Abstract
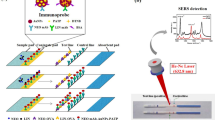
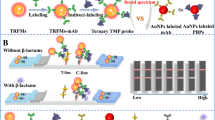
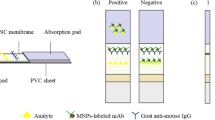
Nowadays, the overuse of antibiotics in cow breeding is a common issue worldwide. Real-time monitoring is critical for antibiotic control of dairy products. Herein, we report a rapid detection method for the duplex detection of two beta-lactam antibiotics (cephalexin and ampicillin) in milk powder using lateral flow (LF) combined with surface-enhanced Raman spectroscopy (SERS). The duplex detection was accomplished using a single test line loaded with two antigens targeting the two corresponding antibody-gold-Raman reporter conjugates. The obtained SERS spectra contributed by the Raman reporter labeled to the two antibodies were analyzed using both peak height method and multiple linear regression (MLR). A good regression fitting was achieved by these two methods (R2 = 0.99 and 0.98, respectively). The limit of detection (LOD) was 10−4 ng/mL for both peak height method and MRL. This study indicated that LF-SERS is a good candidate for real-time multiplex and quantitative analysis for antibiotics in dairy products.




Similar content being viewed by others
Data Availability
Not applicable.
Change history
09 February 2022
A Correction to this paper has been published: https://doi.org/10.1007/s12161-022-02227-0
References
Anfossi L, Di Nardo F, Profiti M, Nogarol C, Cavalera S, Baggiani C, Giovannoli C, Spano G, Ferroglio E, Mignone W, Rosati S (2018) A versatile and sensitive lateral flow immunoassay for the rapid diagnosis of visceral leishmaniasis. Anal Bioanal Chem 410:4123–4134. https://doi.org/10.1007/s00216-018-1067-x
Bian S, Chu X, Jin Y, Xing S, Zhang Y, Hu H (2013) A novel microsphere-based fluorescence immunochromatographic assay for monitoring cefalexin residues in plasma, milk, muscle and liver. Anal Methods 5:6441–6448. https://doi.org/10.1039/c3ay41487f
Byzova NA, Zvereva EA, Zherdev AV, Dzantiev BB (2011) Immunochromatographic technique for express determination of ampicillin in milk and dairy products. Appl Biochem Microbiol 47:627–634. https://doi.org/10.1134/S0003683811060032
Di Nardo F, Alladio E, Baggiani C, Cavalera S, Giovannoli C, Spano G, Anfossi L (2019) Colour-encoded lateral flow immunoassay for the simultaneous detection of aflatoxin B1 and type-B fumonisins in a single test line. Talanta 192:288–294. https://doi.org/10.1016/j.talanta.2018.09.037
Zhang D, Huang L, Liu B, Ni H, Sun L, Su E, Chen H, Gu Z, Zhao X (2018a) Quantitative and ultrasensitive detection of multiplex cardiac biomarkers in lateral flow assay with core-shell SERS nanotags. Biosens Bioelectron 106:204–211. https://doi.org/10.1016/j.bios.2018.01.062
Zhang D, Huang L, Liu B, Su E, Chen H-Y, Gu Z, Zhao X (2018b) Quantitative detection of multiplex cardiac biomarkers with encoded SERS nanotags on a single T line in lateral flow assay. Sensors Actuators B-Chem 277:502–509. https://doi.org/10.1016/j.snb.2018.09.044
Zhang D, Huang L, Liu B, Ge Q, Dong J, Zhao X (2019) Rapid and ultrasensitive quantification of multiplex respiratory tract infection pathogen via lateral flow microarray based on SERS nanotags. Theranostics 9:4849–4859. https://doi.org/10.7150/thno.35824
Dzantiev BB, Byzova NA, Urusov AE, Zherdev AV (2014) Immunochromatographic methods in food analysis. TrAC Trends Anal Chem 55:81–93. https://doi.org/10.1016/j.trac.2013.11.007
Frens G (1973) Controlled nucleation for the regulation of the particle size in monodisperse gold suspensions. Nat Phys Sci 241:20–22. https://doi.org/10.1038/physci241020a0
Guo J, Liu L, Xue F, Xing C, Song S, Kuang H, Xu C (2015) Development of a monoclonal antibody-based immunochromatographic strip for cephalexin. Food Agric Immunol 26:282–292. https://doi.org/10.1080/09540105.2014.907242
Harlow E, Lane D (1988) Antibodies: a laboratory manual. Cold Spring Harbor Laboratory, New York
Kenkel J (2003) Analytical chemistry for technicians, 3rd edn. Lewis Publishers, Boca Raton
Landers TF, Cohen B, Wittum TE, Larson EL (2012) A review of antibiotic use in food animals: perspective, policy, and potential. Public Health Rep 127:4–22. https://doi.org/10.1177/003335491212700103
Laxminarayan R, Duse A, Wattal C, Zaidi AKM, Wertheim HFL, Sumpradit N, Vlieghe E, Hara GL, Gould IM, Goossens H, Greko C, So AD, Bigdeli M, Tomson G, Woodhouse W, Ombaka E, Peralta AQ, Qamar FN, Mir F, Kariuki S, Bhutta ZA, Coates A, Bergstrom R, Wright GD, Brown ED, Cars O (2013) Antibiotic resistance—the need for global solutions. Lancet Infect Dis 13:1057–1098. https://doi.org/10.1016/S1473-3099(13)70318-9
Lutz BR, Dentinger CE, Nguyen LN, Sun L, Zhang J, Allen AN, Chan S, Knudsen BS (2008) Spectral analysis of multiplex Raman probe signatures. ACS Nano 2:2306–2314. https://doi.org/10.1021/nn800243g
Posthuma-Trumpie GA, Korf J, van Amerongen A (2009) Lateral flow (immuno)assay: its strengths, weaknesses, opportunities and threats. A literature survey. Anal Bioanal Chem 393:569–582. https://doi.org/10.1007/s00216-008-2287-2
Quesada-González D, Merkoçi A (2015) Nanoparticle-based lateral flow biosensors. Biosens Bioelectron 73:47–63. https://doi.org/10.1016/j.bios.2015.05.050
Sachi S, Ferdous J, Sikder MH, Azizul Karim Hussani SM (2019) Antibiotic residues in milk: past, present, and future. J Adv Vet Anim Res 6:315–332. https://doi.org/10.5455/javar.2019.f350
Shi Q, Huang J, Sun Y, Deng R, Teng M, Li Q, Yang Y, Hu X, Zhang Z, Zhang G (2018) A SERS-based multiple immuno-nanoprobe for ultrasensitive detection of neomycin and quinolone antibiotics via a lateral flow assay. Mikrochim Acta 185:84. https://doi.org/10.1007/s00604-017-2556-x
Stiles PL, Dieringer JA, Shah NC, van Duyne RP (2008) Surface-enhanced Raman spectroscopy. Annu Rev Anal Chem (Palo Alto, Calif) 1:601–626. https://doi.org/10.1146/annurev.anchem.1.031207.112814
Tao WY, Zhao AW, Sun HH, Gan ZB, Zhang MF, Li D, Guo HY (2014) Periodic silver nanodishes as sensitive and reproducible surface-enhanced Raman scattering substrates. RSC Adv 4:3487–3493. https://doi.org/10.1039/c3ra45935g
Urusov AE, Zherdev AV, Dzantiev BB (2019) Towards lateral flow quantitative assays: detection approaches: detection approaches. Biosensors-Basel 9:89. https://doi.org/10.3390/bios9030089
Funding
This work was supported by the Shenyang Industrial Science and Technology Program (grant number F11-049-2-00).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Informed Consent
Not applicable.
Code Availability
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, X., Wang, X., Wang, L. et al. Duplex Detection of Antibiotics in Milk Powder Using Lateral-Flow Assay Based on Surface-Enhanced Raman Spectroscopy. Food Anal. Methods 14, 165–171 (2021). https://doi.org/10.1007/s12161-020-01870-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01870-9