Abstract
Computer planning software and patient-specific instrumentation have been investigated in multiple subspecialties of orthopedics with mixed results. Shoulder arthroplasty has evolved over the last decade with improvements in implant design and surgical instrumentation. Despite these advances, glenoid positioning in shoulder arthroplasty continues to be a difficult problem. Recent advances in three-dimensional imaging techniques and the use of computer planning software may potentially address some of the common difficulties encountered by surgeons. The addition of patient-specific instrumentation and guides provide an option for patients with significant glenoid deformity that may allow improved accuracy of glenoid component implantation compared to using standard instrumentation. Studies have reported improved positioning of the glenoid component in both anatomic and reverse total shoulder arthroplasty with patient-specific instrumentation and guides. More research is needed to determine whether these improvements lead to better patient-reported outcomes or implant survival. In addition, further studies will be needed to address whether this technology is cost effective for large-scale implementation in the orthopedic community.

Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Singh JA, Sperling J, Buchbinder R, McMaken K. Surgery for shoulder osteoarthritis. Cochrane Database Syst Rev. 2010;10, CD008089.
Wiater JM, Fabing MH. Shoulder arthroplasty: prosthetic options and indications. J Am Acad Orthop Surg. 2009;17:415–25.
Samitier G, Alentorn-Geli E, Torrens C, Wright TW. Reverse shoulder arthroplasty. Part 1: systematic review of clinical and functional outcomes. Int J Should Surg. 2015;9:24–31.
Boileau P, Watkinson DJ, Hatzidakis AM, Balg F. Grammont reverse prosthesis: design, rationale, and biomechanics. J Should Elb Surg. 2005;14:S147–61.
Sassoon A, Nam D, Nunley R, Barrack R. Systematic review of patient-specific instrumentation in total knee arthroplasty: new but not improved. Clin Orthop Relat Res. 2015;473:151–8.
Venkatesan M, Mahadevan D, Ashford RU. Computer-assisted navigation in knee arthroplasty: a critical appraisal. J Knee Surg. 2013;26:357–61.
Atesok K, Schemitsch EH. Computer-assisted trauma surgery. J Am Acad Orthop Surg. 2010;18:247–58.
Farron A, Terrier A, Büchler P. Risks of loosening of a prosthetic glenoid implanted in retroversion. J Should Elb Surg. 2006;15:521–6.
Ho JC, Sabesan VJ, Iannotti JP. Glenoid component retroversion is associated with osteolysis. J Bone Joint Surg Am. 2013;95. Sixty-six total shoulder arthroplasties were evaluated with radiographs at an average of 3.8 years postoperative to determine factors affecting osteolysis around the central peg of an all-polyethylene press fit pegged glenoid component. Thirty percent of implants had osteolysis around the center peg at follow-up and excessive postoperative retroversion >15° was associated with an increased odds of osteolysis, whereas preoperative retroversion was not associated with no change in the odds of osteolysis.
Shapiro TA, McGarry MH, Gupta R, Lee YS, Lee TQ. Biomechanical effects of glenoid retroversion in total shoulder arthroplasty. J Should Elb Surg. 2007;16:S90–5.
Nowak DD, Bahu MJ, Gardner TR, Dyrszka MD, Levine WN, Bigliani LU, et al. Simulation of surgical glenoid resurfacing using three-dimensional computed tomography of the arthritic glenohumeral joint: the amount of glenoid retroversion that can be corrected. J Should Elb Surg. 2009;18:680–8.
Gillespie R, Lyons R, Lazarus M. Eccentric reaming in total shoulder arthroplasty: a cadaveric study. Orthopedics. 2009;32:21.
Clavert P, Millett PJ, Warner JJP. Glenoid resurfacing: what are the limits to asymmetric reaming for posterior erosion? J Should Elb Surg. 2007;16:843–8.
Walch G, Young AA, Boileau P, Loew M, Gazielly D, Molé D. Patterns of loosening of polyethylene keeled glenoid components after shoulder arthroplasty for primary osteoarthritis: results of a multicenter study with more than five years of follow-up. J Bone Joint Surg Am. 2012;94:145–50. Five hundred eighteen total shoulder arthroplasties with a cemented, keeled all-polyethylene glenoid component were evaluated retrospectively with radiographs at a mean of 122 months postoperative for humeral head subluxation, periprosthetic radiolucent lines and shifting of the glenoid component. Definite radiographic evidence of glenoid loosening was seen in 32 % of shoulders with subsidence and posterior tilting of the glenoid component associated with the use of reaming past subchondral bone to optimize glenoid component seating and position. The authors recommend preserving subchondral bone to maintain long-term longevity of the glenoid component.
Sabesan V, Callanan M, Ho J, Iannotti JP. Clinical and radiographic outcomes of total shoulder arthroplasty with bone graft for osteoarthritis with severe glenoid bone loss. J Bone Joint Surg Am. 2013;95:1290–6.
Hill JM, Norris TR. Long-term results of total shoulder arthroplasty following bone-grafting of the glenoid. J Bone Joint Surg Am. 2001;83-A:877–83.
Klika BJ, Wooten CW, Sperling JW, Steinmann SP, Schleck CD, Harmsen WS, et al. Structural bone grafting for glenoid deficiency in primary total shoulder arthroplasty. J Should Elb Surg. 2014;23:1066–72.
Mizuno N, Denard PJ, Raiss P, Walch G. Reverse total shoulder arthroplasty for primary glenohumeral osteoarthritis in patients with a biconcave glenoid. J Bone Joint Surg Am. 2013;95:1297–304.
Rice RS, Sperling JW, Miletti J, Schleck C, Cofield RH. Augmented glenoid component for bone deficiency in shoulder arthroplasty. Clin Orthop Relat Res. 2008;466:579–83.
Cil A, Sperling JW, Cofield RH. Nonstandard glenoid components for bone deficiencies in shoulder arthroplasty. J Should Elb Surg. 2014;23:e149–57.
Wang T, Abrams GD, Behn AW, Lindsey D, Giori N, Cheung EV. Posterior glenoid wear in total shoulder arthroplasty: eccentric anterior reaming is superior to posterior augment. Clin Orthop Relat Res. 2015;473:3928–36.
Sabesan V, Callanan M, Sharma V, Iannotti JP. Correction of acquired glenoid bone loss in osteoarthritis with a standard versus an augmented glenoid component. J Should Elb Surg. 2014;23:964–73.
Sabesan V, Callanan M, Sharma V. Guidelines for the selection of optimal glenoid augment size for moderate to severe glenohumeral osteoarthritis. J Should Elb Surg. 2014;23:974–81.
Hermida JC, Flores-Hernandez C, Hoenecke HR, D’Lima DD. Augmented wedge-shaped glenoid component for the correction of glenoid retroversion: a finite element analysis. J Should Elb Surg. 2014;23:347–54.
Knowles NK, Ferreira LM, Athwal GS. Augmented glenoid component designs for type B2 erosions: a computational comparison by volume of bone removal and quality of remaining bone. J Should Elb Surg. 2015;24:1218–26. Three augmented component designs—full-wedge, posterior-wedge and posterior-step—were studied by virtual implantation in a cohort of 16 patients with B2 glenoids with version correction to 0° and 10°. Posterior wedge implants resulted in substantially less glenoid bone removal with the remaining supporting bone being better quality than a full wedge or posterior step component.
Kersten AD, Flores-Hernandez C, Hoenecke HR, D’Lima DD. Posterior augmented glenoid designs preserve more bone in biconcave glenoids. J Should Elb Surg. 2015;24:1135–41.
Neyton L, Boileau P, Nové-Josserand L, Edwards TB, Walch G. Glenoid bone grafting with a reverse design prosthesis. J Should Elb Surg. 2007;16:S71–8.
Melis B, Bonnevialle N, Neyton L, Lévigne C, Favard L, Walch G, et al. Glenoid loosening and failure in anatomical total shoulder arthroplasty: is revision with a reverse shoulder arthroplasty a reliable option? J Should Elb Surg. 2012;21:342–9.
Werner BS, Böhm D, Abdelkawi A, Gohlke F. Glenoid bone grafting in reverse shoulder arthroplasty for long-standing anterior shoulder dislocation. J Should Elb Surg. 2014;23:1655–61.
Wagner E, Houdek MT, Griffith T, Elhassan BT, Sanchez-Sotelo J, Sperling JW, et al. Glenoid bone-grafting in revision to a reverse total shoulder arthroplasty. J Bone Joint Surg Am. 2015;97:1653–60.
Gonzalez J-F, Alami GB, Baque F, Walch G, Boileau P. Complications of unconstrained shoulder prostheses. J Should Elb Surg. 2011;20:666–82.
Papadonikolakis A, Neradilek MB, Matsen FA. Failure of the glenoid component in anatomic total shoulder arthroplasty. J Bone Joint Surg Am. 2013;95:2205.
Deutsch A, Abboud JA, Kelly J, Mody M, Norris T, Ramsey ML, et al. Clinical results of revision shoulder arthroplasty for glenoid component loosening. J Should Elb Surg. 2007;16:706–16.
Cheung E, Willis M, Walker M, Clark R, Frankle MA. Complications in reverse total shoulder arthroplasty. J Am Acad Orthop Surg. 2011;19:439–49.
Lévigne C, Boileau P, Favard L, Garaud P, Molé D, Sirveaux F, et al. Scapular notching in reverse shoulder arthroplasty. J Should Elb Surg. 2008;17:925–35.
Klein SM, Dunning P, Mulieri P, Pupello D, Downes K, Frankle MA. Effects of acquired glenoid bone defects on surgical technique and clinical outcomes in reverse shoulder arthroplasty. J Bone Joint Surg Am. 2010;92:1144–54.
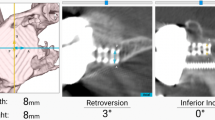
Scalise JJ, Codsi MJ, Bryan J, Brems JJ, Iannotti JP. The influence of three-dimensional computed tomography images of the shoulder in preoperative planning for total shoulder arthroplasty. J Bone Joint Surg Am. 2008;90:2438–45.
Ganapathi A, McCarron JA, Chen X, Iannotti JP. Predicting normal glenoid version from the pathologic scapula: a comparison of 4 methods in 2- and 3-dimensional models. J Should Elb Surg. 2011;20:234–44.
Scalise JJ, Codsi MJ, Bryan J, Iannotti JP. The three-dimensional glenoid vault model can estimate normal glenoid version in osteoarthritis. J Should Elb Surg. 2008;17:487–91.
Iannotti JP, Weiner S, Rodriguez E, Subhas N, Patterson TE, Jun BJ, et al. Three-dimensional imaging and templating improve glenoid implant positioning. J Bone Joint Surg Am. 2015;97:651–8. Forty-six patients with 3D CT preoperative templating were compared to 17 patients with 2D imaging and templating with regards to achieving desired implant position. The use of 3D templating significantly improved achievement of the desired implant position within 5° of inclination or 10° of version when compared to 2D imaging.
Iannotti J, Baker J, Rodriguez E, Brems J, Ricchetti E, Mesiha M, et al. Three-dimensional preoperative planning software and a novel information transfer technology improve glenoid component positioning. J Bone Joint Surg Am. 2014;96:e71–1. Nine bone models from patients with glenohumeral arthritis were implanted with a glenoid component in vitro using standard instrumentation alone, standard instrumentation and 3D planning and a patient specific instrument and 3D planning. Postoperative 3D CT scans were made to register to the preoperative plan. The use of 3D planning with standard instruments improved accuracy of guide pin placement for the glenoid compared to no planning with standard instruments. The use of 3D planning with patient specific instruments further improved guide pin placement in comparison to 3D planning with standard instruments.
Walch G, Vezeridis PS, Boileau P, Deransart P, Chaoui J. Three-dimensional planning and use of patient-specific guides improve glenoid component position: an in vitro study. J Should Elb Surg. 2015;24:302–9. Eighteen cadaveric scapulae were imaged with CT and glenoid implants were placed virtually and patient specific guides were created to direct the central guide pin to position the glenoid. Glenoid component placement was performed and then postoperative CT scans were performed. Accuracy of glenoid component placement on final CT compared to preoperative plan was within 2° of planned inclination and version.
Stübig T, Petri M, Zeckey C, Hawi N, Krettek C, Citak M, et al. 3D navigated implantation of the glenoid component in reversed shoulder arthroplasty. Feasibility and results in an anatomic study. Int J Med Robot Comput Assist Surg. 2013;9:480–5.
Venne G, Rasquinha B, Pichora D, Ellis RE, Bicknell R. Comparing conventional and computer-assisted surgery baseplate and screw placement in reverse shoulder arthroplasty. J Should Elb Surg. 2014;1–8.
Denard PJ, Walch G. Current concepts in the surgical management of primary glenohumeral arthritis with a biconcave glenoid. J Should Elb Surg. 2013;22:1589–98.
Budge MD, Lewis GS, Schaefer E, Coquia S, Flemming DJ, Armstrong AD. Comparison of standard two-dimensional and three-dimensional corrected glenoid version measurements. J Should Elb Surg. 2011;20:577–83.
Terrier A, Ston J, Farron A. Importance of a three-dimensional measure of humeral head subluxation in osteoarthritic shoulders. J Should Elb Surg. 2015;24:295–301.
Jacxsens M, Van Tongel A, Willemot LB, Mueller AM, Valderrabano V, De Wilde L. Accuracy of the glenohumeral subluxation index in nonpathologic shoulders. J Should Elb Surg. 2015;24:541–6.
Hendel MD, Bryan JA, Barsoum WK, Rodriguez EJ, Brems JJ, Evans PJ, et al. Comparison of patient-specific instruments with standard surgical instruments in determining glenoid component position. J Bone Joint Surg Am. 2012;94:2167. Thirty-one patients were randomized to glenoid component placement with 3D CT planning software and patient specific instrumentation or conventional CT scanning and surgical instruments. Postoperative CT scans were used to define and compare the actual implant location with the preoperative plan. The patient specific instrumentation decreased the average deviation of implant position for inclination and medial-lateral offset. The average deviation in version from the preoperative plan was 7° in the standard surgical group and 4° in the patient specific instrumentation group. In patients with preoperative retroversion over 16°, the average deviation in version from the preoperative plan was 10° in the standard instrument group and 1.2° in the patient specific instrument group.
Levy JC, Everding NG, Frankle MA, Keppler LJ. Accuracy of patient-specific guided glenoid baseplate positioning for reverse shoulder arthroplasty. J Should Elb Surg. 2014;23:1563–7. Fourteen cadaveric shoulders were CT scanned and then a patient specific guide was created based upon a virtual preoperative 3D plan. Using the guide was then used and a post-procedure CT scan of the scapula was performed to compare the preoperative plan trajectory with the final position of the drill pathway. The drill pathway was within 1.2° of inclination and 2.6° of version of the planned pathway.
Verborgt O, Vanhees M, Heylen S, Hardy P, Declercq G, Bicknell R. Computer navigation and patient-specific instrumentation in shoulder arthroplasty. Sports Med Arthrosc. 2014;22:e42–9.
Throckmorton TW, Gulotta LV, Bonnarens FO, Wright SA, Hartzell JL, Rozzi WB, et al. Patient-specific targeting guides compared with traditional instrumentation for glenoid component placement in shoulder arthroplasty: a multi-surgeon study in 70 arthritic cadaver specimens. J Should Elb Surg. 2015;24:965–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
James D. Wylie declares that he has no conflict of interest.
Robert Z. Tashjian currently receives royalties from IMASCAP, a planning software company, as well as Shoulder Innovations, a company designing a total shoulder arthroplasty system.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Shoulder Arthroplasty
Rights and permissions
About this article
Cite this article
Wylie, J.D., Tashjian, R.Z. Planning software and patient-specific instruments in shoulder arthroplasty. Curr Rev Musculoskelet Med 9, 1–9 (2016). https://doi.org/10.1007/s12178-016-9312-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12178-016-9312-4