Abstract
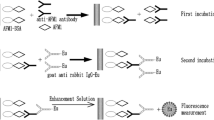
Streptomycin (STR), used extensively in the treatment of bovine mastitis, may cause damage such as ototoxicity, allergic reaction, and increasing bacterial resistance to consumers on account of remnant in milk. A time-resolved fluoroimmunoassay (TRFIA) was developed to quantify STR for the first time to ensure food safety. Using secondary antibody labeled with europium (Eu3+) chelate as a tracer, the proposed TRFIA showed that the linear working range was 0.32–5.0 ng/mL under the optimal conditions. Milk samples were deproteinized by trichloroacetic acid and the limit of detection of STR in milk was 1.8 μg/kg. The recoveries of milk samples fortified with 4.0, 20, and 40 μg/kg of STR ranged from 86.2 to 96.3% with relative standard deviations less than 11%. Results of TRFIA for the authentic samples were coincided with those of UHPLC-MS/MS analyses. This study confirmed that the established TRFIA was sensitive as well as reliable and could be an alternative method to monitor STR residue in milk.






Similar content being viewed by others
References
Aksu H, Cetin O, Arun O, Ergun O (2004) Determination of tetracyclin and streptomycin residues by means of ELISA in pasteurized and UHT-sterilized milk. Med Weter 60:1171–1173
Arsand JB, Jank L, Martins MT, Hoff RB, Barreto F, Pizzolato TM, Sirtori C (2016) Determination of aminoglycoside residues in milk and muscle based on a simple and fast extraction procedure followed by liquid chromatography coupled to tandem mass spectrometry and time of flight mass spectrometry. Talanta 154:38–45. doi:10.1016/j.talanta.2016.03.045
Byzova NA, Zvereva EA, Zherdev AV, Eremin SA, Sveshnikov PG, Dzantiev BB (2011) Pretreatment-free immunochromatographic assay for the detection of streptomycin and its application to the control of milk and dairy products. Anal Chim Acta 701:209–217. doi:10.1016/j.aca.2011.06.001
Commission CA (2014) Maximum residue limits for veterinary drugs in foods. Updated as at the 35th session of the Codex Alimentarius Commission (July 2012) CAC/MRL:2–2012
de Oliveira RC, Paschoal JAR, Sismotto M, Airoldi FPD, Reyes FGR (2009) Development and validation of an LC-APCI-MS-MS analytical method for the determination of streptomycin and dihydrostreptomycin residues in milk. J Chromatogr Sci 47:756–761
Deng A et al (2003) Residue analysis of the pharmaceutical diclofenac in different water types using ELISA and GC-MS. Environ Sci Technol 37:3422–3429
European Commission (2002) Commission decision (2002)/657/EC of 12 August (2002) implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. Off J Eur Comm L 221:8–28
European Commission (2011) Action plan against the rising threats from antimicrobial resistance. http://ec.europa.eu/dgs/health consumer/docs/communication amr 2011 748 en.pdf. Accessed 16 January 2012
Furusawa N (2001) Determining the procedure for routine residue monitoring of sulfamethazine in edible animal tissues. Biomed Chromatogr 15:235–239. doi:10.1002/bmc.48
Gaudin V, Hedou C, Verdon E (2013) Validation of two ELISA kits for the screening of tylosin and streptomycin in honey according to the European decision 2002/657/EC. Food Addit Contam A 30:93–109. doi:10.1080/19440049.2012.722696
Granja R, Nino AMM, Zucchetti RAM, Nino REM, Patel R, Salerno AG (2009) Determination of streptomycin residues in honey by liquid chromatography-tandem mass spectrometry. Anal Chim Acta 637:64–67. doi:10.1016/j.aca.2009.01.006
Gremilogianni AM, Megoulas NC, Koupparis MA (2010) Hydrophilic interaction vs ion pair liquid chromatography for the determination of streptomycin and dihydrostreptomycin residues in milk based on mass spectrometric detection. J Chromatogr A 1217:6646–6651. doi:10.1016/j.chroma.2010.04.059
Haasnoot W, Bienenmann-Ploum M, Kohen F (2003) Biosensor immunoassay for the detection of eight sulfonamides in chicken serum. Anal Chim Acta 483:171–180. doi:10.1016/s0003-2670(02)01545-3
Jiang WX, Wang ZH, Nolke G, Zhang J, Niu LL, Shen JZ (2013) Simultaneous determination of aflatoxin B-1 and aflatoxin M-1 in food matrices by enzyme-linked immunosorbent assay. Food Anal Methods 6:767–774. doi:10.1007/s12161-012-9484-5
Kolosova AY, Shim WB, Yang ZY, Eremin SA, Chung DH (2006) Direct competitive ELISA based on a monoclonal antibody for detection of aflatoxin B-1. Stabilization of ELISA kit components and application to grain samples. Analytical Anal Bioanal Chem 384:286–294
Le T, Yan PF, Liu J, Wei S (2013) Simultaneous detection of sulfamethazine and sulfaquinoxaline using a dual-label time-resolved fluorescence immunoassay. Food Addit Contam A 30:1264–1269. doi:10.1080/19440049.2013.801084
Liang QN, Chen PQ, Liu TC, Zhou JW, Chen JJ, Wu YS (2015) Development of a time-resolved fluoroimmunoassay for Epstein-Barr virus viral capsid antigen IgA antibody in human serum. J Virol Methods 222:16–21. doi:10.1016/j.jviromet.2015.03.024
Lin GF, Liu TC, Zou LP, Hou JY, Wu YS (2013) Development of a dual-label time-resolved fluoroimmunoassay for the detection of α-fetoprotein and hepatitis B virus surface antigen. Luminescence 28:401–406
Mishra GK, Sharma A, Bhand S (2015) Ultrasensitive detection of streptomycin using flow injection analysis-electrochemical quartz crystal nanobalance (FIA-EQCN) biosensor. Biosens Bioelectron 67:532–539. doi:10.1016/j.bios.2014.09.033
MOA (2002) No. 235 Bulletin of the Ministry of Agriculture of the People’s Republic of China. Available at http://www.moa.gov.cn/zwllm/tzgg/gg/200302/t20030226_59300.htm Accessed 28.09.12
NHFPC (2016) National action plan on antimicrobial resistance 2016 to 2020. National Health and Family Planning Commission of China, available at http://www.gov.cn/xinwen/2016-08/25/content_5102348.htm
Organization WH (2015) Global action plan on antimicrobial resistance
Peippo P, Lovgren T, Tuomola M (2005) Rapid screening of narasin residues in poultry plasma by time-resolved fluoroimmunoassay. Anal Chim Acta 529:27–31. doi:10.1016/j.aca.2004.07.013
Regulation C (2010) No. 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin. Off J Eur Union L 15:1–72
Reverberi R, Reverberi L (2007) Factors affecting the antigen-antibody reaction. Blood Transfus 5:227
Samsonova JV, Bashkurov ML, Ivanova NL, Rubtsova MY, Egorov AM (2005) ELISA of streptomycin in buffer and milk: effect of reagents’ structure and analysis format on assay performance. Food Agr Immunol 16:47–57. doi:10.1080/09540100400029944
Shen JZ, Zhang Z, Yao Y, Shi WM, Liu YB, Zhang SX (2007) Time-resolved fluoroimmunoassay for ractopamine in swine tissue. Anal Bioanal Chem 387:1561–1564. doi:10.1007/s00216-006-1063-4
Sheng W, Yang L, Wang JP, Zhang Y, Wang S (2013) Development of an enzyme-linked immunosorbent assay for the detection of gentamycin residues in animal-derived foods. LWT-Food Sci Technol 50:204–209. doi:10.1016/j.lwt.2012.05.028
Shi HY, Sheng EZ, Feng L, Zhou LL, Hua XD, Wang MH (2015) Simultaneous detection of imidacloprid and parathion by the dual-labeled time-resolved fluoroimmunoassay. Environ Sci Pollut R 22:14882–14890. doi:10.1007/s11356-015-4697-y
Taghdisi SM, Danesh NM, Nameghi MA, Ramezani M, Abnous K (2016) A label-free fluorescent aptasensor for selective and sensitive detection of streptomycin in milk and blood serum. Food Chem 203:145–149. doi:10.1016/j.foodchem.2016.02.017
Unusan N (2009) Occurrence of chloramphenicol, streptomycin and tetracycline residues in ultra-heat-treatment milk marketed in Turkey. Int Food Sci Nutr 60:359–364. doi:10.1080/09637480701664555
van Bruijnsvoort M, Ottink SJM, Jonker KM, de Boer E (2004) Determination of streptomycin and dihydrostreptomycin in milk and honey by liquid chromatography with tandem mass spectrometry. J Chromatogr A 1058:137–142. doi:10.1016/j.chroma.2004.07.101
Vinas P, Balsalobre N, Hernandez-Cordoba M (2007) Liquid chromatography on an amide stationary phase with post-column derivatization and fluorimetric detection for the determination of streptomycin and dihydrostreptomycin in foods. Talanta 72:808–812. doi:10.1016/j.talanta.2006.12.006
Wan GH, Cui H, Zheng HS, Pang YQ, Liu LJ, Yu XF (2006) Flow-injection determination of streptomycin residues in milk using the luminol-periodate-Mn2+ chemiluminescence system. Luminescence 21:36–42. doi:10.1002/bio.880
Wang JP, Zhang HY, Sheng W, Liu W, Zheng LL, Zhang XZ, Wang S (2013) Determination of streptomycin residues in animal-derived foods by a reliable and accurate enzyme-linked immunosorbent assay. Anal Methods 5:4430–4435. doi:10.1039/c3ay40131f
Wei S, Le T, Chen Y, Xu J, He HQ, Niu XD, Luo JH (2013) Time-resolved fluoroimmunoassay for quantitative determination of tylosin and tilmicosin in edible animal tissues. Chin Sci Bull 58:1838–1842. doi:10.1007/s11434-013-5749-7
Wu JX, Zhang SE, Zhou XP (2010) Monoclonal antibody-based ELISA and colloidal gold-based immunochromatographic assay for streptomycin residue detection in milk and swine urine. J Zhejiang Univ Sci 11:52–60. doi:10.1631/jzus.B0900215
Zhang J, Guo J-Z, Xiao H-L, Zhu L, Liu H-Y, Zhang Y, Huang B (2010) Simultaneous detection of different serum pepsinogens and its primary application. World J Gastroenterol 16:3072–3077. doi:10.1007/s11356-015-4697-y
Zhou B, Zhang J, Fan J, Zhu L, Zhang Y, Jin J, Huang BA (2015) A new sensitive method for the detection of chloramphenicol in food using time-resolved fluoroimmunoassay. European Eur Food Res Technol 240:619–625. doi:10.1007/s00217-014-2363-0
Acknowledgements
This study was supported financially by grants from the Ministry of Science and Technology of China (2015BAK36B03). The authors would like to thank colleagues at the Department of Veterinary Pharmacology & Toxicology, College of Veterinary Medicine of China Agricultural University, for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was supported financially by grants from the Ministry of Science and Technology of China (2015BAK36B03).
Conflict of Interest
Yuanze Sun declares that she has no conflict of interest. Jie Xie declares that she has no conflict of interest. Tao Peng declares that he has no conflict of interest. Jianyi Wang declares that he has no conflict of interest. Sanlei Xie declares that he has no conflict of interest. Kai Yao declares that he has no conflict of interest. Cheng Wang declares that he has no conflict of interest. Shujuan Sun declares that she has no conflict of interest. Xi Xia declares that he has no conflict of interest. Haiyang Jiang declares that he has no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Rights and permissions
About this article
Cite this article
Sun, Y., Xie, J., Peng, T. et al. A New Method Based on Time-Resolved Fluoroimmunoassay for the Detection of Streptomycin in Milk. Food Anal. Methods 10, 2262–2269 (2017). https://doi.org/10.1007/s12161-017-0797-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-017-0797-2