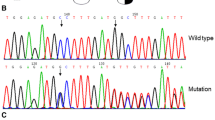
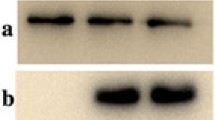
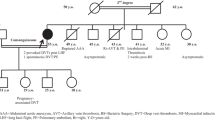
Dysfibrinogenemia is characterized by blood coagulation dysfunction induced by an abnormal molecular structure of fibrinogen. Here, we describe a new case. A 32-year-old female was suspected of having dysfibrinogenemia during routine laboratory screening, based on her decreased functional fibrinogen level, normal fibrinogen antigen level, and prolonged thrombin time. We extracted DNA and performed polymerase chain reaction and DNA sequencing to identify genetic mutation. Fibrin polymerization, the kinetics of the fibrinopeptide release, scanning electron microscopy, mass spectrometric analysis, fibrin cross-linking, sodium dodecyl sulfate polyacrylamide gel electrophoresis and western blot were conducted. DNA sequencing identified a heterozygous point mutation, Gly13Arg in Aα chain. Fibrin polymerization was markedly impaired (prolonged lag phase and decreased final turbidity). The rate and extent of fibrinopeptide A release from the patient were abnormal and reduced. The mass spectrometry analysis revealed the presence of mutant fibrinogen chains in the patient’s circulation. Electron micrographs revealed abnormal fibrin clots. Fibrin cross-linking was normal. Sodium dodecyl sulfate polyacrylamide gel electrophoresis and western blot showed no difference. We report a new case with a mutation in the fibrinopeptide A region, AαGly13Arg. These results indicated that the functional abnormalities were related to delayed and defective fibrinopeptide A cleavage and likely impaired thrombin binding.






Similar content being viewed by others
References
Asselta R, Duga S, Tenchini ML. The molecular basis of quantitative fibrinogen disorders. J Thromb Haemost. 2006;4:2115–29.
Kent WJ, Sugnet CW, Furey TS, Roskin KM, Pringle TH, Zahler AM, et al. The human genome browser at UCSC. Genome Res. 2002;12:996–1006.
Kreuz W, Meili E, Peter-Salonen K, Haertel S, Devay J, Krzensk U, et al. Efficacy and tolerability of a pasteurised human fibrinogen concentrate in patients with congenital fibrinogen deficiency. Transfus Apher Sci. 2005;32:247–53.
Mosesson MW. Fibrinogen structure and fibrin clot assembly. Semin Thromb Hemost. 1998;24:169–74.
Mosesson MW, Siebenlist KR, Meh DA. The structure and biological features of fibrinogen and fibrin. Ann N Y Acad Sci. 2001;936:11–30.
Walter S, Stabler S, Lefkowitz JB. Fibrinogen Denver: a dysfibrinogenemia associated with an abnormal reptilase time and significant bleeding. Haemophilia. 2006;12:393–7.
Cunningham MT, Brandt JT, Laposata M, Olson JD. Laboratory diagnosis of dysfibrinogenemia. Arch Pathol Lab Med. 2002;126:499–505.
Acharya SS, Dimichele DM. Rare inherited disorders of fibrinogen. Haemophilia. 2008;14:1151–8.
Meyer M, Kutscher G, Sturzebecher J, Riesener G, Lutze G. Fibrinogen Magdeburg I: a novel variant of human fibrinogen with an amino acid exchange in the fibrinopeptide A (Aalpha 9, Leu → Pro). Thromb Res. 2003;109:145–51.
Reber P, Furlan M, Beck EA, Barbui T. Fibrinogen Bergamo III and fibrinogen Torino: two further variants with hereditary molecular defects in fibrinopeptide A. Thromb Res. 1987;46:163–7.
Bogli C, Cofrancesco E, Cortellaro M, Della Volpe A, Hofer A, Furlan M, et al. Fibrinogen Milano. VI: a heterozygous dysfibrinogenemia (A alpha 16 Arg—His) with bleeding tendency. Eur J Haematol. 1990;45:26–30.
Imafuku Y, Tanaka K, Takahashi K, Ogawa K, Sanpei M, Yamada H, et al. Identification of a dysfibrinogenemia of gammaR275C (Fibrinogen Fukushima). Clin Chim Acta. 2002;325:151–6.
Kotlin R, Chytilova M, Suttnar J, Riedel T, Salaj P, Blatny J, et al. Fibrinogen Novy Jicin and Praha II: cases of hereditary Aalpha 16 Arg → Cys and Aalpha 16 Arg → His dysfibrinogenemia. Thromb Res. 2007;121:75–84.
Gralnick H, Givelber H, Shainoff J, Finlayson J. Fibrinogen Bethesda: a congenital dysfibrinogenemia with delayed fibrinopeptide release. J Clin Invest. 1971;50:1819.
Terasawa F, Okumura N, Higuchi Y, Ishikawa S, Tozuka M, Ishida F, et al. Fibrinogen Matsumoto III: a variant with gamma275 Arg → Cys (CGC → TGC)–comparison of fibrin polymerization properties with those of Matsumoto I (gamma364 Asp → His) and Matsumoto II (gamma308 Asn → Lys). Thromb Haemost. 1999;81:763–6.
Gharahdaghi F, Weinberg CR, Meagher DA, Imai BS, Mische SM. Mass spectrometric identification of proteins from silver-stained polyacrylamide gel: a method for the removal of silver ions to enhance sensitivity. Electrophoresis. 1999;20:601–5.
Tanaka H, Terasawa F, Ito T, Tokunaga S, Ishida F, Kitano K, et al. Fibrinogen Matsumoto V: a variant with Aalpha19 Arg → Gly (AGG → GGG). Comparison between fibrin polymerization stimulated by thrombin or reptilase and fibrin monomer polymerization. Thromb Haemost. 2001;85:108–13.
Bin Q, Liang F, Ou DY, Cui HR, Luo JM. Two symptomatic cases of dysfibrinogenemia in China: one with gamma-chain Arg275Cys mutation and another without detectable mutation in fibrinogen genes. Blood Coagul Fibrinolysis. 2015;26:564–71.
Finlayson JS, Morton RO. Gel electrophoresis for assessing fibrin crosslinking: a precaution. Clin Chim Acta. 1972;36:254–6.
Mosesson MW. Fibrinogen and fibrin polymerization: appraisal of the binding events that accompany fibrin generation and fibrin clot assembly. Blood Coagul Fibrinolysis. 1997;8:257–67.
Bogli C, Hofer A, Baudo F, Redaelli R, Furlan M. Fibrinogen Milano IV, another case of congenital dysfibrinogenemia with an abnormal fibrinopeptide A release (A alpha 16 Arg—His). Haemostasis. 1992;22:7–11.
Garbis S, Lubec G, Fountoulakis M. Limitations of current proteomics technologies. J Chromatogr A. 2005;1077:1–18.
Kotlin R, Suttnar J, Capova I, Hrachovinova I, Urbankova M, Dyr JE. Fibrinogen Sumperk II: dysfibrinogenemia in an individual with two coding mutations. Am J Hematol. 2012;87:555–7.
Kotlin R, Zichova K, Suttnar J, Reicheltova Z, Salaj P, Hrachovinova I, et al. Congenital dysfibrinogenemia Aalpha Gly13Glu associated with bleeding during pregnancy. Thromb Res. 2011;127:277–8.
Mathonnet F, Peltier JY, Detruit H, de Raucourt E, Alvarez JC, Mazmanian GM, et al. Fibrinogen Saint-Germain I: a case of the heterozygous Aalpha GLY 12 → VAL fibrinogen variant. Blood Coagul Fibrinolysis. 2002;13:149–53.
Denninger MH, Finlayson JS, Reamer LA, Parquet-Gernez A, Goudemand M, Menache D. Congenital dysfibrinogenemia: fibrinogen Lille. Thromb Res. 1978;13:453–66.
Pechik I, Yakovlev S, Mosesson MW, Gilliland GL, Medved L. Structural basis for sequential cleavage of fibrinopeptides upon fibrin assembly. Biochemistry. 2006;45:3588–97.
Martin PD, Robertson W, Turk D, Huber R, Bode W, Edwards BF. The structure of residues 7-16 of the A alpha-chain of human fibrinogen bound to bovine thrombin at 2.3-A resolution. J Biol Chem. 1992;267:7911–20.
Casini A, Neerman-Arbez M, Ariens RA, de Moerloose P. Dysfibrinogenemia: from molecular anomalies to clinical manifestations and management. J Thromb Haemost. 2015;13:909–19.
Patil R, Mukaddam A, Ghosh K, Shetty S. Management of pregnancy in dysfibrinogenemia cases: a dilemma. Blood Coagul Fibrinolysis. 2016.
Munoz J, Schering J, Lambing A, Neal S, Goyert G, Green PM, et al. The dilemma of inherited dysfibrinogenemia during pregnancy. Blood Coagul Fibrinolysis. 2012;23:775–7.
Santacroce R, Cappucci F, Pisanelli D, Perricone F, Papa ML, Santoro R, et al. Inherited abnormalities of fibrinogen: 10-year clinical experience of an Italian group. Blood Coagul Fibrinolysis. 2006;17:235–40.
Haverkate F, Samama M. Familial dysfibrinogenemia and thrombophilia. Report on a study of the SSC Subcommittee on Fibrinogen. Thromb Haemost. 1995;73:151–61.
Yamanaka Y, Takeuchi K, Sugimoto M, Sato A, Nakago S, Maruo T. Dysfibrinogenemia during pregnancy treated successfully with fibrinogen. Acta Obstet Gynecol Scand. 2003;82:972–3.
Miesbach W, Galanakis D, Scharrer I. Treatment of patients with dysfibrinogenemia and a history of abortions during pregnancy. Blood Coagul Fibrinolysis. 2009;20:366–70.
Yan J, Deng D, Luo M, Cheng P, Chi B, Yuan Y, et al. Dysfibrinogenemia in a patient undergoing artificial abortion after misdiagnosis and review of the literature. Clin Chim Acta. 2015;447:86–9.
Bolton-Maggs PH, Perry DJ, Chalmers EA, Parapia LA, Wilde JT, Williams MD, et al. The rare coagulation disorders—review with guidelines for management from the United Kingdom Haemophilia Centre Doctors’ Organisation. Haemophilia. 2004;10:593–628.
Acknowledgements
We would like to thank the Department of Hematology and Clinical Laboratory of the First Affiliated Hospital of Guangxi Medical University, China. This research was supported by Grants from the National Natural Science Foundation of China (81560342).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
J. Yan and M. Luo contributed equally to this work and are co-first authors.
About this article
Cite this article
Yan, J., Luo, M., Cheng, P. et al. A novel mutation in the fibrinogen Aα chain (Gly13Arg, fibrinogen Nanning) causes congenital dysfibrinogenemia associated with defective peptide A release. Int J Hematol 105, 506–514 (2017). https://doi.org/10.1007/s12185-016-2157-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-016-2157-1