Abstract
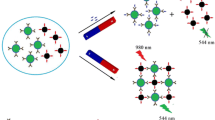
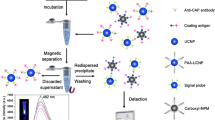
A visual fluorescence quenching immunoaffinity test column (FQ-ITC) assay was developed for the simultaneous detection of malachite green (MG) and crystal violet (CV) residues in aquatic products. The proposed FQ-ITC assay was based on fluorescence resonance energy transfer from the fluorescence donor (quantum dot) to the fluorescence acceptor (MG or CV) and the specific identification between the analytes and their polyclonal antibodies. The process can be completed within 10 min, and the results can be observed by the naked eye. After the evaluation of the specificity and sample matrix effect, the established FQ-ITC was found to be successfully applied in the detection of MG or CV in grass carp, striped bass, carp, and shrimp, with a visual limit of detection of 2 μg/kg. The accuracy of the FQ-ITC assay was verified by comparison with liquid chromatography-tandem mass spectrometry. The simple, time-saving, sensitive, and visual observation characteristics make the established FQ-ITC assay an effective analytical tool for the simultaneous detection of MG and CV in aquatic products.






Similar content being viewed by others
References
Alderman DJ, Clifton-Hadley RS (1993) Malachite green: a pharmacokinetic study in rainbow trout, Oncorhynchus mykiss (Walbaum). J Fish Dis 16:297–311. https://doi.org/10.1111/j.1365-2761.1993.tb00864.x
Andersena WC, Turnipseed SB, Karbiwnyk CM, Lee RH, Clark SB, Rowe WD, Madson MR, Miller KE (2009) Multiresidue method for the triphenylmethane dyes in fish: malachite green, crystal (gentian) violet, and brilliant green. Anal Chim Acta 637:279–289. https://doi.org/10.1016/j.aca.2008.09.041
Cha CJ, Doerge DR, Cerniglia CE (2001) Biotransformation of malachite green by the fungus Cunninghamella elegans. Appl Environ Microbiol 67:4358–4360. https://doi.org/10.1128/AEM.67.9.4358-4360.2001
Chen LY, Tse WH, Chen Y, McDonald MW, Melling J, Zhang J (2017) Nanostructured biosensor for detecting glucose in tear by applying fluorescence resonance energy transfer quenching mechanism. Biosens Bioelectron 91:393–399. https://doi.org/10.1016/j.bios.2016.12.044
Day MJ (2015) Introduction to antigen and antibody assays. Top Companion Anim Med 30:128–131. https://doi.org/10.1053/j.tcam.2015.12.001
Doerge DR, Churchwell MI, Gehring TA, Pu YM, Plakas SM (1998) Analysis of malachite green and metabolites in fish using liquid chromatography atmospheric pressure chemical ionization mass spectrometry. Rapid Commun Mass Spectrom 12:1625–1634. https://doi.org/10.1002/(SICI)1097-0231(19981115)12:21<1625::AID-RCM373>3.0.CO;2-I
Dutta AK (1995) Excitation energy transfer between erythrosin B and malachite green adsorbed on a microheterogeneous polymer latex. Solid State Commun 95:159–165. https://doi.org/10.1016/0038-1098(95)00234-0
Fang L, Deng JW, Yu Y, Yang YY, Wang XW, Liu HT, Luan TG (2016) Coupling liquid-phase microextraction with paper spray for rapid analysis of malachite green, crystal violet and their metabolites in complex samples using mass spectrometry. Anal Methods 8:6651–6656. https://doi.org/10.1039/c6ay01466f
Feng XB, Gan N, Zhang HR, Yan Q, Li TH, Cao YT, Hu FT, Yu HW, Jiang QL (2015) A novel “dual-potential” electrochemiluminescence aptasensor array using CdS quantum dots and luminol-gold nanoparticles as labels for simultaneous detection of malachite green and chloramphenicol. Biosens Bioelectron 74:587–593. https://doi.org/10.1016/j.bios.2015.06.048
Fu WL, Zhen SJ, Huang CZ (2013) One-pot green synthesis of graphene oxide/gold nanocomposites as SERS substrates for malachitegreen detection. Analyst 138(10):3075–3081. https://doi.org/10.1039/c3an00018d
Ghasemi E, Kaykhaii M (2016) Application of micro-cloud point extraction for spectrophotometric determination of malachite green, crystal violet and rhodamine B in aqueous samples. Spectrochim Acta A 164:93–97. https://doi.org/10.1016/j.saa.2016.04.001
Gu X, Zhou J, Zhou L, Xie SS, Petti L, Wang SM, Wang FY (2018) Specific binding of antigen-antibody in physiological environment: measurement, force characteristics and analysis. Opt Lasers Eng 104:252–258. https://doi.org/10.1016/j.optlaseng.2017.09.016
Hidayah APN, Faridah S, Azura MSN, Gayah AR, Othman M, Fatimah AB (2016) Malachite green and leuco-malachite green detection in fish using modified enzyme biosensor. Proc Chem 20:85–89. https://doi.org/10.1016/j.proche.2016.07.014
Kong LY, Li Y, Ma C, Liu BH, Tan L (2018) Sensitive immunoassay of von Willebrand factor based on fluorescence resonance energy transfer between graphene quantum dots and Ag@Au nanoparticles. Colloids Surf B: Biointerfaces 165:286–292. https://doi.org/10.1016/j.colsurfb.2018.02.049
Li L, Peng AH, Lin ZZ, Zhong HP, Chen XM, Huang ZY (2017) Biomimetic ELISA detection of malachite green based on molecularly imprinted polymer film. Food Chem 229:403–408. https://doi.org/10.1016/j.foodchem.2017.02.090
Lopez-Gutierrez N, Romero-Gonzalez R, Plaza-Bolanos P, Martinez-Vidaal JL, Garrido-Frenich A (2013) Simultaneous and fast determination of malachite green, leucomalachite green, crystal violet, and brilliant green in seafood by ultra high-performance liquid chromatography-tandem mass spectrometry. Food Anal Methods 6:406–414. https://doi.org/10.1007/s12161-012-9456-9
Manzo V, Navarro O, Honda L, Sanchez K, Toral MI, Richter P (2013) Determination of crystal violet in water by direct solid phase spectrophotometry after rotating disk sorptive extraction. Talanta 106:305–308. https://doi.org/10.1016/j.talanta.2012.11.004
Panandiker A, Fernandes C, Rao TK, Rao KV (1993) Morphological transformation of Syrian hamster embryo cells in primary culture by malachite green correlates well with the evidence for formation of reactive free radicals. Cancer Lett 74:31–36. https://doi.org/10.1016/0304-3835(93)90040-G
Rolinski OJ, Birch DJS, McCartney LJ, Pickup JC (1999) Fluorescence resonance energy transfer from allophycocyanin to malachite green. Chem Phys Lett 309:395–401. https://doi.org/10.1016/S0009-2614(99)00707-1
Shalaby AR, Emam WH, Anwar MM (2017) Mini-column assay for rapid detection of malachite green in fish. Food Chem 226:8–13. https://doi.org/10.1016/j.foodchem.2017.01.045
Song YQ, Liu SP, Liu ZF, Hu XL (2011) Resonance rayleigh scattering spectra, non-linear scattering spectra of malachite green-12-tungstophosphoric acid system and its analytical application in fish. Spectrochim Acta A 78:148–152. https://doi.org/10.1016/j.saa.2010.09.014
Song D, Yang R, Wang CW, Xiao R, Long F (2016) Reusable nanosilver-coated magnetic particles for ultrasensitive SERS-based detection of malachite green in water samples. Sci Rep 6:22870. https://doi.org/10.1038/srep22870
Van de Riet JM, Murphy CJ, Pearce JN, Potter RA, Burns BG (2005) Determination of malachite green and leucomalachite green in a variety of aquacultured products by liquid chromatography with tandem mass spectrometry detection. J AOAC Int 88:744–749
Wang X, Xu GH, Wei FD, Ma YS, Ma YJ, Song YY, Cen Y, Hu Q (2017) Highly sensitive and selective aptasensor for detection of adenosine based on fluorescence resonance energy transfer from carbon dots to nano-graphite. J Colloid Interface Sci 508:455–461. https://doi.org/10.1016/j.jcis.2017.07.028
Wu L, Lin ZZ, Zhong HP, Peng AH, Chen XM, Huang ZY (2017b) Rapid detection of malachite green in fish based on CdTe quantum dots coated with molecularly imprinted silica. Food Chem 299:847–853. https://doi.org/10.1016/j.foodchem.2017.02.144
Wu L, Lin ZZ, Zhong HP, Chen XM, Huang ZY (2017a) Rapid determination of malachite green in water and fish using a fluorescent probe based on CdTe quantum dots coated with molecularly imprinted polymer. Sensors Actuators B Chem 239:69–75. https://doi.org/10.1016/j.snb.2016.07.166
Xie J, Peng T, Chen DD, Zhang QJ, Wang GM, Wang X, Guo Q, Jiang F, Chen D, Deng J (2013) Determination of malachite green, crystal violet and their leuco-metabolites in fish by HPLC after immunoaffinity column clean-up. J Chromatogr B 913-914:123–128. https://doi.org/10.1016/j.jchromb.2012.12.002
Xu NN, Zhang Q, Guo W, Li QT, Xu J (2016) Au@PVP core-shell nanoparticles used as surface-enhanced raman spectroscopic substrate to detect malachite green. Chin J Anal Chem 44:1378–1384. https://doi.org/10.1016/S1872-2040(16)60956-8
Yi HC, Qu WY, Huang WS (2008) Electrochemical determination of malachite green using a multi-wall carbon nanotube modified glassy carbon electrode. Microchim Acta 160:291–296. https://doi.org/10.1007/s00604-007-0814-z
Yi KY (2017) A novel method for the quantitative determination of crystal violet utilizing YVO4:Eu3+ nanoparticles. Chem Lett 46:520–523. https://doi.org/10.1246/cl.161157
Zhang Y, Yang JY, Lei HT, Wang H, Xu ZL, Shen YD, Zeng DP, Sun YM (2015) Development of chemiluminescent enzyme immunoassay for the determination of malachite green in seafood. Food Agric Immunol 26:204–217. https://doi.org/10.1080/09540105.2014.884056
Funding
This work was supported by grants from the Ministry of Science and Technology of the People’s Republic of China (Project No. 2016YFD0400401), Tianjin Municipal Science and Technology Commission (Project No. 16PTSYJC00130), and the International Science and Technology Cooperation Program of China (Project No. 2014DFR30350).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
Yiyuan Chen declares that he has no conflict of interest. Minling Ding declares that she has no conflict of interest. Jiaqi Li declares that she has no conflict of interest. Wei Sheng declares that she has no conflict of interest. Bing Liu declares that she has no conflict of interest. Yan Zhang declares that she has no conflict of interest. Shuo Wang declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Chen, Y., Ding, M., Li, J. et al. Fluorescence Quenching Immunoaffinity Test Column with Quantum Dots as Fluorescence Donors for the Quick Detection of Malachite Green and Crystal Violet in Aquatic Products. Food Anal. Methods 11, 3362–3370 (2018). https://doi.org/10.1007/s12161-018-1312-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1312-0