Abstract
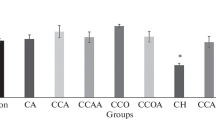
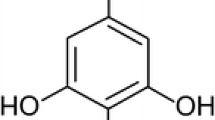
We investigated human blood erythrocytes under oxidative stress in vitro and established a correlation between composition and state of lipids and changes in erythrocytes structure under induced oxidative stress. These changes may serve as an indicator of not only the erythrocyte state but of systemic processes that occur at the level of the whole organism, including various pathologies as well. We found that a pyrimidine derivative xymedon used in the present study is an effective inhibitor of oxidative processes. Xymedon may be useful as an antioxidant for preserving the structural and functional characteristics of the erythrocytes in the treatment of organisms exposed to physical and toxic factors causing oxidative stress.






Similar content being viewed by others
References
Edwards CJ, Fuller J. Oxidative Stress in Erythrocytes. Comp Haematol Int. 1996;6:24–31.
Wagner BA, Buettner GR, Burns CP. Free radical-nediated lipid peroxidation in cells: oxidizability is a function of cell lipid bisallylic hydrogen content. Biochemistry. 1994;33:4449–53.
Izmaĭlov SG, Kochnev OS. The effect of xymedon on healing of linear wounds. Klin Khir. 1991;1:10–2.
Slabnov IuD, Cherepnev GV, Tsibul’kin AP, Karimova FG, Garaev RS. Effect of pyrimidine derivatives on the regulation of active transport of calcium in the immunocompetent cells. Eksp Klin Farmakol. 1997;60:44–6.
Slabnov IuD, Cherepnev GV, Tsibul’kin AP, Karimova FG, Garaev RS. The effect of pyrimidine derivatives on the sulfhydryl status of immunocompetent cells in vivo: the interrelationship with Ca2(+)-ATPase and antimutagenic activity. Eksp Klin Farmakol. 1998;61:40–3.
Cherepnev GV, Tereshchenko VIu, Malyshev KV, Semenov VV, Slabnov IuD, Garaev RS. The immunomodulator ximedon lowers the level of induced DNA damages in bone marrow and peripheral blood cells: the possibilities for immunogenetic correction. Eksp Klin Farmakol. 1999;62:31–5.
Cherepnev GV, Malyshev KV, Slabnov IuD, Semenov VV, Reznik VS. Potential role of the antimutagenic effect of xymedone in modification of immunoreactivity. Eksp Klin Farmakol. 2000;63:43–8.
Izmaĭlov SG, Izmaĭlov GA, Zaikonnikova IV, Reznik VS, Tereshchenko VIu, Gorbunov SM, et al. Clinical efficacy of using ximedon in surgical practice. Antibiot Khimioter. 2002;47:20–6.
Cherepnev GV, Kern F, Garaev RS. Xymedon decreases phosphatidylserine membrane expression induced by proapoptogenic deficit of serum growth factors in Jurkat T-cells. Eksp Klin Farmakol. 2002;65:40–3.
Hoвицкий BB, Pязaнцeвa HB, Cтeпoвaя EA, Фeдopoвa TC, Кpaвeц EБ, Ивaнoв BB и дp. Moлeкyляpныe нapyшeния мeмбpaны эpитpoцитoв пpи пaтoлoгии paзнoгo гeнeзa являютcя типoвoй peaкциeй opгaнизмa: кoнтypы пpoблeмы. Бюллeтeнь cибиpcкoй мeдицины. 2006;5:62–9.
Morimoto Y, Conroy SM, Ollberding NJ, Henning SM, Franke AA, Wilkens LR, et al. Erythrocyte membrane fatty acid composition, serum lipids, and non-Hodgkin’s lymphoma risk in a nested case-control study: the multiethnic cohort. Cancer Causes Control. 2012;23:1693–703.
Mihara M, Uchiyava M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal Biochem. 1978;86:271–8.
Bligh EA, Dyer W. Rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–7.
Broekhuyse RM. Phospholipid in tissues of the eye. Isolation, characterization and quantitative analysis by two-dimensional thin-layer chromatography of diacyl and vinyl-ether phospholipids. Biochim Biophys Acta. 1968;152:307–15.
Morrison WR, Smith ML. Preparation of fatty acid methyl esters and dimethylacetals from lipid with boron fluoride methanol. J Lipid Res. 1964;5:600–8.
Tychinskii VP, Kufal’ GE, Vyshenskaya TV, Perevedentseva EV, Nikandrov SL. Measurements of submicron structures with the airyscan laser phase microscope. Quantum Electron. 1997;27:735–9.
Tychinskii VP. Coherent phase microscopy of intracellular processes. Phys Usp. 2001;44:614.
Yusipovich A, Zagubizhenko M, Levin G, Platonova A, Parshina EY, Grygorzcyk R, et al. Laser interference microscopy of amphibian erythrocytes: impact of cell volume and refractive index. J Microsc. 2011;244:223–9.
Popescu G, Park Y, Choi W, Dasari RR, Feld MS, Badizadegan K. Imaging red blood cell dynamics by quantitative phase microscopy. Blood Cells Mol Dis. 2008;41:10–6.
Rappaz B, Barbul A, Emery Y, Korenstein I, Depeursinge C, Magistretti PJ, et al. Comparative study of human erythrocytes by digital holographic microscopy, confocal microscopy, and impedance volume analyzer. Cytometry. 2008;73A:895–903.
Brazhe NA, Brazhe AR, Pavlov AN, Erokhova NA, Yusipovich AI, Maksimov GV, et al. Unraveling cell processes: interference imaging interwoven with data analysis. J Biol Phys. 2006;32:191–208.
Brazhe AR, Brazhe NA, Maksimov GV, Ignatyev PS, Rubin AB, Mosekilde E et al. Phase-modulation laser interference microscopy: an advance in cell imaging and dynamics study. J Biomed Opt. 2008;13(3):034004.
Brandon D, Kaplan WD. Microstructural characterization of materials. 2nd ed. Chichester, England: Wiley; 2008. p. 536.
Yusipovich AI, Bryzgalova NYu, Parshina EYu, Lomakin AG, Rodnenkov OV, Levin GG, et al. Evaluation of erythrocyte shape and status by laser interference microscopy. Bull Exp Biol Med. 2008;145:382–5.
Yusipovich AI, Parshina EY, Brysgalova NYu, Brashe AR, Brashe NA, Lomakin AG, et al. Laser interference microscopy in erythrocyte study. J Appl Phys. 2009;105:102037.
Park Y, Diez-Silva M, Popescu G, Lykotrafitis G, Choi W, Feld MS, et al. Refractive index maps and membrane dynamics of human red blood cells parasitized by Plasmodium falciparum. PNAS. 2008;105:13730–5.
Shvets VI, Stepanov AE, Schmitt L, Spiess B, Schlewer G. Synthesis and complexation properties of inositol- phosphates. In: Reitz AB, editors. Inositol phosphates and derivetses: synthesis, biochemistry and therapeutic potential, Chapter 12, ACS Symposium Series, 463, Washington; 1991. p. 155–71.
Steinbrecher UP, Parthasarathy S, Leake DS, Witztum JL, Steinberg D. Modification of low density lipoprotein by endothelial cells involves lipid peroxidation and degradation of low density lipoprotein phospholipids. PNAS. 1984;81:3883–7.
Sheetz MP, Singer SJ. Equilibrium and kinetic effects of drugs on the shapes of human erythrocytes. J Cell Biol. 1976;70:247–51.
Cassera MB, Silber AM, Gennaro AM. Differential effects of cholesterol on acyl chain order in erythrocyte membranes as a function of depth from the surface: an electron paramagnetic resonance (EPR) spin label study. Biophys Chem. 2002;99:117–27.
Kiefer CR, Snyder LM. Oxidation and erythrocyte senescence. Curr Opin Hematol. 2000;7:113–6.
Шпepлинг ИA, Pязaнцeвa HB, Кyпpинa HП, Филиппoвa OH, Poгoв OA, Aкимoвa BB и дp. O cocтaвe мeмбpaннo-cвязaннoгo гeмoглoбинa пpи мeтгeмoглoбинeмияx. In: Snyder LM Coвpeмeнныe нayкoeмкиe тexнoлoгии. 2004;6:104–5.
Kumar R. Biochemical changes in erythrocyte membrane in type 2 diabetes mellitus. Indian J Med Sci. 2012;66:131–5.
Reddy KR, Reddy VD, Padmavathi P, Kavitha G, Saradamma B, Varadacharyulu NC. Gender differences in alcohol-induced oxidative stress and altered membrane properties in erythrocytes of rats. Indian J Biochem Biophys. 2013;50:32–9.
Rodnenkov OV, Luneva OG, Ulyanjva NA, Maksimov GV, Rubin AB, Orlov SN, et al. Erytrocy membrane fluidity and hemoglobin hemoporphyrin conformation: features revealed in patients with heart failure. Pathophysiology. 2005;11:209–13.
Acknowledgments
This work was supported in part by grants from the Ogarev Mordovia State University.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Revin, V.V., Filatova, S.M., Syusin, I.V. et al. Study of correlation between state and composition of lipid phase and change in erythrocytes structure under induction of oxidative processes. Int J Hematol 101, 487–496 (2015). https://doi.org/10.1007/s12185-015-1758-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-015-1758-4