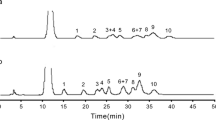
Abstract
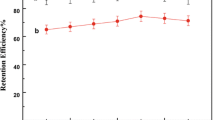
In this work an HPLC/hydrophilic interaction liquid chromatography (HILIC) system for intact glucosinolates was optimized using a weak anion exchange resin to remove carboxylic phenolics that absorbed at the same wavelength (229 nm) as glucosinolates, by selectively protonating contaminating phenolics with pKas from 2.98 to 4 while leaving the second OH of the sulfate of the thioglycosidic linkage (pKa = 1.92) ionized. Glucosinolates bound to the anion exchange resin, while the contaminating phenolics washed through. The extraction was performed in 2-ml snap top centrifuge tubes with 50 to 80 mg of fresh tissue in 0.3 ml of extractant. The corresponding spin tubes contained 20–22 mg of anion exchange resin and were loaded with 100 μl of extractant. A 48-well pressure plate was used to load, wash, and finally elute the glucosinolates without a centrifuge into autosampler vials. Seventeen glucosinolates found in common vegetables were separated in a 16-min isocratic run vs 50 min for the C18 gradient used in the desulfoglucosinolate method. The recovery of a spiked sample of phenethyl glucosinolate was greater than 90%, and the limit of detection (LOD) and limit of quantitation (LOQ) were 6.85 and 20.78 nmol respectively. The time from sample weighing to the end of the first chromatographic run was approximately 2 h.








Similar content being viewed by others
Abbreviations
- HILIC column:
-
Hydrophilic interaction liquid chromatography
- DEAE:
-
Diethylaminoethyl
- PTFE:
-
Polytetrafluoroethylene
- PolyWAXLP:
-
Weak anion exchange resin with linear polyethyleneimine
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantitation
- DAD:
-
diode array detector
- PEEK:
-
Polyetheretherketone
- mAU:
-
Milli-absorbance unit
- pKa:
-
Negative log of the acid dissociation constant
- HPLC:
-
High pressure liquid chromatography
References
Ares AM, Bernal J, Nozal MJ, Turner C, Plaza M (2015) Fast determination of intact glucosinolates in broccoli leaf by pressurized liquid extraction and ultra-high-performance liquid chromatography coupled to quadrupole time-of-flight mass spectrometry. Food Res Int 76:498–505
Bianco G, Pascale R, Lelario F, Bufo SA, Tommaso R (2016) The investigation of glucosinolates by mass spectrometry. In: Kopriva, S Glucosinolates Advances in Botanical Research, 80: 3-30. ISBN-13: 978-0081003275
Buchner, R (1987) Approach to determination of HPLC response factors for glucosinolates. in Wathelet, J.-P (Ed) Glucosinolates in rapeseeds: analytical aspects Springer, Dordrecht, pp. 50–58
Capriotti AL, Cavaliere C, La Barbera G, Montone CM, Piovesana S, Chiozzi RZ, Laganà A (2018) Chromatographic column evaluation for the untargeted profiling of glucosinolates in cauliflower by means of ultra-high-performance liquid chromatography coupled to high resolution mass spectrometry. Talanta 179:792–802
Clarke DB (2010) Glucosinolates, structures and analysis in food. Anal Methods 2:310–325
Erdemgil FZ, Şanli S, Şanli N, Özkan G, Barbosa J, Guiteras J, Beltran JL (2007) Determination of pKa values of some hydroxylated benzoic acids in methanol–water binary mixtures by LC methodology and potentiometry. Talanta 72:489–496
Fahey JW, Zalcmann AT, Talalay P (2001) The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochem 56:5–51
Felker P, Bunch R, Leung AM (2016) Concentrations of thiocyanate and goitrin in human plasma, their precursor concentrations in brassica vegetables, and associated potential risk for hypothyroidism. Nutr Rev 74:248–258
Glauser G, Schweizer F, Turlings TC, Reymond P (2012) Rapid profiling of intact glucosinolates in Arabidopsis leaves by UHPLC-QTOFMS using a charged surface hybrid column. Phytochem Anal 23(5):520–528
Haupt GW (1952) An alkaline solution of potassium chromate as a transmittancy standard in the ultraviolet. JOSA 42:441–447
James D, Devaraj S, Bellur P, Lakkanna S, Vicini J, Boddupalli S (2012) Novel concepts of broccoli sulforaphanes and disease: induction of phase II antioxidant and detoxification enzymes by enhanced-glucoraphanin broccoli. Nutr Rev 70:654–665
Klopsch R, Witzel K, Börner A, Schreiner M, Hanschen FS (2017) Metabolic profiling of glucosinolates and their hydrolysis products in a germplasm collection of Brassica rapa turnips Food Res. Int. 100:392–403
Kopriva, S. (Editor) (2016). Glucosinolates advances in botanical research. Volume 80, Pages 1–350. ISBN-13: 978–0081003275
Olsen CE, Huang X-C, Hansen CIC, Cipollini D, Ørgaard M, Matthes A, Geu-Flores F, Koch MA, Agerbirk N (2016) Glucosinolate diversity within a phylogenetic framework of the tribe Cardamineae (Brassicaceae) unraveled with HPLC-MS/MS and NMR-based analytical distinction of 70 desulfoglucosinolates. Phytochem 132:33–56
Park JE, Sun Y, Lim SK, Tam JP, Dekker M, Chen H, Sze SK (2017) Dietary phytochemical PEITC restricts tumor development via modulation of epigenetic writers and erasers. Sci Rep 7:40569
Schwimmer S (1961) Spectral changes during action of myrosinase on sinigrin. Acta Chem Scand A 15:535–544
Troyer JK, Stephenson KK, Fahey JW (2001) Analysis of glucosinolates from broccoli and other cruciferous vegetables by hydrophilic interaction liquid chromatography. J Chromatogr A A 919:299–304
Truscott RJ, Burke DG, Minchinton IR (1982) The characterization of a novel hydroxindole glucosinolate. Biochem Biophys Res Com 107:258–1264
Wade KL, Garrard IJ, Fahey JW (2007) Improved hydrophilic interaction chromatography method for the identification and quantification of glucosinolates. J Chromatogr A 1154:469–472
Witzel K, Hanschen FS, Schreiner M, Krumbein A, Ruppel S, Grosch R (2013) Verticillium suppression is associated with the glucosinolate composition of Arabidopsis thaliana leaves. PLoS One 8(9):e71877
Yang B, Quiros CF (2010) Survey of glucosinolate variation in leaves of Brassica rapa crops. Genet Resour Crop Evol 57:1079–1089
Acknowledgments
The authors gratefully acknowledge the advice and support of K. Wade and J. Fahey of Johns Hopkins in getting started, Niels Agerbirk for assistance with the relative absorbances at 229 nm, and Amos Heckendorf of the Nest Group for many helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding Information
All the funding for this research was D’Arrigo Bros in-house funding.
Conflict of Interest
The authors are employees of a major agricultural company growing brassica vegetables and would like to see more of these vegetables consumed and their beneficial properties reported in the literature. Peter Felker declares that he has no conflict of interest. Ronald Bunch declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Felker, P., Bunch, R.A. Intact Glucosinolate Determination in Brassica Vegetables Using Anion Exchange Cleanup and HILIC (Hydrophilic Interaction Liquid Chromatography) HPLC Chromatography. Food Anal. Methods 13, 1817–1828 (2020). https://doi.org/10.1007/s12161-020-01798-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01798-0