Abstract
Introduction
There is considerable interest in identifying potential drivers for human atrial fibrillation (AF), in order to improve therapy. Ablation via pulmonary vein isolation (PVI) is broadly used, yet is insufficient in many patients yet its outcomes are unimproved by adding extensive ablation of lines or complex electrogram sites, particularly in patients with persistent AF.
Novelty
Rotational and focal sources for AF represent novel mechanistic and therapeutic targets, often remote from the PVs and proven to drive AF in many studies. This chapter discusses this issue.
Aspects of Clinical Relevance
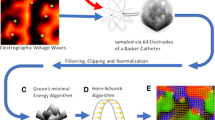
AF sources can now routinely be identified clinically by many methods, yet discrepant results have been reported. AF drivers identified by Focal Impulse and Rotor Mapping (FIRM), the most widely applied method, are also seen in simultaneous optical maps of human atria and have now been detected by other mapping methods applied to the exact same signals. In proof-of-concept studies, ablation of drivers can terminate persistent AF and, in over a thousand patients reported thus far, yield favorable long-term outcomes versus PVI alone. Nevertheless, some centers show disappointing results. This review focuses on discrepant results, which may reflect challenging patients, operator unfamiliarity with basket catheter use, or the technical ablation of drivers, amongst other factors. We discuss challenges, potential solutions, and future directions for map-guided AF driver ablation including basket-data collection, interpreting AF maps, ablation guidance, and extent.
Conclusions
Mapping and ablation of AF drivers is a rapidly growing field which, with continued scientific discovery and procedural advances, offers a strong mechanistic foundation to improve patient-tailored ablation for complex arrhythmias.





Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
January CT, et al. AHA/ACC/HRS guideline for the Management of Patients with Atrial Fibrillation: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014; doi:10.1016/j.jacc.2014.03.021.
Verma A, et al. Approaches to catheter ablation for persistent atrial fibrillation. N Engl J Med. 2015;372:1812–22. doi:10.1056/NEJMoa1408288.
Calkins H. Demonstrating the value of contact force sensing: more difficult than meets the eye. Circulation. 2015;132:901–3. doi:10.1161/CIRCULATIONAHA.115.018354.
Kuck KH, et al. Cryoballoon or radiofrequency ablation for paroxysmal atrial fibrillation. N Engl J Med. 2016;74:2235–45. doi:10.1056/NEJMoa1602014.
Wong KC, et al. No benefit OF Complex Fractionated Atrial Electrogram (CFAE) ablation in addition to circumferential pulmonary vein ablation and linear ablation: BOCA study. Circ Arrhythm Electrophysiol. 2015;8:1316–24. doi:10.1161/CIRCEP.114.002504.
Vogler J, et al. Pulmonary vein isolation versus defragmentation: the CHASE-AF clinical trial. J Am Coll Cardiol. 2015;66:2743–52. doi:10.1016/j.jacc.2015.09.088.
Narayan SM, et al. Treatment of atrial fibrillation by the ablation of localized sources: the Conventional Ablation for Atrial Fibrillation With or Without Focal Impulse and Rotor Modulation: CONFIRM trial. J Am Coll Cardiol. 2012;60:628–36.
Haissaguerre M, et al. Driver domains in persistent atrial fibrillation. Circulation. 2014;130:530–8. doi:10.1161/CIRCULATIONAHA.113.005421.
Seitz J, et al. AF ablation guided by spatiotemporal electrogram dispersion without pulmonary vein isolation: a wholly patient-tailored approach. J Am Coll Cardiol. 2017;69:303–21. doi:10.1016/j.jacc.2016.10.065.
Lin Y-J, et al. Benefits of atrial substrate modification guided by electrogram similarity and phase mapping techniques to eliminate rotors and focal sources versus conventional defragmentation in persistent atrial fibrillation. JACC: Clinical Electrophysiology. 2016;2:667–78.
Marrouche NF, et al. Association of atrial tissue fibrosis identified by delayed enhancement MRI and atrial fibrillation catheter ablation: the DECAAF study. JAMA. 2014;311:498–506. doi:10.1001/jama.2014.3.
•• Hansen BJ, et al. Atrial fibrillation driven by micro-anatomic intramural re-entry revealed by simultaneous sub-epicardial and sub-endocardial optical mapping in explanted human hearts. Eur Heart J. 2015;36:2390–401. doi:10.1093/eurheartj/ehv233. First study examining optical imaging of human atrial during fibrillation, demonstrating rotational drivers where ablation terminated AF. The study also performs a detailed structural assessment of tissue near and remote from such drivers and identifies other factors whereby drivers may cause fibrillation.
Allessie MA, et al. Electropathological substrate of long-standing persistent atrial fibrillation in patients with structural heart disease: longitudinal dissociation. Circ Arrhythm Electrophysiol. 2010;3:606–15. doi:10.1161/CIRCEP.109.910125.
Miller JM, et al. Initial independent outcomes from focal impulse and rotor modulation ablation for atrial fibrillation: multicenter FIRM registry. J Cardiovasc Electrophys. 2014;25:921–9.
Rashid H, Sweeney A. Approaches for focal impulse and rotor mapping in complex patients: a US private practice perspective. J Innovations in Cardiac Rhythm Management. 2015;6:2193–8.
Gianni C, et al. Acute and short-term outcomes in persistent and long-standing persistent patients undergoing rotors only ablation (abstract). Heart Rhythm. 2015;12:PO01–58.
•• Miller JM, et al. Clinical benefit of ablating localized sources for human atrial fibrillation: the Indiana University FIRM registry. J Am Coll Cardiol. 2017;69:1247–56. Largest study of ablation of focal and rotational drivers for AF to date, with detailed descriptions of patient cohorts, ablation technique and long-term outcome.
Kottkamp H. Human atrial fibrillation substrate: towards a specific fibrotic atrial cardiomyopathy. Eur Heart J. 2013;34:2731–8. doi:10.1093/eurheartj/eht194.
• Pandit SV, Jalife J. Rotors and the dynamics of cardiac fibrillation. Circ Res. 2013;112:849–62. doi:10.1161/CIRCRESAHA.111.300158. Excellent review on the physiology of cardiac fibrillation and the presence of drivers. This covers physiological issues, mechanical anchoring and historical background.
Rappel WJ, Zaman JA, Narayan SM. Mechanisms for the termination of atrial fibrillation by localized ablation: computational and clinical studies. Circ Arrhythm Electrophysiol. 2015;8:1325–33. doi:10.1161/CIRCEP.115.002956.
Buch E, et al. Long-term clinical outcomes of focal impulse and rotor modulation for treatment of atrial fibrillation: a multi-center experience. Heart Rhythm. 2016;13:636–41. doi:10.1016/j.hrthm.2015.10.031.
Steinberg JS, et al. Focal impulse and rotor modulation: acute procedural observations and extended clinical follow-up. Heart Rhythm. 2016; doi:10.1016/j.hrthm.2016.11.008.
Lee S, et al. Simultaneous bi-atrial high density (510 - 512 electrodes) Epicardial mapping of persistent and long-standing persistent atrial fibrillation in patients: new insights into the mechanism of its maintenance. Circulation. 2015;132:2108–17. doi:10.1161/CIRCULATIONAHA.115.017007.
Baykaner T, et al. Clinical outcomes of AF driver ablation: a systematic review (abstract). Heart Rhythm. 2017;14:AB09–4.
Zhao J, et al. Integration of high-resolution optical mapping and 3-dimensional micro-computed tomographic imaging to resolve the structural basis of atrial conduction in the human heart. Circ Arrhythm Electrophysiol. 2015;8:1514–7. doi:10.1161/CIRCEP.115.003064.
Zaman JA, et al. Improved mapping of human atrial fibrillation using repolarization information to exclude far-field signals (abstract). Heart Rhythm. 2016;13
Narayan SM, Zaman JA. Mechanistically-based mapping of human cardiac fibrillation. J Physiol. 2016;594:2399–415. doi:10.1113/JP270513.
Kuklik P, et al. Reconstruction of instantaneous phase of unipolar atrial contact electrogram using a concept of sinusoidal recomposition and Hilbert transform. IEEE Trans Biomed Eng. 2015;62:296–302. doi:10.1109/TBME.2014.2350029.
Alhusseini M, et al. Two independent mapping techniques identify rotational activity patterns at sites of local termination during persistent atrial fibrillation. J Cardiovasc Electrophys. 2017; in press
Zaman JAB, et al. Recurrent post ablation paroxysmal atrial fibrillation shares substrates with persistent atrial fibrillation: an 11 center study. JACC: Clinical Electrophysiology. 2017;3:393–402. doi:10.1016/j.jacep.2016.10.006. PMID: 28596994.
Vijayakumar R, Vasireddi SK, Cuculich PS, Faddis MN, Rudy Y. Methodology considerations in phase mapping of human cardiac arrhythmias. Circ Arrhythm Electrophysiol. 2016;9 doi:10.1161/CIRCEP.116.004409.
Sommer P, et al. Successful repeat catheter ablation of recurrent longstanding persistent atrial fibrillation with rotor elimination as the procedural endpoint: a case series. J Cardiovasc Electrophysiol. 2016;27:274–80. doi:10.1111/jce.12874.
Tomassoni G, et al. Long-term follow-up of FIRM-guided ablation of atrial fibrillation: a single-center experience. J Innovations in Cardiac Rhythm Management. 2015;6:2145–51.
Narayan SM, et al. Ablation of rotor and focal sources reduces late recurrence of atrial fibrillation compared to trigger ablation alone: extended follow-up of the CONFIRM (CONventional ablation with or without focal impulse and rotor modulation) trial. J Am Coll Cardiol. 2014;63:1761–8.
Narayan SM, Krummen DE, Rappel W-J. Clinical mapping approach to identify rotors and focal beats in human atrial fibrillation. J Cardiovasc Electrophysiol. 2012;23:447–54.
Rodrigo M, et al. Body surface localization of left and right atrial high frequency rotors in atrial fibrillation patients: a clinical-computational study. Heart Rhythm. 2014;11:1584–91. doi:10.1016/j.hrthm.2014.05.013.
Benharash P, et al. Quantitative analysis of localized sources identified by focal impulse and rotor modulation mapping in atrial fibrillation. Circ Arrhythm Electrophysiol. 2015;8:554–61. doi:10.1161/CIRCEP.115.002721.
Gianni C, et al. Acute and early outcomes of FIRM-guided rotors-only ablation in patients with non-paroxysmal atrial fibrillation. Heart Rhythm. 2016;13:830–5. doi:10.1016/j.hrthm.2015.12.028.
Jalife J, Filgueiras Rama D, Berenfeld O. Letter by Jalife et al regarding article, quantitative analysis of localized sources identified by focal impulse and rotor modulation mapping in atrial fibrillation. Circ Arrhythm Electrophysiol. 2015;8:1296–8. doi:10.1161/CIRCEP.115.003324.
Allessie MA, Bonke FIM, Schopman FJG. Circus movement in rabbit atrial muscle as a mechanism of tachycardia. III: the “leading circle” concept: a new model of circus movement in cardiac tissue without the involvement of an anatomical obstacle. Circ Res. 1977;41:9–18.
Lalani GG, et al. Organized sources are spatially conserved in recurrent compared to pre-ablation atrial fibrillation: further evidence for non-random electrical substrates. J Cardiovasc Electrophysiol. 2016;27:661–9. doi:10.1111/jce.12964.
Krummen DE, et al. Multicenter procedural safety of FIRM ablation (abstract). Europace. 2016;
Spitzer SG, et al. Treatment of recurrent non-paroxysmal atrial fibrillation using focal impulse and rotor mapping (FIRM)-guided rotor ablation: early recurrence and long-term outcomes. J Cardiovasc Electrophysiol. 2017;28:31–8. doi:10.1111/jce.13110.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
This work was supported by grants from the National Institutes of Health to Dr. Narayan (HL83359, HL103800). Dr. Zaman is supported by a Fulbright foundation award. Dr. Baykaner is supported by a fellowship from the Heart Rhythm Society. Dr. Narayan is co-author of intellectual property owned by the University of California Regents and licensed to Topera Inc., and has held equity in Topera. Dr. Narayan reports consulting fees and honoraria from the American College of Cardiology, Medtronic, St. Jude, Abbott and Uptodate. Dr. Viswanathan reports clinical studies from Medtronic and St. Jude Medical. Dr. Wang reports Honoraria/Consultant from Janssen, St. Jude Medical, Amgen, Medtronic; Fellowship support from Biosense Webster, Boston Scientific, Medtronic, St. Jude Medical; Clinical studies from Medtronic, Siemens, Cardiofocus, ARCA; and Stock options from Vytronus. Drs. Rodrigo and Baykaner, Mr. Kowalewski, Ms. Shenasa and Ms. Meckler report no conflicts.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Arrhythmias
Rights and permissions
About this article
Cite this article
Narayan, S.M., Rodrigo, M., Kowalewski, C.A.B. et al. Ablation of Focal Impulses and Rotational Sources: What Can Be Learned from Differing Procedural Outcomes?. Curr Cardiovasc Risk Rep 11, 27 (2017). https://doi.org/10.1007/s12170-017-0552-7
Published:
DOI: https://doi.org/10.1007/s12170-017-0552-7