Abstract
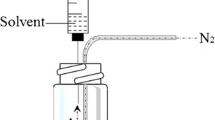
In this study, a green, simple, and sensitive method was developed for the analysis of aliphatic aldehydes from fried meat by using a modified gas purge–microsyringe extraction (GP–MSE) system in combination with high-performance liquid chromatography (HPLC) with fluorescence detection. The modified GP–MSE system possessed two gas channels and showed better recoveries for compounds with diverse density in comparison with one gas channel GP–MSE system. Target compounds in fried meat were effectively extracted without the traditional solvent extraction and lipid removing process, while the HPLC sensitivity of aldehydes was enhanced by introducing 2-(12-benzo[b]acridin-5(12H)-yl)-acetohydrazide (BAAH) with excellent fluorescence property into the molecules. Parameters influencing the extraction efficiency and HPLC sensitivity were optimized. The limits of detection (LODs) ranged from 0.30 to 0.45 μg/kg, and the limits of quantification (LOQs) ranged from 1.0 to 1.5 μg/kg. The recoveries of the target compounds were in the range of 86.9 to 95.6%. The proposed method was successfully applied to the analysis of aldehydes in fried meat samples. Formaldehyde, acetaldehyde, pentanal, hexanal, heptanal, octanal, nonaldehyde, and decanal were all found in fried meat samples with concentrations ranging from 0.05 to 17.8 mg/kg.



Similar content being viewed by others
References
Al Rashidi M, Shihadeh A, Saliba NA (2008) Volatile aldehydes in the mainstream smoke of the narghile waterpipe. Food Chem Toxicol 46(11):3546–3549. https://doi.org/10.1016/j.fct.2008.09.007
Calejo I, Moreira N, Araújo AM, Carvalho M, de Lourdes Bastos M, de Pinho PG (2016) Optimisation and validation of a HS-SPME–GC–IT/MS method for analysis of carbonyl volatile compounds as biomarkers in human urine: application in a pilot study to discriminate individuals with smoking habits. Talanta 148:486–493. https://doi.org/10.1016/j.talanta.2015.09.070
Domínguez R, Gómez M, FonsecaS LJM (2014) Influence of thermal treatment on formation of volatile compounds, cooking loss and lipid oxidation in foal meat. LWT-Food Sci Technol 58(2):439–445. https://doi.org/10.1016/j.lwt.2014.04.006
Dominguez R, Gomez M, Fonseca S, Lorenzo JM (2014) Effect of different cooking methods on lipid oxidation and formation of volatile compounds in foal meat. Meat Sci 97(2):223–230. https://doi.org/10.1016/j.meatsci.2014.01.023
Environmental Protection Agency (US), Integrated Risk Information System on Formaldehyde, National Center for Environmental Assessment, Office of Research and Development, Washington, DC, 1999
Fenech M, Nersesyan A, Knasmueller S (2016) A systematic review of the association between occupational exposure to formaldehyde and effects on chromosomal DNA damage measured using the cytokinesis-block micronucleus assay in lymphocytes. Mutat Res 770(Pt A):46–57
Jerković I, Mastelić J, Tartaglia S (2007) A study of volatile flavour substances in Dalmatian traditional smoked ham: impact of dry-curing and frying. Food Chem 104(3):1030–1039. https://doi.org/10.1016/j.foodchem.2007.01.013
Li X, Xue A, Chen H, Li S (2013) Low-density solvent-based dispersive liquid-liquid microextraction combined with single-drop microextraction for the fast determination of chlorophenols in environmental water samples by high performance liquid chromatography-ultraviolet detection. J Chromatogr A 1280:9–15. https://doi.org/10.1016/j.chroma.2013.01.023
Ma C, Ji J, Tan C, Chen D, Luo F, Wang Y, Chen X (2014) Headspace solid-phase microextraction coupled to gas chromatography for the analysis of aldehydes in edible oils. Talanta 120:94–99. https://doi.org/10.1016/j.talanta.2013.11.021
Malekpour A, Ahmadi N (2017) Surfactant-alumina-coated magnetic nanoparticles as an efficient aldehydes adsorbent prior their determination by HPLC. Food Anal Methods 10(6):1817–1825. https://doi.org/10.1007/s12161-016-0728-7
Mashayekhi HA, Khalilian F (2016) Development of solid-phase extraction coupled with dispersive liquid-liquid microextraction method for the simultaneous determination of three benzodiazepines in human urine and plasma. J Chromatogr Sci 54(6):1068–1073. https://doi.org/10.1093/chromsci/bmw031
Nieto G, Bañón S, Garrido MD (2011) Effect of supplementing ewes’ diet with thyme (Thymus zygis ssp. gracilis) leaves on the lipid oxidation of cooked lamb meat. Food Chem 125(4):1147–1152. https://doi.org/10.1016/j.foodchem.2010.09.090
Osorio VM, de Lourdes Cardeal Z (2011) Determination of acrolein in french fries by solid-phase microextraction gas chromatography and mass spectrometry. J Chromatogr A 1218(21):3332–3336. https://doi.org/10.1016/j.chroma.2010.11.068
Peng C-Y, Lang C-H, Lin P-C, Kuo Y-C (2017) Effects of cooking method, cooking oil, and food type on aldehyde emissions in cooking oil fumes. J Hazard Mater 324(Pt B):160–167. https://doi.org/10.1016/j.jhazmat.2016.10.045
Rahmani M, Kaykhaii M, Ghasemi E, Tahernejad M (2015) Application of in-syringe dispersive liquid-liquid microextraction and narrow-bore tube dispersive liquid-liquid microextraction for the determination of trace amounts of BTEX in water samples. J Chromatogr Sci 53(7):1210–1216. https://doi.org/10.1093/chromsci/bmu163
Ramiärez MR, Esteä M, Morcuende D, Cava R (2004) Effect of the type of frying culinary fat on volatile compounds isolated in fried pork loin chops by using SPME-GC-MS. J Agric Food Chem 52(25):7637–7643. https://doi.org/10.1021/jf049207s
Saieva C, Peluso M, Palli D, Cellai F, Ceroti M, Selvi V, Bendinelli B, Assedi M, Munnia A, Masala G (2016) Dietary and lifestyle determinants of malondialdehyde DNA adducts in a representative sample of the Florence City population. Mutagenesis 31(4):475–480. https://doi.org/10.1093/mutage/gew012
Sheng C, Zhang S, Wang F, Ji Z, Dou K, You J (2016) Determination of aliphatic amines in infant food packaging materials by high performance liquid chromatography. Chinese J Anal Chem 44:1365–1371
Tsuruya A, Kuwahara A, Saito Y, Yamaguchi H, Tenma N, Inai M, Takahashi S, Tsutsumi E, Suwa Y, Totsuka Y, Suda W, Oshima K, Hattori M, Mizukami T, Yokoyama A, Shimoyama T, Nakayama T (2016) Major anaerobic bacteria responsible for the production of carcinogenic acetaldehyde from ethanol in the colon and rectum. Alcohol Alcoholism 51(4):395–401. https://doi.org/10.1093/alcalc/agv135
Varlet V, Prost C, Serot T (2007) Volatile aldehydes in smoked fish: analysis methods, occurrence and mechanisms of formation. Food Chem 105(4):1536–1556. https://doi.org/10.1016/j.foodchem.2007.03.041
Yang C, Piao X, Qiu J, Wang X, Ren C, Li D (2011) Gas purge microsyringe extraction for quantitative direct gas chromatographic-mass spectrometric analysis of volatile and semivolatile chemicals. J Chromatogr A 1218(12):1549–1555. https://doi.org/10.1016/j.chroma.2011.01.018
Yang C, Qiu J, Ren C, Piao X, Li X, Wu X, Li D (2009) Gas flow headspace liquid phase microextraction. J Chromatogr A 1216(45):7694–7699. https://doi.org/10.1016/j.chroma.2009.09.039
Yang C, Zhao J, Wang J, Yu H, Piao X, Li D (2013) Water-based gas purge microsyringe extraction coupled with liquid chromatography for determination of alkylphenols from sea food Laminaria japonica Aresh. J Chromatogr A 1300:38–42. https://doi.org/10.1016/j.chroma.2013.02.087
Zamora R, Aguilar I, Granvogl M, Hidalgo FJ (2016) Toxicologically relevant aldehydes produced during the frying process are trapped by food phenolics. J Agric Food Chem 64(27):5583–5589. https://doi.org/10.1021/acs.jafc.6b02165
Zhang Q, Saleh AS, Chen J, Shen Q (2012) Chemical alterations taken place during deep-fat frying based on certain reaction products: a review. Chemistry Phys Lipids 165(6):662–681. https://doi.org/10.1016/j.chemphyslip.2012.07.002
Zhang S, Li C, Zhou G, Che G, You J, Suo Y (2013) Determination of the carbohydrates from Notopterygium forbesii Boiss by HPLC with fluorescence detection. Carbohyd polym 97(2):794–799. https://doi.org/10.1016/j.carbpol.2013.05.041
Zhang S, Sheng C, Zhang J, Li Y, You J (2017) Gas purge microsyringe extraction coupled with dispersive liquid-liquid microextraction for the determination of acidic compounds in food packaging materials. Food Anal Methods 10(5):1164–1171. https://doi.org/10.1007/s12161-016-0660-x
Zhang Y, Lee HK (2013) Liquid phase microextraction using knitting wool as the extractant phase holder before chromatographic analysis: a new approach for trace analysis. J Chromatogr A 1273:12–17. https://doi.org/10.1016/j.chroma.2012.11.084
Acknowledgements
The work was supported by the Natural Science Foundation of Shandong Province (ZR2014BQ009, ZR2015CM040), the National Undergraduate Training Program for Innovation and Entrepreneurship of China (No. 201710446036), and the National Science Foundation of China (No. 21275089).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
Yanxin Li declares that she has no conflict of interest. Shijuan Zhang declares that she has no conflict of interest. Fengli Lu declares that she has no conflict of interest. Qian Liu declares that he has no conflict of interest. Jinmao You declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Li, Y., Zhang, S., Lu, F. et al. Gas Purge–Microsyringe Extraction Coupled with Liquid Chromatography and Fluorescence Detection for the Determination of Aldehydes from Fried Meat. Food Anal. Methods 11, 1390–1397 (2018). https://doi.org/10.1007/s12161-017-1121-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-017-1121-x