Abstract
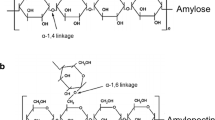
Chemical composition and grain quality traits are important factors related to the end-use quality of sorghum. Thus, rapid and non-destructive analytical methods for screening sorghum for desirable grain quality traits benefit the industries using sorghum for food, feed, and biofuels as well as sorghum breeders and seed companies working to improve the grain quality of sorghum. In the present study, a method based on Fourier-transform infrared spectroscopy (FTIR) was developed to characterize the major chemical components in sorghum flour. Sorghum flour had three major FTIR active regions from 1800 to 800 cm−1. As expected, FTIR spectra revealed that starch was the primary compound in sorghum flour, followed by protein with only minor amounts of lipids and phenolic acids. Phenolic acids were characterized by a peak at 1709 cm−1, which varied in intensity across sorghum varieties. Tannin sorghum could be discriminated from non-tannin sorghum by a shift in O–H stretch band around 3300 cm−1 and their C=C absorption peaks at 1608 and 1522 cm−1. Flour from the outer part of the kernel (corneous endosperm) had greater protein and lipid absorbance while the inner kernel (floury endosperm) had stronger starch absorbance. Pearson’s correlation analyses between FTIR peak intensities and protein/starch content showed that starch content did not have a significant correlation with infrared peaks, whereas the amide I peak at 1652 cm−1 (C=O stretching group), highly correlated with protein content (P ≤ 0.05). A calibration curve (Y = 127.79X-14.345, R2 = 0.9454) was built using peak height at 1652 cm−1 to predict protein content and this was applied to determining protein digestibility.









Similar content being viewed by others
Abbreviations
- FTIR:
-
Fourier-transform infrared spectroscopy
- ATR:
-
Attenuated total reflection
- DA:
-
Discrimination analysis
- SEM:
-
Scan electron microscopy
- IVPD:
-
In vitro protein digestibility
References
AACC International (1999) Approved Methods of Analysis, 11th Ed. Method 46-30.01 Crude protein-combustion method. AACC International, St. Paul, MN. https://doi.org/10.1094/AACCIntMethod-46-30.01
Awika JM, Rooney LW (2004) Sorghum phytochemicals and their potential impact on human health. Phytochemistry 65:1199–1221
Babushkin V, Spiridonov A, Kozhukhar A (2016) Application of NIR and FTIR in food analysis. J Phys Sci Appl 6:47–50
Barth A (2007) Infrared spectroscopy of proteins. Biochim Biophys Acta 1767:1073–1101
Bean SR, Wilson JD, Moreau R, Gallant A, Awika JM, Kaufman RC, Adrianos S, Ioerger BP (2016) Structure and composition of the grain. In: Ciampitti I, Prasad V, editors, Sorghum: State of the Art and Future Perspectives, Agron. Monogr. 58. ASA and CSSA, Madison, WI. https://doi.org/10.2134/agronmonogr58.2014.0081
Bean SR, Zhu L, Smith BM, Wilson JD, Ioerger BP, Tilley M (2019) Starch and protein chemistry and functional properties. In: Taylor JRN, Duodu K (eds) Sorghum and millets: chemistry and technology, 2nd edn. Elsevier, San Diego, CA, pp 131–170
Bunaciu AA, Fleschin S, Aboul-Enein HY (2014) Infrared microscopy applications; review. Curr Anal Chem 10:132–139
Byler DM, Sui H (1986) Examination of secondary structure of proteins by deconvolved FTIR spectra. Biopolymers 25:469–487
Carbonaro M, Nucara A (2010) Secondary structure of food proteins by Fourier transform spectroscopy in the mid-infrared region. Amino Acids 38:679–690
Casa AM, Pressoir G, Brown PJ, Mitchell SE, Rooney WL, Tuinstra MR, Franks CD, Kresovitch S (2008) Community resources and strategies for association mapping in sorghum. Crop Sci 48:30–40
Chandrashekar A, Satyanarayana KV (2006) Disease and pest resistance in grains of sorghum and millets. J Cereal Sci 44:287–304
Chang SKC, Zhang Y (2017) Protein analysis. In: Nielsen S (ed) Food analysis. Food Science Text Series, Springer, Cham. https://doi.org/10.1007/978-3-319-45776-5_18
Chen Y, Ye, Liu J (2014) Effects of different concentrations of ethanol and isopropanol on physicochemical properties of zein-based films. Ind Crop Prod 53:140–147
Ciacci C, Maiuri L, Caporaso N, Bucci C, Giudice LD, Massardo DR, Pontieri P, Fonzo ND, Bean SR, Ioerger B, Londei M (2007) Celiac disease: in vitro and in vivo safety and palatability of wheat free sorghum food products. Clin Nutr 26:799–805
Coates J (2000) Interpretation of infrared spectra, a practical approach, encyclopedia of analytical chemistry. R.A. Meyers (Ed.) pp. 10815–10837 John Wiley & Sons Ltd, Chichester
Corredor DY, Bean SR, Wang D (2007) Pretreatment and enzymatic hydrolysis of sorghum bran. Cereal Chem 84:61–66
Dean JA (1999) Lange’s handbook of chemistry, section 7.5, 5th edn. McGraw Hill, United States of America
Dong A, Huang P, Caughey WS (1990) Protein secondary structure in water from second-derivative amide infrared spectra. Biochemistry 29:3303–3308
Duodu KG, Tang H, Grant A, Wellner N, Belton PS, Taylor JRN (2001) FTIR and solid state 13C NMR spectroscopy of proteins of wet cooked and popped sorghum and maize. J Cereal Sci 33:261–269
Elkhalifa AEO, Georget DMR, Barker SA, Belton PS (2009) Study of the physical properties of kafirin during the fabrication of tablets for pharmaceutical applications. J Cereal Sci 50:159–165
Ezeogu LI, Duodu KG, Taylor JRN (2005) Effects of endosperm texture and cooking conditions on the in vitro starch digestibility of sorghum and maize flours. Cereal Chem 42:33–44
Gao Y, Taylor J, Wellner N, Byaruhanga YB, Parker ML, Mills CEN, Belton PS (2005) Effect of preparation conditions on protein secondary structure and biofilm formation of kafirin. J Agric Food Chem 53:306–312
Hagerman AE, Butler LG (1980) Condensed tannin purification and characterization of tannin-associated proteins. J Agric Food Chem 28:947–952
Hahn DH, Faubion JM, Rooney LW (1983) Sorghum phenolic acids, their high-performance liquid chromatography separation and their relation to fungal resistance. Cereal Chem 60:255–259
Henley EC, Taylor JRN, Obukosia SD (2010) The importance of dietary protein in human health: combating protein deficiency in sub-Saharan Africa through transgenic biofortified sorghum. Adv Food Nutr Res 60:21–52
Herald T, Gadgil P, Perumal R, Bean SR, Wilson TJ (2014) High-throughput micro-plate HCl-vanillin assay for screening tannin content in sorghum grain. J Sci Food Ag 94:2133–2136
Holding DR (2014) Recent advances in the study of prolamin storage protein organization and function. Front Plant Sci 5:article 216, 1-9
Ioerger B, Bean SR, Tuinstra MR, Pedersen JF, Erpelding J, Lee K, Herrman T (2007) Characterization of polymeric proteins from vitreous and floury sorghum endosperm. J Agric Food Chem 55:10232–10239
Jeong S, Kim HW, Lee S (2017) Rheological and secondary structural characterization of rice flour-zein composites for noodles slit from gluten-free sheeted dough. Food Chem 221:1539–1545
Klopfenstein CF, Hoseney RC (1995) Nutritional properties of sorghum and the millets. In: Dendy DSAV (ed) Sorghum and millet: chemistry and technology. American Association of Cereal Chemists, St. Paul, MN, pp 125–168
McCleary BV, Gibson TS, Solah V, Mugford DC (1994) Total starch measurement in cereal products: interlaboratory evaluation of a rapid enzymic test procedure. Cereal Chem 71:501–505
Mertz ET, Hassen MM, Carins-Whittern C, Kirleis AW, Tu L, Axtell JD (1984) Pepsin digestibility of proteins in sorghum and other major cereals. PNAS 81:1–2
Nunn S, Nishikida K (2008) Advanced ATR correction algorithm, application note:50581. Thermo Fisher Scientific, Madison, WI, SUA
Prates LL, Refat B, Lei Y, Louzada-Prates M, Yu P (2018) Relationship of carbohydrates and lignin molecular structure spectral profiles to nutrient profile in newly developed oats cultivars and barley grain. Spectrochim Acta A 188:495–506
Price ML, Van Scoyoc S, Butler LG (1978) A critical evaluation of the vanillin reaction as an assay for tannin in sorghum grain. J Agri Food Chem 26:1214–1218
Rhodes DH, Hoffman L, Rooney WL, Ramu P, Morris GP, Kresovich S (2014) Genome-wide association study of grain polyphenol concentrations in global sorghum [Sorghum bicolor (L.) Moench] germplasm. J Agric Food Chem 62:10916–10927
Rhodes D, Gadgil P, Perumal R, Tesso T, Herald TJ (2017) Natural variation and genome-wide association study of antioxidants in a diverse sorghum collection. Cereal Chem 94:190–198
Robbins RJ (2003) Phenolic acids in foods: an overview of analytical methodology. J Agric Food Chem 51:2866–2887
Shi H, Yu P (2017) Comparison of grating-based near-infrared (NIR) and Fourier transform mid-infrared (ATR-FT/MIR) spectroscopy based on spectral preprocessing and wavelength selection for the determination of crude protein and moisture content in wheat. Food Control 82:57–65
Silverstein RM, Webster FX, Kiemle DJ, Bryce DL (2014) Spectrometric identification of organic compounds, 8th edn ISBN: 978-0-470-61637-6
Sun SQ, Chen JB, Zhou Q (2011) Infrared spectroscopy for complex mixtures. Chemical industry press, China
Taylor JRN, Emmambux MN (2010) Developments in our understanding of sorghum polysaccharides and their benefits. Cereal Chem 87:263–271
van de Voort FR (1992) Fourier transform infrared spectroscopy applied to food analysis. Food Res Int 5:397–403
Vu TH, Bean S, Hsieh CF, Shi YC (2017) Preparation and characterization of sorghum flour with increased resistant starch content. J Sci Food Agric 97:4770–4779
Wang J, Yue Y, Liu T, Zhang B, Wang Z, Zhang C (2017) Change in glutenin macropolymer secondary structure in wheat sourdough fermentation by FTIR. Interdiscip Sci Comput Life Sci 9:247–253
Williams DH, Fleming I (1995) Spectroscopic methods in organic chemistry, chapter 2. McGraw Hill
Wu Y, Guo T, Mu Q, Wang J, Li X, Wu Y, Tian B, Wang ML, Bai G, Perumal R, Trick HN, Dweikat IM, Tuinstra MR, Bean SR, Morris G, Tesso TT, Li X (2019) Allelochemicals targeted to balance competing selections in African agroecosystems. Nature Plants 5:1229–1263
Yang H, Yang S, Kong J, Dong A, Yu S (2015) Obtaining information about protein secondary structures in aqueous solution suing Fourier transform FTIR spectroscopy. Nat Protoc 10:382–396
Zhao R, Bean SR, Ioerger BP, Wang D, Boyle DL (2008) Impact of mashing on sorghum proteins and its relationship to ethanol fermentation. J Agric Food Chem 56:946–953
Funding
This research was partially supported by the United Sorghum Checkoff Program (USCP) [grant number Cl011 – 15].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
H. Lin declares that he has no conflict of interest. S. R. Bean declares that he has no conflict of interest. M. Tilley declares that he has no conflict of interest. K. H. S. Peiris declares that he has no conflict of interest. D. Brabec declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Informed consent is not applicable.
Disclaimer
Names are necessary to report factually on available data; however, the U.S. Department of Agriculture neither guarantees nor warrants the standard of the product and use of the name by the U.S. Department of Agriculture implies no approval of the product to the exclusion of others that may also be suitable. USDA is an equal opportunity provider and employer.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lin, H., Bean, S.R., Tilley, M. et al. Qualitative and Quantitative Analysis of Sorghum Grain Composition Including Protein and Tannins Using ATR-FTIR Spectroscopy. Food Anal. Methods 14, 268–279 (2021). https://doi.org/10.1007/s12161-020-01874-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01874-5