Abstract
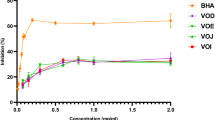
The results of GC-MS analysis are used to characterise the bioactive compounds in the infusion. The obtained results of the current research clearly revealed that the phenols, flavonoids and monomeric anthocyanins are abundant and a high-antioxidant activity of Pimpinella anisum L. infusions was detected. As expected, the studied plant’s infusion showed a stronger activity against free radicals than the enzymes that are attributed to generation of various chronic diseases. Among the characterised compounds, the fatty acids were detected with the highest quantity with 47.68% followed by triterpenoids and sterols, with 15.56 and 7.29%, respectively. Fatty acids (linoleic, oleic and palmitic acids), triterpenoids (lupeol, β-Amyrin and betulinic acids (BAs)) as well as sterols (β-sitosterol and stigmasterol) were detected as the main bioactive compounds in the studied infusion. In the current research, these compounds were detected to have substantial impacts on revealing of the potential health benefits of P. anisum L. seeds.



Similar content being viewed by others
References
Abidhusen HM, Acharya SS, Gajjar AV (2012) Corriandrum Sativum-review of advances in phytopharmacology. Int J Pharm Sci Res 3(5):1233–1239. https://doi.org/10.13040/IJPSR.0975-8232.3
Aspenstrom-Fagerlund B, Tallkvist J, Ilback NG, Glynn AW (2012) Oleic acid decreases BCRP mediated efflux of mitoxantrone in Caco-2 cell monolayers. Food Chem Toxicol 50(10):3635–3645. https://doi.org/10.1016/j.fct.2012.07.015
Barros L, Helano SA, Carvalho AM, Ferreira ICFR (2010) Lamiaceae often used in Portuguese folk medicine as a source of powerful antioxidants: vitamins and phenolics. LWT-Food Sci Technol 43:544–550. https://doi.org/10.1016/j.lwt.2009.09.024
Bedard K, Krause KH (2007) The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev 87(1):245–313. https://doi.org/10.1152/physrev.00044.2005
Benzie IF, Strain JJ (1999) Ferric reducing antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol 299:15–27. https://doi.org/10.1016/S0076-6879(99)99005-5.
Bergamo P, Luongo D, Miyamoto J, Cocca E, Kishino S, Ogawa J, Tanabe S, Rossi M (2014) Immunomodulatory activity of a gut microbial metabolite of dietary linoleic acid, 10-hydroxy-cis-12-octadecenoic acid, associated with improved antioxidant/detoxifying defences. J Funct Foods 11:192–202. https://doi.org/10.1016/j.jff.2014.10.007
Bradford PG, Awad AB (2007) Phytosterols as anticancer compounds. Mol Nutr Food Res 51(2):161–170. https://doi.org/10.1002/mnfr.200600164
Butterfield DA, Reed T, Newman SF, Sultana R (2007) Roles of amyloid beta-peptide-associated oxidative stress and brain protein modifications in the pathogenesis of Alzheimer’s disease and mild cognitive impairment. Free Radic Biol Med 43(5):658–677. https://doi.org/10.1016/j.freeradbiomed.2007.05.037
Camps P, El Achab R, Morral J, Munoz-Torrero D, Badia A, Banos JE, Vivas NM, Barril X, Orozco M, Luque FJ (2000) New tacrine-huperzine A hybrids (huprines): highly potent tight-binding acetylcholinesterase inhibitors of interest for the treatment of Alzheimer’s disease. J Med Chem 43(24):4657–4666. https://doi.org/10.1021/jm000980y
Colla ARS, Oliveira Á, Pazini FL, Rosa JM, Manosso LM, Cunha MP, Rodrigues AL (2014) Serotonergic and noradrenergic systems are implicated in the antidepressant-like effect of ursolic acid in mice. Pharmacol Biochem 124:108–116. https://doi.org/10.1016/j.pbb.2014.05.015
Conforti F, Statti G, Loizzo MR, Sacchetti G, Poli F, Menichini F (2005) In vitro antioxidant effect and inhibition of alpha-amylase of two varieties of Amaranthus caudatus seeds. Biol Pharm Bull 28(6):1098–1102. https://doi.org/10.1248/bpb.28.1098
Cui J, Huang L, Zhao A, Lew JL, Yu J, Sahoo S, Meinke PT, Royo I, Pelaez F, Wright SD (2003) Guggulsterone is a farnesoid X receptor antagonist in coactivator association assays but acts to enhance transcription of bile salt export pump. J Biol Chem 278(12):10214–10220. https://doi.org/10.1074/jbc.M209323200
Dolatabadi Z, Elhami Rad AH, Farzaneh V, Akhlaghi Feizabad SH, Estiri SH, Bakhshabadi H (2016) Modeling of the lycopene extraction from tomato pulps. Food Chem 190:968–973. https://doi.org/10.1016/j.foodchem.2015.06.069
Ellman GL, Courtney KD, Andres jr V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Farzaneh V, Carvalho IS (2015) A review of the health benefit potentials of herbal plant infusions and their mechanism of actions. Ind Crop Prod 65:247–258. https://doi.org/10.1016/j.indcrop.2014.10.057
Farzaneh V, Carvalho IS (2017) Modelling of microwave assisted extraction (MAE) of Anthocyanins (TMA). J Appl Res Med Aromat Plants 6:92–100. https://doi.org/10.1016/j.jarmap.2017.02.005
Farzaneh V, Ghodsvali A, Bakhshabadi H, Ganje M, Dolatabadi Z, Carvalho IS (2016) Modelling of the selected physical properties of the fava bean with various moisture contents using fuzzy logic design. J Food Process Eng. https://doi.org/10.1111/jfpe.12366
Farzaneh V, Bakhshabadi H, Hamid M, Gharekhani GM, Farzaneh F, Rashidzadeh S, Isabel SC (2017) Application of an adaptive neuro_fuzzy inference system (ANFIS) in the modelling of rapeseeds’ oil extraction. J Food Process Eng 40(6). https://doi.org/10.1111/jfpe.12562
Farzaneh V, Alireza G, Hamid B, Dolatabadi Z, Farzaneh F, Isabel SC, Khashayar S (2018) Screening of the alterations in qualitative characteristics of grape under the impacts of storage and harvest times using artificial neural network. Evol Syst 9(1):81–89. https://doi.org/10.1007/s12530-017-9212-x
Ghodsvali A, Farzaneh V, Bakhshabadi H, Zare Z, Karami Z, Mokhtarian M, Carvalho IS (2016) Screening of the aerodynamic and biophysical properties of barley malt. Int Agrophys 30. https://doi.org/10.1515/intag-2016-0017
Giusti MM, Wrolstad RE (2001) Characterization and measurement of anthocyanins by UV-visible spectroscopy. Current protocols in food analytical chemistry. John Wiley & Sons, Inc. doi: https://doi.org/10.1002/0471142913.faf0102s00
Huang YC, Chang YH, Shao YY (2006) Effects of genotype and treatment on the antioxidant activity of sweet potato in Taiwan. Food Chem 98(3):529–538. https://doi.org/10.1016/j.foodchem.2005.05.083
Jabrayili S, Farzaneh V, Zare Z, Bakhshabadi H, Babazadeh Z, Mokhtarian M, Carvalho IS (2016) Modelling of mass transfer kinetic in osmotic dehydration of kiwifruit. Int Agrophys 30(2):185–191. https://doi.org/10.1515/intag-2015-0091.
Khan AG, Kuek C, Chaudhry TM, Khoo CS, Hayes WJ (2000) Role of plants, mycorrhizae and phytochelators in heavy metal contaminated land remediation. Chemosphere 41(1–2):197–207. https://doi.org/10.1016/S0045-6535(99)00412-9.
Kim KT, Rioux LE, Turgeon SL (2014) Alpha-amylase and alpha-glucosidase inhibition is differentially modulated by fucoidan obtained from Fucus vesiculosus and Ascophyllum nodosum. Phytochemistry 98(0):27–33. https://doi.org/10.1016/j.phytochem.2013.12.003.
Lou-Bonafonte JM, Arnal C, Navarro MA, Osada J (2012) Efficacy of bioactive compounds from extra virgin olive oil to modulate atherosclerosis development. Mol Nutr Food Res 56(7):1043–1057. https://doi.org/10.1002/mnfr.201100668
Maedler K, Oberholzer J, Bucher P, Spinas GA, Donath MY (2003) Monounsaturated fatty acids prevent the deleterious effects of palmitate and high glucose on human pancreatic β-cell turnover and function. Diabetes 52(3):726–733. https://doi.org/10.2337/diabetes.52.3.726
Masso-Welch PA, Zangani D, Ip C, Vaughan MM, Shoemaker S, Ramirez RA, Ip MM (2002) Inhibition of angiogenesis by the cancer chemopreventive agent conjugated linoleic acid. Cancer Res 62(15):4383–4389
Nichenametla SN, Taruscio TG, Barney DL, Exon JH (2006) A review of the effects and mechanisms of polyphenolics in cancer. Crit Rev Food Sci Nutr 46(2):161–183. https://doi.org/10.1080/10408390591000541
Okada Y, Miyauchi N, Suzuki K, Kobayashi T, Tsutsui C, Mayuzumi K, Nishibe S, Okuyama T (1995) Search for naturally occurring substances to prevent the complications of diabetes. II. Inhibitory effect of coumarin and flavonoid derivatives on bovine lens aldose reductase and rabbit platelet aggregation. Chem Pharm Bull (Tokyo) 43(8):1385–1387
Ortiz-Andrade RR, Garcia-Jimenez S, Castillo-Espana P, Ramirez-Avila G, Villalobos-Molina R, Estrada-Soto S (2007) Alpha-glucosidase inhibitory activity of the methanolic extract from Tournefortia hartwegiana: an anti-hyperglycemic agent. J Ethnopharmacol 109(1):48–53. https://doi.org/10.1016/j.jep.2006.07.002
Otuki MF, Ferreira J, Lima FV, Meyre-Silva C, Malheiros A, Muller LA, Cani GS, Santos AR, Yunes RA, Calixto JB (2005) Antinociceptive properties of mixture of alpha-amyrin and beta-amyrin triterpenes: evidence for participation of protein kinase C and protein kinase A pathways. J Pharmacol Exp Ther 313(1):310–318. https://doi.org/10.1124/jpet.104.071779.
Özcan M (2004) Mineral contents of some plants used as condiments in Turkey. Food Chemistry 84:437–440. https://doi.org/10.1016/S0308-8146(03)00263-2
Özcan MM, Chalchat CJ (2006) Chemical composition and antifungal effect of anise (Pimpinella anisum L.) fruit oil at ripening stage. Ann Microbiol 56:353–358
Pathak R, Pathak A (2012) Study of life style habits on risk of type 2 diabetes. Int J Appl Basic Med Res 2(2):92–96. https://doi.org/10.4103/2229-516X.106349
Preetha SP, Kanniappan M, Selvakumar E, Nagaraj M, Varalakshmi P (2006) Lupeol ameliorates aflatoxin B1-induced peroxidative hepatic damage in rats. Comp Biochem Physiol C Toxicol Pharmacol 143(3):333–339. https://doi.org/10.1016/j.cbpc.2006.03.008
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269(2):337–341. https://doi.org/10.1006/abio.1999.4019
Quettier-Deleu C, Gressier B, Vasseur J, Dine T, Brunet C, Luyckx M, Cazin M, Cazin JC, Bailleul F, Trotin F (2000) Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J Ethnopharmacol 72(1–2):35–42. https://doi.org/10.1016/S0378-8741(00)00196-3.
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Bio Med 26(9–10):1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Rodrigues VM, Rosa PTV, Marques MOM, Petenate AJ, Meireles MAA (2003) Supercritical extraction of essential oil from Pimpinella anisum led (Pimpinella anisum L.) using CO2: solubility, kinetics, and composition data. J Agric Food Chem 51(6):1518–1523. https://doi.org/10.1021/jf0257493
Rostami M, Farzaneh V, Boujmehrani A, Mohammadi M, Bakhshabadi H (2014) Optimizing the extraction process of sesame seed's oil using response surface method on the industrial scale. Ind Crop Prod 58:160–165. https://doi.org/10.1016/j.indcrop.2014.04.015
Sancheti S, Um BH, Seo SY (2010) 1,2,3,4,6-penta-O-galloyl-β-d-glucose: a cholinesterase inhibitor from Terminalia chebula. S Afr J Bot 76(2):285–288. https://doi.org/10.1016/j.sajb.2009.11.006
Sancheti S, Sancheti S, Seo SY (2013) Antidiabetic and antiacetylcholinesterase effects of ethyl acetate fraction of Chaenomeles sinensis (Thouin) Koehne fruits in streptozotocin-induced diabetic rats. Exp Toxicol Pathol 65(1–2):55–60. https://doi.org/10.1016/j.etp.2011.05.010
Shobha R, Rajeshwari C, Andallu B (2013) Anti-peroxidative and anti-diabetic activities of aniseeds (Pimpinella anisum l.) and identification of bioactive compounds. Am J Phytomed Clin Ther 1(5):516–527. https://doi.org/10.1016/b978-0-12-375688-6.10020-9
Singh AB, Yadav DK, Maurya R, Srivastava AK (2009) Antihyperglycaemic activity of alpha-amyrin acetate in rats and db/db mice. Nat Prod Res 23(9):876–882. https://doi.org/10.1080/14786410802420416
Skujins S 1988 Handbook for ICP-AES (Varian-Vista). A short guide to Vista Series ICP-AES operation. Version 1.0, Varian Int. AG, Zug.
Sudhahar V, Ashokkumar S, Varalakshmi P (2006a) Effect of lupeol and lupeol linoleate on lipemic--hepatocellular aberrations in rats fed a high cholesterol diet. Mol Nutr Food Res 50(12):1212–1219. https://doi.org/10.1002/mnfr.200600134
Sudhahar V, Kumar SA, Varalakshmi P (2006b) Role of lupeol and lupeol linoleate on lipemic-oxidative stress in experimental hypercholesterolemia. Life Sci 78(12):1329–1335. https://doi.org/10.1016/j.lfs.2005.07.011
Sudhahar V, Kumar SA, Mythili Y, Varalakshmi P (2007a) Remedial effect of lupeol and its ester derivative on hypercholesterolemia-induced oxidative and inflammatory stresses. Nutr Res 27(12):778–787. https://doi.org/10.1016/j.nutres.2007.09.012
Sudhahar V, Kumar SA, Sudharsan PT, Varalakshmi P (2007b) Protective effect of lupeol and its ester on cardiac abnormalities in experimental hypercholesterolemia. Vasc Pharmacol 46(6):412–418. https://doi.org/10.1016/j.vph.2006.12.005
Trayhurn P, Wood IS (2004) Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br J Nutr 92(3):347–355. https://doi.org/10.1079/BJN20041213
Yen GC, Duh P, Chuang D (2000) Antioxidant activity of anthraquinones and anthrone. Food Chem 70(4):437–441. https://doi.org/10.1016/S0308-8146(00)00108-4
Yuk JE, Woo JS, Yun C-Y, Lee J-S, Kim J-H, Song G-Y, Yang EJ, Hur IK, Kim IS (2007) Effects of lactose-β-sitosterol and β-sitosterol on ovalbumin-induced lung inflammation in actively sensitized mice. Int Immunopharmacol 7(12):1517–1527. https://doi.org/10.1016/j.intimp.2007.07.026
Acknowledgments
This work is a part of Ph.D. study of Vahid Farzaneh. The authors greatly acknowledge the Food Science Lab (FSL), Faculty of Science and Technology, University of Algarve for providing the necessary facilities during this study period.
Funding Source
The authors would like to acknowledge Erasmus Mundus program SALAM-2013 for the financial support during this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Vahid Farzaneh has received research grants from Erasmus Mundus program SALAM-2013. Jorge Gominho declares that he has no conflict of interest. Helena Pereira declares that she has no conflict of interest. Isabel S. Carvalho declares that she has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Farzaneh, V., Gominho, J., Pereira, H. et al. Screening of the Antioxidant and Enzyme Inhibition Potentials of Portuguese Pimpinella anisum L. Seeds by GC-MS. Food Anal. Methods 11, 2645–2656 (2018). https://doi.org/10.1007/s12161-018-1250-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-018-1250-x