Abstract
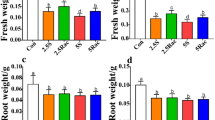
The enantioselective phytotoxicity of chiral pesticides is a new area of research in phyto-biochemical studies. Glufosinate (GL) is a chiral herbicide, comprised of the two enantiomers. The aim of this study was to investigate the enantioselective phytotoxicity of glufosinate ammonium enantiomers on root and shoot dry weight as well as catechin biosynthesis of wheat seedlings. Separation of GL enantiomers was achieved by using high-performance liquid chromatography on Eurocel 01 column. The optical rotation of separated enantiomers confirmed the purity of each isomer. The I50 parameter, which is the concentration resulting in a 50 % inhibition in root and shoot dry weight estimated using dose–response curves, showed that (+)-GL was 4.2 times more active than the (−)-GL enantiomer. It demonstrated that the effect of enantioselectivity of GL on catechin biosynthesis in wheat shoots was sensible, while the effect of enantioselectivity of GL in catechin content in wheat roots was not distinct.







Similar content being viewed by others
References
Ali I, Aboul-Enein HY, Ghanem A (2005) Enantioselective toxicity and carcinogenesis. Curr Pharm Anal 1:109–125
Aliferis KA (2011) Metabolomics—a robust bioanalytical approach for the discovery of the modes-of-action of pesticides: a review. Pestic Biochem Physiol 100:105–117
Contardo Jara V, Wiegand C (2008) Biotransformation and antioxidant enzymes of Lumbriculus variegates as biomarkers of contaminated sediment exposure. Chemosphere 70:1879–1888
Dewick PM (2002) Medicinal natural products: a biosynthetic approach, 2nd edn. Wiley, Chennai
Diao J, Xu P, Wang P, Lu D, Lu Y, Zhou Z (2010) Enantioselective degradation in sediment and aquatic toxicity to Daphnia magna of the herbicide lactofen enantiomers. J Agric Food Chem 58:2439–2445
Dinehart SK, Smith LM, McMurry ST, Anderson TA, Smith PN, Haukos DA (2009) Toxicity of a glufosinate- and several glyphosate-based herbicides to juvenile amphibians from the Southern High Plains USA. Sci Total Environ 407:1065–1071
Ghassempour A, Mollayi S, Farzaneh M, Sharifi-Tehrani A, Aboul-Enein HY (2011) Variation of catechin, epicatechin and their enantiomers concentrations before and after wheat cultivar-Puccinia triticina infection. Food Chem 125:1287–1290
Inderjit JL, Pollock RM, Callaway WH (2008) Phytotoxic effects of (+/−)-catechin in vitro, in soil, and in the field. PLoS One 3:1–11
Jing Y, Zhao M, Liu J, Liu W (2010) Enantioselectivity in environmental risk assessment of modern chiral pesticides. Environ Pollut 158:2371–2383
Kajdzanoska M, Gjamovski V, Stefova M (2010) HPLC-DAD-ESI-MSn identification of phenolic compounds in cultivated strawberries from Macedonia. Macedonian J Chem Chem Eng 29:181–194
Lacuesta M, Munoz Rueda A, Gonzalez-Murua C, Sivak MN (1992) Effect of phosphinothricin(glufosinate) on photosynthesis and chlorophyll fluorescence emission by barley leaves illuminated under photorespiratory and non-photorespiratory conditions. J Exp Bot 2:43
Liu H, Zhao M, Zhang C, Ma Y, Liu W (2008) Enantioselective cytotoxicity of the insecticide bifenthrin on a human amnion epithelial (FL) cell line. Toxicol Appl Pharmacol 253:89–96
Liu W, Lin K, Gan J (2006) Separation and aquatic toxicity of enantiomers of the organophosphorus insecticide trichloronate. Chirality 18:713–716
Muller BZ, Zumdick A, Schuphan I, Schmidt B (2001) Metabolism of the herbicide glufosinate ammonium in plant cell cultures of transgenic (rhizomania-resistant) and non-transgenic sugarbeet (Beta vulgaris), carrot (Daucus carota), purple foxglove (Digitalis purpurea) and thorn apple (Datura stramonium). Pest Manag Sci 57:46–56
Pizzutti IR, Andre K, Hiemstra M, Wickert C, Prestes OD (2009) Method validation and comparison of acetonitrile and acetone extraction for the analysis of 169 pesticides in soya grain by liquid chromatography–tandem mass spectrometry. J Chrom A 1216:4539–4552
Qian HF, Hu HJ, Mao YY, Ma J, Zhang AP, Liu WP, Fu ZW (2009) Enantioselective phytotoxicity of the herbicide imazethapyr in rice. Chemosphere 76:885–892
Qin S, Budd R, Bondarenko S, Liu W, Gan J (2006) Enantioselective degradation and chiral stability of pyrethroids in soil and sediment. J Agric Food Chem 54:5040–5045
Ramezani MK, Oliver DP, Kookana RS, Lao W, Gill GG, Preston C (2010) Faster degradation of herbicidally-active enantiomer of imidazolinones in soil. Chemosphere 79:1040–1045
Ruhland M, Engelhardt G, Pawlizki K (2002) A comparative investigation of the metabolism of the herbicide glufosinate in cell cultures of transgenic glufosinate-resistant and non-transgenic oilseed rape (Brassica napus) and corn (Zea mays). Environ Biosafty Res 1:29–37
Ruhland M, Engelhardt G, Pawlizki K (2004) Distribution and metabolism of d/l-, l-and d-glufosinate in transgenic, glufosinate-tolerant crops of maize (Zea mays L ssp. mays) and oilseed rape (Brassica napus L var napus). Pest Manag Sci 60:691–696
Seefeldt SS, Jensen JE, Fuerst EP (1995) Log-logistic analysis of herbicide dose–response relationships. Weed Technol 9:218–227
Song NH, Yin XL, Chen GF, Yang H (2007) Biological responses of wheat (Triticum aestivum) plants to the herbicide chlorotoluron in soils. Chemosphere 68:1779–1787
Subramanian G (2007) Chiral separation techniques: a practical approach. Wiley, Weinheim
Xu C, Tu W, Lou C, Hong Y, Zhao M (2009) Enantioselective separation and zebrafish embryo toxicity of insecticide beta-cypermethrin. J Environ Sci 22:738–743
Conflict of Interest
Mohammad Khodadady declares that she has no conflict of interest. Mohammad Kazem Ramezani declares that he has no conflict of interest. Vehideh Mahdavi declares that she has no conflict of interest. Alireza Ghassempour declares that she has no conflict of interest. Hassan Y. Aboul-Enein declares that he has no conflict of interest. This article does not contain any studies with human or animal subjects.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Khodadady, M., Ramezani, M.K., Mahdavi, V. et al. Enantioseparation and Enantioselective Phytotoxicity of Glufosinate Ammonium on Catechin Biosynthesis in Wheat. Food Anal. Methods 7, 747–753 (2014). https://doi.org/10.1007/s12161-013-9677-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-013-9677-6