Abstract
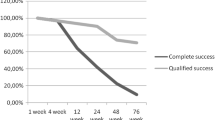
This study aims to evaluate the efficacy and safety of post-trabeculectomy topical bevacizumab, in preventing bleb failure. A total of 11 females and 12 males were enrolled for the study. After a complete ophthalmic evaluation, the patients underwent trabeculectomy. Full ophthalmic examination was done the next day, which included visual acuity, intraocular pressure (IOP), and bleb morphology. Next, two drops of bevacizumab [0.25 mg (0.01 ml)] were instilled over the wound area with a tuberculin (1 ml) syringe. Pressure was applied at the medial canthus to prevent it from going into the lacrimal passage. Similar postoperative procedures were repeated at 1, 2, 3, and 4 weeks. So in all, five instillations of bevacizumab were completed. Later, the patients were followed at 1-month interval for 6 months. Target IOP was achieved in all eyes and was maintained during the study period of 6 months without any anti-glaucoma medication. Average IOP before trabeculectomy was 27.27 ± 8.92 mmHg, which decreased postoperatively to 8.77 ± 0.97 mmHg at 3 months and 9.27 ± 1.16 mmHg at 6 months. This drop in IOP was statistically significant on both occasions (p = 0.00). A nice low filtering bleb was formed after the surgery, and it remained the same during the study period. The other parameters like cup-to-disc ratio and visual field did not change during the 6 months of study as expected because glaucoma is a slow progressive disease. Modulating the wound at multiple sites by bevacizumab (inflammation, proliferative, and remodeling stages) can lead to success of trabeculectomy.



Similar content being viewed by others
References
Addicks EM, Quigley HA, Green WR, Robin AL. Histologic characteristics of filtering blebs in glaucomatous eyes. Arch Ophthalmol. 1983;101:795–8.
Mills KB. Trabeculectomy: a retrospective long-term follow-up of 444 cases. Br J Ophthalmol. 1981;65:790–5.
Ridgway AE, Rubinstein K, Smith VH. Trabeculectomy. A study of 86 cases. Br J Ophthalmol. 1972;56:511–6.
Ridgway AE. Trabeculectomy. A follow-up study. Br J Ophthalmol. 1974;58:680–6.
Schwartz AL, Anderson DR. Trabecular surgery. Arch Ophthalmol. 1974;92:134–8.
Schwartz PL, Ackerman J, Beards J, Wesseley Z, Goodstein S, Ballen PH. Further experience with trabeculectomy. Ann Ophthalmol. 1976;8:207–17.
Kahook MY, Camejo L, Noecker RJ. Trabeculectomy with intraoperative retrobulbar triamcinolone acetonide. Clin Ophthalmol. 2009;3:29–31.
Lu D, Tai MC, Chiang C. Subconjunctival retention of perfluoropropane gas increases the success of rate of trabeculectomy. Asian J Ophthalmol. 1999;1:8–9.
Lu DW, Tai MC, Chiang CH. Subconjunctival retention of perfluoropropane (C3F8) gas increased the success rates of trabeculectomy. J Ocul Pharmacol Ther. 1997;13:235–42.
Skuta GL. Antifibrotic agents in glaucoma filtering surgery. Int Ophthalmol Clin. 1993;33:165–82.
Rockwood EJ, Parrish 2nd RK, Heuer DK, et al. Glaucoma filtering surgery with 5-fluorouracil. Ophthalmology. 1987;94:1071–8.
Akarsu C, Onol M, Hasanreisoglu B. Postoperative 5-fluorouracil versus intraoperative mitomycin C in high-risk glaucoma filtering surgery: extended follow up. Clin Exp Ophthalmol. 2003;31:199–205.
Membrey WL, Poinoosawmy DP, Bunce C, Hitchings RA. Glaucoma surgery with or without adjunctive antiproliferatives in normal tension glaucoma: 1 intraocular pressure control and complications. Br J Ophthalmol. 2000;84:586–90.
Yoon PS, Singh K. Update on antifibrotic use in glaucoma surgery, including use in trabeculectomy and glaucoma drainage implants and combined cataract and glaucoma surgery. Curr Opin Ophthalmol. 2004;15:141–6.
McGuigan LJ, Cook DJ, Yablonski ME. Dexamethasone, d-penicillamine, and glaucoma filtering surgery in rabbits. Invest Ophthalmol Vis Sci. 1986;27:1755–7.
Mc Guigan LJB, Mason RP, Sanchez R, Quigley HA. d-Penicillamine and beta-aminopropionitrile effects on experimental filtering surgery. Invest Ophthalmol Vis Sci. 1987;28:1625–9.
Moorhead LC. Inhibition of collagen cross-linking: a new approach to ocular scarring. Curr Eye Res. 1981;1:77–83.
Moorhead LC, Smith J, Stewart R, Kimbrough R. Effects of beta-aminopropionitrile after glaucoma filtration surgery; pilot human study. Ann Ophthalmol. 1987;19:223–5.
Cameron ME. Beta irradiation as an adjunct to surgery in refractory glaucoma. Trans Aust Coll Ophthalmol. 1970;2:53–60.
Skuta GL, Parrish RK. Wound healing in glaucoma filtering surgery. Surv Ophthalmol. 1987;32:149–70.
Jordan JF, Diestelhorst M, Grisanti S, Krieglstein GK. Photodynamic modulation of wound healing in glaucoma filtration surgery. Br J Ophthalmol. 2003;87:870–5.
Lu DW, Chang CJ, Chiang CH, Yeh MK, Chou PI. Wound modulation after trabeculectomy by different formulations of antimetabolites in rabbits. J Ocul Pharmacol Ther. 2000;16:529–38.
Demir T, Turgut B, Celiker U, et al. Effects of octreotide acetate and amniotic membrane on wound healing in experimental glaucoma surgery. Doc Ophthalmol. 2003;107:87–92.
Vandewalle E, Spielberg L, Zeyen T, Stalmans I. Can preoperative bevacizumab improve trabeculectomy outcome? “Avastin Trab Study”. Bull Soc Belge Ophthalmol. 2010;314:51–2.
Choi JY, Choi J, Y-De K. Subconjunctival bevacizumab as an adjunct to trabeculectomy in eyes with refractory glaucoma: a case series. Korean J Ophthalmol. 2010;24:47–52.
Li Z, Van Bergen T, Van de Veire S, et al. Inhibition of vascular endothelial growth factor reduces scar formation after glaucoma filtration surgery. Invest Ophthalmol Vis Sci. 2009;50:5217–25.
Cantor LB, Mantravadi A, WuDunn D, Swamynathan K, Cortes A. Morphologic classification of filtering blebs after glaucoma filtration surgery: the Indiana bleb appearance grading scale. Journal of Glaucoma. 2003;12:266–71.
World Health Organization and International Agency for the Prevention of Blindness. The state of the world’s sight. VISION 2020: the Right to Sight: 1999–2005. Geneva: WHO; 2005.
McEvan WK. Application of Poiseuille’s law to aqueous outflow. Arch Ophthalmol. 1958;60:290–4.
Hitchings RA, Grierson I. Clinico pathological correlation in eyes with failed fistulizing surgery. Trans Ophthalmol Soc UK. 1983;103:84–8.
Lama PJ, Fechtner RD. Antifibrotics and wound healing in glaucoma surgery. Surv Ophthalmol. 2003;48:314–46.
Esson DW, Popp MP, Liu L, Schultz GS, Sherwood MB. Microarray analysis of the failure of filtering “blebs” in a rat model of glaucoma filtering surgery. Invest Ophthalmol Vis Sci. 2004;45:4450–62.
Chang MR, Cheng Q, Lee DA. Basic science and clinical aspects of wound healing in glaucoma filtering surgery. J Ocul Pharmacol Ther. 1998;14:75–95.
Grierson I, Joseph J, Miller M, Day JE. Wound repair: the fibroblast and the inhibition of scar formation. Eye. 1988;2(Pt 2):135–48.
Tahery MM, Lee DA. Review: pharmacologic control of wound healing in glaucoma filtration surgery. J Ocul Pharmacol. 1989;5:155–79.
Cordeiro MF, Schultz GS, Ali RR, Bhattacharya SS, Khaw PT. Molecular therapy in ocular wound healing. Br J Ophthalmol. 1999;83:1219–24.
Siriwardena D, Kotecha A, Minassian D, Dart JKG, Khaw PT. Anterior chamber flare after trabeculectomy and after phacoemulsification. Br J Ophthalmol. 2000;84:1056–7.
Gordon CR, Rojavin Y, Patel M, et al. A review on bevacizumab and surgical wound healing: an important warning to all surgeons. Ann Plast Surg. 2009;62(6):707–9.
Wilgus TA, Ferreira AM, Oberyszyn TM, Bergdall VK, Dipietro LA. Regulation of scar formation by vascular endothelial growth factor. Lab Invest. 2008;88:579–90.
Methew R, Barton K. Anti vascular endothelial growth factor therapy in glaucoma filtration surgery. Am J Ophthalmol. 2011;152:10–5.
O’Neill EC, Qin Q, Van Bergen NJ, et al. Antifibrotic activity of bevacizumab on human Tenon’s fibroblast in vitro. Invest Ophthalmol Vis Sci. 2010;51:6524–32.
Nomoto H, Shiraga F, Kuno N, et al. Pharmacokinetics of bevacizumab after topical, subconjunctival, and intravitreal administration in rabbits. Invest Ophthalmol Vis Sci. 2009;50:4807–13.
Dastjerdi MH, Al-Arfaz KM, Nallasamy N, et al. Topical bevacizumab in the treatment of corneal neovascularization: results of a prospective, open-label, non-comparative study. Arch Ophthalmol. 2009;127:381–9.
Bock F, Onderka J, Rummelt C, et al. Safety profile of topical VEGF neutralization at the cornea. Invest Ophthalmol Vis Sci. 2009;50:2095–102.
Kim SW, Ha BJ, Kim EK, Tchah H, Kim TI. The effect of topical bevacizumab on corneal neovascularization. Ophthalmology. 2008;115:33–8.
Koenig Y, Bock F, Horn F, Kruse F, Straub K, Cursiefen C. Short- and long-term safety profile and efficacy of topical bevacizumab (Avastin) eye drops against corneal neovascularization. Graefes Arch Clin Exp Ophthalmol. 2009;247:1375–82.
Hosseini H, Khalili MR. Therapeutic potential of bevacizumab (Avastin) in herpetic stromal keratitis (HSK). Med Hypotheses. 2007;69:568–70.
Heindl LM, Cursiefen C. Pterygium. Etiology, clinical aspects and novel adjuvant therapies. Ophthalmologe. 2010;107(517–20):522–4.
Wu PC, Kuo HK, Tai MH, Shin SJ. Topical bevacizumab eyedrops for limbal-conjunctival neovascularization in impending recurrent pterygium. Cornea. 2009;28:103–4.
Uy HS, Chan PS, Ang RE. Topical bevacizumab and ocular surface neovascularization in patients with Stevens-Johnson syndrome. Cornea. 2008;27:70–3.
Broadway DC, Grierson I, O’Brien C, Hitchings RA. Adverse effects of topical antiglaucoma medication. I. The conjunctival cell profile. Arch Ophthalmol. 1994;112:1437–45.
Liesegang TJ. Conjunctival changes associated with glaucoma therapy: implications for the external disease consultant and the treatment of glaucoma. Cornea. 1998;17:574–83.
Cevenkel B, Kopitar AN, Ihan A. Mediat Inflamm: Inflammatory molecule in aqueous humour and on ocular surface and glaucoma surgery outcome; 2010. doi:10.1155/2010/939602.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bhasker, S.K., Saxena, S. & Singh, K. Post-trabeculectomy topical bevacizumab preventing bleb failure: a preliminary study. j ocul biol dis inform 5, 89–95 (2012). https://doi.org/10.1007/s12177-013-9108-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12177-013-9108-3