Abstract
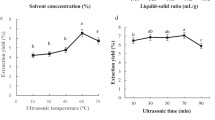
The optimum saponification extracting conditions of sterol of Pleurotus tuber-regium (Fr.) Sing Sclerotium (PTRSS) with ultrasonic were studied by the Response Surface Methodology (RSM). The results of optimum extraction conditions from Box-Behnken model were as follow: ultrasonic time, 48 min; solid-to-liquid ratio, 1:12.5 g/mL; saponification temperature, 81 ℃; saponification concentration, 0.61 mol/L. Under these conditions, the sterol content was 497.89 ± 0.75 µg/g. Fourier transform infrared spectroscopy was used to analyze the structures of the substance and shows that it has the characteristic peak of sterol. In a certain concentration range, the dose-effect relationship between sterol of PTRSS and antioxidant activity. The binding capacity of 4 mg/mL sterol PTRSS to sodium cholate, sodium glycylcholate and sodium taurocholate in vitro was 36.05 ± 0.76%, 29.41 ± 0.64%, 8.90 ± 0.34%, respectively, indicating that it possessed good hypolipodemic effect in vitro.





Similar content being viewed by others
References
Adu JK, Amengor CDK, Kabiri N, Orman E, Patamia SAG, Okrah BK (2019) Validation of a simple and Robust Liebermann–Burchard Colorimetric Method for the assay of cholesterol in selected milk products in Ghana International. J Food Sci 2019:1–7. https://doi.org/10.1155/2019/9045938
Blumfield M, Abbott K, Duve E, Cassettari T, Marshall S, Fayet-Moore F (2020) Examining the health effects and bioactive components in Agaricus Bisporus mushrooms: a scoping review. J Nutr Biochem 84. https://doi.org/10.1016/j.jnutbio.2020.108453
Chen Z, Shen N, Wu X, Jia J, Wu Y, Chiba H, Hui S (2023) Extraction and quantitation of phytosterols from Edible Brown seaweeds: optimization, validation, and Application Foods. 12. https://doi.org/10.3390/foods12020244
Dunford NT, Irmak S, Jonnala R (2009) Effect of the Solvent type and temperature on phytosterol contents and compositions of wheat straw, Bran, and germ extracts. J Agric Food Chem 57:10608–10611. https://doi.org/10.1021/jf9022383
Geng H-X, Yu R-C, Chen Z-F, Peng Q-C, Yan T, Zhou M-J (2017) Analysis of sterols in selected bloom-forming algae. In China Harmful Algae 66:29–39. https://doi.org/10.1016/j.hal.2017.04.008
Gong Y, Luo W, Chen H, Ren B, Hu W, Li L (2022) Systematical Ingredient investigations of Ficus Tikoua Bur. Fruit and Immunoregulatory and antioxidant effects of different fractions molecules. 27. https://doi.org/10.3390/molecules27206880
Hakalin NLS, Molina-Gutiérrez M, Prieto A, Martínez MJ (2018) Optimization of lipase-catalyzed synthesis of β-sitostanol esters by response surface methodology. Food Chem 261:139–148. https://doi.org/10.1016/j.foodchem.2018.04.031
Hammann S, Vetter W (2016) Method Development for the determination of Free and Esterified sterols in Button mushrooms (Agaricus Bisporus). J Agric Food Chem 64:3437–3444. https://doi.org/10.1021/acs.jafc.6b00383
Hasan M, Azhar M, Nangia H, Bhatt PC, Panda BP (2016) Influence of high-pressure homogenization, ultrasonication, and supercritical fluid on free astaxanthin extraction from β-glucanase-treated Phaffia rhodozyma cells. Prep Biochem Biotechnol 46:116–122. https://doi.org/10.1080/10826068.2014.995807
He W-S, Yin J, Xu H-S, Qian Q-Y, Jia C-S, Ma H-L, Feng B (2014) Efficient synthesis and characterization of Ergosterol Laurate in a solvent-free system. J Agric Food Chem 62:11748–11755. https://doi.org/10.1021/jf504516q
He W-S et al (2019a) Cholesterol-reducing effect of ergosterol is modulated via inhibition of cholesterol absorption and promotion of cholesterol excretion. J Funct Foods 57:488–496. https://doi.org/10.1016/j.jff.2019.04.042
He W-S et al (2019b) Candida sp. 99–125 lipase-catalyzed synthesis of ergosterol linolenate and its characterization. Food Chem 280:286–293. https://doi.org/10.1016/j.foodchem.2018.12.076
He WS et al (2022) Enhanced antioxidant capacity of lipoic acid in different food systems through lipase-mediated esterification with phytosterols. J Sci Food Agric 102:7115–7125. https://doi.org/10.1002/jsfa.12073
Heleno SA, Diz P, Prieto MA, Barros L, Rodrigues A, Barreiro MF, Ferreira ICFR (2016) Optimization of ultrasound-assisted extraction to obtain mycosterols from Agaricus Bisporus L. by response surface methodology and comparison with conventional. Soxhlet Extr Food Chem 197:1054–1063. https://doi.org/10.1016/j.foodchem.2015.11.108
Heleno SA et al (2017) Development of dairy beverages functionalized with pure ergosterol and mycosterol extracts: an alternative to phytosterol-based beverages. Food Funct 8:103–110. https://doi.org/10.1039/c6fo01600f
Kalsait RP, Khedekar PB, Saoji AN, Bhusari KP (2011) Isolation of phytosterols and antihyperlipidemic activity of Lagenaria siceraria. Arch Pharm Res 34:1599–1604. https://doi.org/10.1007/s12272-011-1003-5
Meza-Menchaca T et al (2020) Ergosterol peroxide isolated from Oyster Medicinal Mushroom, Pleurotus ostreatus (Agaricomycetes), potentially induces radiosensitivity in cervical. Cancer Int J Med Mushrooms 22:1109–1119. https://doi.org/10.1615/IntJMedMushrooms.2020036673
Qian F, Wang Y, Wen Z-j, Jiang S-j, Tuo Y-f, Mu G-q (2018) Plastein reaction enhanced bile-acid binding capacity of soybean protein hydrolysates and whey protein hydrolysates. J Food Sci Technol 55:1021–1027. https://doi.org/10.1007/s13197-017-3015-8
Quintero-Cabello KP et al (2021) Antioxidant properties and Industrial Uses of Edible Polyporales. J Fungi 7. https://doi.org/10.3390/jof7030196
Rangsinth P et al (2023) Potential Beneficial effects and Pharmacological Properties of Ergosterol, a common bioactive compound in Edible Mushrooms Foods. 12. https://doi.org/10.3390/foods12132529
Sabbione AC, Añón MC, Scilingo A (2023) Characterization and bile acid binding capacity of Dietary Fiber obtained from three different Amaranth Products Plant Foods for Human Nutrition. https://doi.org/10.1007/s11130-023-01116-z
Schlag S, Huang Y, Vetter W (2021) GC/EI-MS method for the determination of phytosterols in vegetable oils. Anal Bioanal Chem 414:1061–1071. https://doi.org/10.1007/s00216-021-03730-9
Shen J-J, Zhu A-S, Gao T-T, Zhao J-Z (2021) Optimization of extraction technology of sterols from discarded soybean pod by response surface methodology Environmental challenges. 5. https://doi.org/10.1016/j.envc.2021.100272
Sommer K, Vetter W (2020) Gas chromatography with mass spectrometry detection and characterization of 27 sterols in two truffle (tuber) species. J Food Compos Anal 94. https://doi.org/10.1016/j.jfca.2020.103650
Sommer K, Krauß S, Vetter W (2020) Differentiation of European and Chinese truffle (tuber sp.) species by means of sterol fingerprints. J Agric Food Chem 68:14393–14401. https://doi.org/10.1021/acs.jafc.0c06011
Souilem F et al (2017) Wild mushrooms and their mycelia as sources of bioactive compounds: Antioxidant, anti-inflammatory and cytotoxic properties. Food Chem 230:40–48. https://doi.org/10.1016/j.foodchem.2017.03.026
Srihera N, Li Y, Zhang T-T, Wang Y-M, Yanagita T, Waiprib Y, Xue C-H (2022) Preparation and characterization of astaxanthin-loaded liposomes stabilized by Sea Cucumber Sulfated Sterols instead of cholesterol. J Oleo Sci 71:401–410. https://doi.org/10.5650/jos.ess21233
Sun S et al (2022) New insights on the analysis of phytosterols in pollen and anther wall of tree peony (Paeonia ostii ‘Fengdan’) revealed by GC-MS/MS Analytica Chimica Acta 1212. https://doi.org/10.1016/j.aca.2022.339891
Tan Z, Shahidi F (2011) Optimization of enzymatic synthesis of Phytosteryl Caprylates using response surface methodology. J Am Oil Chem Soc 89:657–666. https://doi.org/10.1007/s11746-011-1949-y
Wang L, Weller CL (2006) Recent advances in extraction of nutraceuticals from plants Trends. Food Sci Technol 17:300–312. https://doi.org/10.1016/j.tifs.2005.12.004
Wang C, Liu Y, Lan Y, Yuan J (2022) Extraction of a triterpene solution and evaluation of the Hypolipidemic efficacy of the Pleurotus tuber-regium (Fr). Sing Sclerotium Foods 11. https://doi.org/10.3390/foods11182881
Wu G-H, Hu T, Li Z-Y, Huang Z-L, Jiang J-G (2014) In vitro antioxidant activities of the polysaccharides from Pleurotus tuber-regium (Fr). Sing Food Chemistry 148:351–356. https://doi.org/10.1016/j.foodchem.2013.10.029
Yu G, Yue C, Zang X, Chen C, Dong L, Liu Y (2019) Purification, characterization and in vitro bile salt-binding capacity of polysaccharides from Armillaria mellea mushroom Czech. J Food Sci 37:51–56. https://doi.org/10.17221/182/2018-cjfs
Zheng M-M et al (2012) Ultrasonic pretreatment for lipase-catalyed synthesis of phytosterol esters with different acyl donors. Ultrason Sonochem 19:1015–1020. https://doi.org/10.1016/j.ultsonch.2012.02.004
Acknowledgements
This work was supported by the Basic Public Welfare Research Program of Zhejiang Province (No. LGN22C200001) and the school-level Student Research Training Program of Zhejiang A & F University (No. S202210341206).
Author information
Authors and Affiliations
Contributions
Chao and Yuan wrote the main manuscript text. Yuanhong and Jianing prepared figures ang tables. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Wang, C., Lan, Y. et al. Optimization of Extraction Process of Sterol of Pleurotus Tuber-Regium (Fr.) Sing Sclerotium and Their Antioxidant and Hypolipidemic Effects. Food Anal. Methods 17, 349–357 (2024). https://doi.org/10.1007/s12161-024-02572-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-024-02572-2