Abstract
Nutrients in dairy wastewaters can be remediated through assimilation into algal biomass. Anaerobically digested manure creates an effluent (ADE) that is useful for algal cultivation while alternate processing of manure through a polyhydroxyalkanoate reactor generates a distinct effluent (PHAE), not previously characterized for algal cultivation. Each effluent was evaluated for growth rate, biomass production, and nutrient recovery using type algae species Chlorella vulgaris. Growth rates were elevated in 5, 10, and 20 % dilutions of PHAE (0.59, 0.53, 0.42 days−1) compared to equal concentrations of ADE (0.40, 0.36, 0.37 days−1). In addition, the growth phase lasted up to twice as long for PHAE, resulting in a fourfold higher stationary phase algal concentration (cells∙mL−1) compared to ADE. Growth in ADE was limited by specific inhibitory properties: high concentrations of dissolved organic matter, ammonia, and elevated bacterial load. Maximum nutrient removal rates for ADE and PHAE were 0.95 and 3.46 mg·L−1·day−1 for nitrogen and 0.67 and 0.04 mg·L−1·day−1 for phosphorus, respectively. Finally, biomass derived from PHAE was higher in lipids (11.3 % versus 7.2 %) and thus has a greater potential as a feedstock for biofuel compared to ADE.
Similar content being viewed by others
Introduction
Algae biomass is poised to become an important biofuel crop in the future, although reductions in production costs are needed in order to achieve a financially competitive product [1]. The US National Research Council (NRC) has determined that in order to create a commercially sustainable algae biofuel, additional research and development of algal production systems utilizing wastewater as a nutrient source is needed [1]. In this regard, the US dairy industry produces large quantities of nutrient laden wastes. The 9.1 million dairy cows in the US generate an estimated 226 billion kg of wet manure each year (2010) [2]. Each ton of manure contains approximately 6.6 kg of nitrogen (N) and 1.1 kg of phosphorus (P) [3]. Pate et al. projects each ton of dry algae biomass requires 88 kg of N and 12 kg of P [4]. If we apply these projections based on N and P content and assume 100 % nutrient uptake, we can estimate the amount of N and P produced by the dairy industry has the potential to produce between 16.95 and 20.72 billion kg of algal biomass∙year−1, assuming no recycling of nutrients. This biomass may be converted to biofuel through various pathways, including anaerobic digestion to biogas, hydrothermal liquefaction to bio-crude, and transesterification of lipids to biodiesel [5]. This amount of algal biomass at 20 % oil content has the potential to produce 0.99 to 1.18 billion gallons of biodiesel∙year−1 [6]. The US consumed 1.37 billion gallons of biodiesel in 2013 [7]; therefore, dairy based algae biodiesel could replace 72–86 % of the annual US biodiesel consumption.
The dairy industry has been proactive in seeking to recover nutrients and carbon from their wastewaters; for example, anaerobic digesters (AD) are utilized to capture value from solid animal wastes in numerous systems [8]. Utilization of ADs as a manure management strategy is projected to increase [8]. A complementary strategy to AD being developed for manure management, involves bacterial utilization of manure-based organic carbon (sugars, organic acids, lipids) to synthesize polyesters of hydroxyalkanoates known as polyhydroxyalkanoates (PHAs)—a polyester molecule that can be formed into biodegradable plastics [9–11]. The PHA process can operate in series with the AD system at the same dairy location because each utilizes different molecular compounds as biological metabolites [12]. The AD and PHA processes both capture a portion of energy from dairy waste, although they also produce wastewater streams containing significant quantities of mineral nutrients (N and P) that require extensive treatment before discharge [13–15]. Leveraging these nutrient-rich effluents, co-locating dairy AD and PHA sites with algae biomass cultivation in an integrated bio-refinery has the potential to improve economic outcomes by transitioning dairy installations from single output systems to multiple commodity production platforms (Fig. 1), thereby improving environmental benefits through increased carbon and nutrient sequestration [16, 17]. However, to maximize the potential of such an integrated system, the process of cultivating algae solely using nutrients from PHAE needs to be better understood, both in the context of ADE and in terms of nutrient remediation of these wastewaters.
Microalgae can be cultivated on dairy AD effluent (ADE) [13, 18] while providing concomitant nutrient removal [19]; however, PHA reactor effluent (PHAE) has not previously been characterized for algal cultivation. Here, we describe the algal growth kinetics and biomass production potential based on the nutrients in PHAE in comparison to ADE, thereby providing insight into a novel wastewater-based nutrient source for algal cultivation, and further demonstrating the potential of an integrated AD-PHA-Algae system. The quality of the algal biomass produced was analyzed in terms of suspended solids, total protein, carbohydrate, and lipid content as well as the types of lipids present. Nutrient removal rates were calculated to assess the relative remediation potential of these wastewaters in consort to algal production. Optical properties as well as key potential inhibitors present in one or both of the wastewaters (e.g., organic acids and ammonia) were assessed to determine which factors influence algal growth inhibition, if any. Finally, the value of an integrated AD-PHAE-Algae system in the context of algal production is discussed relative to algal production on AD effluents alone.
Materials and Methods
Dairy Manure Media
The dairy anaerobic digester effluent (ADE) and polyhydroxyalkanoate reactor effluent (PHAE) used for this study were obtained from systems maintained by Dr. Erik Coats laboratory (University of Idaho). The ADE was centrifuged at 8000 rpm for 5 min at 10 °C, and the PHAE was centrifuged at 5000 rpm for 5 min at 10 °C to remove a majority of solids, both were stored frozen at −20 °C until used to prepare the algal growth medium. After being previously centrifuged and frozen, both effluents were analyzed for nitrogen, phosphorus, and chemical oxygen demand, organic acid composition, and colony forming units (CFU).
Dairy Wastewater Analysis
Organic Acids
Volatile fatty acids (VFA) (acetic, propionic, butyric, isobutyric, valeric, isovaleric, and caproic acids) and methanol were quantified using a Hewlett-Packard 6890 series gas chromatograph (Agilent Technologies, Inc., Santa Clara, CA, USA) equipped with a flame-ionization detector and a Hewlett-Packard 7679 series injector. The system was interfaced with the Hewlett-Packard GC ChemStation software version A.06.01. VFA separation was achieved using a capillary column (Heliflex® AT™-AquaWax-DA, 30 m x 0.25 mm Ø, W. R. Grace & Co., Deerfield, IL, USA) which was ramped from an initial 50 to 200 °C in three steps (following 2 min at 50 °C, ramp to 95 °C at 30 °C∙min−1 then to 150 °C at 10 °C∙min−1 and hold for 3 min; finally, ramp to 200 °C at 25 °C∙min−1 and hold for 12 min) with helium as the carrier gas (1.2 mL∙min−1). The split/split less injector and detector were operated isothermally at 210 and 300 °C, respectively. Prior to analysis, samples were acidified to a pH of 2 using HCl; 0.5 μL of each sample was injected in 20:1 split mode. VFA and methanol concentrations were determined through retention time matching with known standards (Sigma-Aldrich Co., St. Louis, MO, USA; Thermo Fisher Scientific Inc., Waltham, MA, USA) and linear standard curves (R 2 > 0.99).
Colony forming units (CFU)
Tryptic Soy Agar (TSA) and Reasoner’s 2A agar (R2A) plates were prepared using the following media recipes: TSA (per 1000 mL of purified water): 15 g tryptone, 5 g soytone, 5 g sodium chloride, 15 g agar. R2A (per 1000 mL of purified water): 0.5 g proteose peptone, 0.5 g casamino acids, 0.5 g yeast extract, 0.5 g dextrose, 0.5 g soluble starch, 0.3 g dipotassium phosphate, 0.5 g MgSO4·H2O, 0.3 g Sodium pyruvate, 15 g Agar, final pH to 7.2 ± 0.2 at 25 °C. For plating, aliquots of 100 μL of sample was dispensed on both TSA and R2A plates, and incubated for 48 h at 22 °C. Samples were diluted at 1:20 (5 μl of sample + 95 μl of sterile DH2O) for ADE only. CFUs were counted and recorded after 48 h.
Algal Cultures
Chlorella vulgaris (UTEX 2714) was acquired from the University of Texas at Austin Culture Collection (UTEX). Prior to use as an inoculum source in the wastewater experiments, the C. vulgaris stocks were cultured in triplicate in glass dilution bottles using Proteose Medium [20] (per liter): 0.25 g NaNO3, 0.025 g CaCl2·2H2O, 0.075 g MgSO4·7H2O, 0.075 g K2HPO4, 0.175 g KH2PO4, 0.025 g NaCl, 1.0 g Proteose peptone, pH adjusted to 6.8. Cultures were transferred to fresh media on 4-week intervals. Before use, algal inoculums were grown for 2 weeks in Proteose media and rinsed to remove excess Proteose media from the inoculum. The cells were rinsed by centrifugation at 10,000 rpm for 15 min, washed in an equal volume of sterile water, centrifuged again and re-suspended in an equal volume of sterile water prior to inoculation.
Algal Growth in Diluted ADE and PHAE
Dilutions of dairy manure media were made in triplicate using previously frozen non-sterile ADE or PHAE and sterile nanopure filtered water to obtain the desired concentration (5 10 or 20 % by volume). A 500-mL volume of each concentration was dispensed into 1000-mL Erlenmeyer flasks and fitted with air tubing and an air stone for introduction of supplemental 2.5 % v/v CO2 in air; each treatment was performed in triplicate (n = 3). Treatments were inoculated with a 5 % (v/v) amount of washed algal cells (ranging from 2.0 × 105 to 6.0× 105 cells∙mL−1). Each treatment was supplied with a constant gas stream composed of 2.5 % (v/v) CO2 in air to control pH. Compact fluorescent bulbs provided light on a 12-h light/dark cycle. Flasks were shaken and sampled daily; OD680 and chlorophyll were analyzed immediately. Weekly samples were taken and stored frozen at −20 °C for nutrient measurements. All experiments were run as batch cultures.
Determination of Algal Growth
The following methods were used to track algal biomass production throughout each experiment. Since the high turbidity of the cultivation medias can interfere with absorbance-based methods, multiple methods were employed in order to obtain a complete picture of algal growth kinetics.
Total Chlorophyll Method
Methanol solvent extraction was used to extract chlorophyll as adapted from Hipkins [21]. After extraction, absorbance of the chlorophyll/methanol mixture was measured at 650 and 665 nm using an Aquamate spectrophotometer (Thermo Scientific). Chlorophyll content was then calculated using the following equations [21]: chlorophyll a = (16.5*A 665) − (8.3*A 650), chlorophyll b = (33.8*A 650) − (12.5*A 665), total chlorophyll = (25.8*A 650) + (4*A 665).
Direct cell counts
Algal cell density was measured using a hemocytometer. Ten-microliter samples were dispensed into the counting chamber and algal cells were counted at ×10 magnification.
Absorbance at 680 nm
The direct absorbance (optical density) was measured at 680 nm on an Aquamate spectrophotometer (Thermo Scientific), as an indication of turbidity attributed to a combination of algal cells, bacteria cells, and dissolved organic matter. The machine was zeroed using deionized water.
Total solids (TS)
On day 21 of each experiment, 30 mL of algal suspension was removed from the flask and placed into a pre-weighed drying tin. Samples were dried overnight at 50 °C and 25″ Hg vacuum. Dried samples were weighed in the tin and the difference taken as the total solids weight. Total volatile solids (TVS) was determined by combusting each sample in a muffle furnace at 550 °C for 4 h, and subtracting the weight of the combusted sample from the total solids weight.
Specific growth rate
The data collected from the direct cell counts was used to calculate specific growth rate (μ). The exponential growth phase was dependent on the wastewater and concentration, encompassing the following days: ADE media at 5 (0–4), 20 (0–6) and 10 % (0–6); PHAE media at 5 (2–6), 10 (2–6), and 20 % (2–6). The cell counts for these days were used to calculate corresponding specific growth rates using the following equation:
where
- μ :
-
Specific growth rate
- N o :
-
Cell count at initial time point
- N t :
-
Cell count at final time point
- Δt :
-
Time span used for cell count measurement
Biomass Characteristics
Lipid Composition and Fatty Acid Profile
Lipid and fatty acid profile methods were adapted from Osman et al. [22]. Freeze-dried algae of known weight (20–50 mg) was extracted with CH2Cl2 (10 mL) with constant stirring in a glass culture tube for 36 h. The solution was centrifuged and the supernatant transferred to a vial and evaporated to dryness under a stream of nitrogen gas and the yield recorded. A portion of the extract (1–2 mg) of known weight was transferred to a 5-mL reacti-vialTM to which 2 mL of MeOH/H2SO4/CHCl3 (1.7:0.3:2.0 v/v/v) was added, and the mixture was heated for 90 min at 90 °C. CHCl3 contained 1-napthalaneacetic acid as an internal standard (50 μg∙mL−1). A 1-mL volume of water was added to the cooled mixture, vigorously shaken, centrifuged, the CHCl3 layer removed, dried through anhydrous sodium sulfate, and transferred to a GC vial. The prepared fatty acid methyl ester (FAME) derivatives were analyzed by GC-MS (FOCUS-ISQ, ThermoScientific); temperature profile: 40 °C (1 min) → 5 °C∙min−1 to 250 °C; GC capillary column: RTx-5MS (30 m x 0.25 mm Ø, Restek). The eluted compounds were identified with authentic standards and by spectral matching with the 2008 NIST mass spectral library.
Carbohydrate and protein analysis
Algae were freeze dried (20–50 mg), and carbohydrate content was analyzed as alditol acetate derived by GC-MS as described by Chakraborty et al. [23]. Protein content was determined on lyophilized cell pellets using the Lowry method after extraction of total protein in 24 % trichloroacetic acid and expressed as mg protein per mg of biomass as described in Slocombe et al. 2013 [24].
Nutrient Measurements
All nutrient measurements were performed using colorimetric methods and an Aquamate spectrophotometer (Thermo Scientific). Samples were passed through a 0.45-μm filter prior to all nutrient analysis to remove particulates and algal cells, except for the COD test. Test methods used were as follows: total dissolved nitrogen (Hach persulfate digestion method #10071), dissolved ammonia nitrogen (NH3-N) (Hach salicylate method #10023 as adapted from Reardon et al. [25]), dissolved nitrate nitrogen (NO3 --N) (Hach cadmium reduction method #8171), total dissolved phosphorus (Hach PhosVer 3 with acid persulfate digestion method #8190), and Chemical oxygen demand (Hach reactor digestion method #8000, USEPA standard method 5220 D). Samples were diluted accordingly in order to obtain the appropriate concentration range for each test.
Testing of Potential Inhibitory Components
To determine the chemical components in the wastewaters that influenced algal growth, separate batch experiments with defined media amended with organic acids or ammonia at levels present in the ADE were performed. Modified Chu 13 mineral media was used for all inhibitory tests [26] (per liter): 200 mg KNO3, 100 mg MgSO4·7H2O, 52 mg K2HPO4·3H2O, 54 mg CaCl2·2H2O, 10 mg FeNaEDT, and 5 mL trace elements solution (572 mg H3BO3, 308 mg MnSO4∙H2O, 44 mg ZnSO4·7H2O, 16 mg CuSO4·5H2O, 12 mg NaMoO4·2H2O, 18 mg CoSO4·7H2O in 1 L or water).
Each experimental treatment received 100 mL of sterile Modified Chu 13 media and one of the potentially inhibitory compound in the following concentrations: 111.9 mg·L−1 acetate [CH3COO−] (simulating 20 % v/v ADE), 44.69 mg·L−1 propionate [C2H5COO−] (simulating 20 % v/v ADE), or 33.7 mg·L−1 ammonia-nitrogen [NH3-N] (simulating 5 % v/v ADE) as the sole nitrogen source. To account for changes in ionic strength in the media containing the organic acid treatment, the controls were constructed with equivalent concentrations of NaCl. For treatments receiving ammonia, controls were constructed with an equal molar nitrogen [N] concentration of KNO3 as the sole nitrogen source. Treatments were inoculated with C. vulgaris, sparged with 2.5 % (v/v) CO2 in air and incubated in flasks as described above. Bottles were shaken daily before sampling; OD680 and chlorophyll were then measured immediately.
Statistical Analysis
Statistical analysis was performed using raw data and SigmaPlot 12.3. A one-way repeated measures ANOVA was performed to determine if nutrient removal was significant over time for each wastewater and for each dilution. All sample sets passed normality test using Shapiro-Wilk with p value to reject of 0.050. Equal variance p value to reject was 0.050. Post hoc multiple comparisons were performed using Tukey analysis.
Results and Discussion
To understand the dynamics at play in a complicated wastewater-algae system, our approach was based on two goals: (1) perform algal growth experiments employing Chlorella vulgaris, as a type algal species, using various dilutions of each wastewater to determine growth kinetics (growth rates, duration of growth phase) and determine differences in final biomass, lipid, protein, and carbohydrate content and (2) analyze and quantify the properties in each wastewater that potentially influenced growth of C. vulgaris along with identifying specific compounds that caused growth inhibition.
Growth of Chlorella Vulgaris in ADE and PHAE
ADE has been shown to support algae growth [13, 27], although PHAE has not previously been characterized as a feedstock for algal production. Cultivation experiments were performed to determine the differences in growth response of C. vulgaris to ADE versus PHAE wastewaters at concentrations of 5 10 and 20 % (v/v). Results from C. vulgaris cultivation experiments were compared with respect to equivalent levels of dilution among the effluent types, followed by normalization to initial nitrogen load in each treatment.
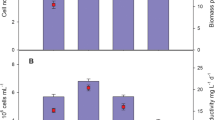
Stationary phase algal cell concentrations were 3–4 times higher in the 5 and 10 % PHAE (29.30E + 06 and 40.42E + 06 cells·mL−1) compared to the same concentrations of ADE (10.02E + 06 and 10.12E + 06 cells · mL−1); no difference was seen between the 20 % wastewater treatments (Fig. 2 a, b). Contradictory to the cell counts, the stationary phase total solid (TS) measurements were higher for all concentrations of ADE (0.89, 1.39, 2.23 g·L−1) compared to PHAE (0.67, 1.07, 1.57 g·L−1) (Table 2). This discrepancy may be a result of higher concentrations of indigenous microorganisms and/or particulate organic matter in the ADE compared to the PHAE; pre-inoculated wastewater testing showed that all dilutions of ADE were twice as high in TS compared to PHAE (Table 2), and consequentially, chemical oxygen demand (COD) was also twice as high in ADE (Table 1). No observations were made to distinguish algal cell size between the different treatments; therefore, we cannot contribute cell size to TS differences. In addition, the cellular mass (g·cell−1) of C. vulgaris is reported to vary widely depending on nutrient conditions [28]; therefore, a TS calculation based on cell counts was unrealistic.
Growth Kinetics of C. Vulgaris Between ADE and PHAE
Exponential growth rates were elevated for PHAE media (0.59, 0.53, 0.42 days−1) compared to ADE media (0.40, 0.36, 0.37 days−1) (Table 2), although a divergence in growth patterns between the two wastewaters emerged for the remainder of the time to stationary phase (Fig. 2). After the initial exponential period, algal cultures in all dilutions of the ADE experienced reduced and/or negative growth whereas the cultures in the PHAE exhibited continued positive growth for the 5 and 10 % concentrations, the 20 % PHAE concentration also displayed a reduced growth rate similar to the ADE. The longer positive growth phase in the 5 and 10 % PHAE translated into higher stationary cell concentrations compared to the ADE treatments.
Equal concentrations of wastewaters did not contain equivalent concentrations of nutrients (N and P) (Fig. 3); therefore, nitrogen loading may have been a factor in determining growth rates. To compensate for this, a normalized growth rate was calculated per unit (g∙L−1) of nitrogen loading; growth rates for 5, 10, and 20 % PHAE media (22.88, 4.73, 3.14 days−1 per g∙L−1 N) compared to ADE media (6.47, 4.42, 3.48 days−1 per g∙L−1 N). The higher growth rate for 5 and 10 % PHAE indicates that nitrogen loading was not a significant factor in determining growth rates since this media had the lowest nitrogen loading. This is consistent with Converti et al. where C. vulgaris growth rates were not significantly changed when nitrogen (NaNO3) loading was half and one quarter of the original growth media [29]. We can conclude that although the two wastewaters are not directly comparable because of a multitude of physicochemical differences, nitrogen loading was not directly responsible for differences in the observed growth rates.
Total dissolved phosphorus (TDP) concentrations in solution over the 21-day time course of the algal growth experiments in ADE (top) and PHAE (bottom) for 5, 10, and 20 % dilutions. Treatment levels that share a letter are not significantly different from one another (p > 0.05). Mean values (n = 3), ± one standard deviation
Biomass Lipid, Carbohydrate, and Protein Content
The composition of the algal biomass determines the bio-crude yield in a hydrothermal liquefaction process, with lipids being the main contributor to the bio-crude product [30]. The cellular macromolecules that contribute to the bio-crude yield follow the trend lipids > proteins > carbohydrates [30]. The total lipid and fatty acid profile of the algae cultivated in ADE and PHAE was assessed for the stationary phase biomass in the 5 % wastewater dilutions only (Table 3). The lipid fraction of the dry biomass was 7.2 and 11.3 % w/w for ADE and PHAE, respectively, revealing that PHAE wastewater produced a biomass that will theoretically have a higher bio-oil yield in a hydrothermal liquefaction process. In comparison, Wang et al. found that Chlorella cultivated in various dilutions of AD dairy manure resulted in an algal biomass with a lipid content ranging from 9.0 to 13.7 % of dry biomass [13]. Higher lipid contents in PHAE media can be attributed to lower nitrogen loading, resulting in a longer duration of N-deprivation during stationary phase. For example, the levels of dissolved nitrate and ammonia were exhausted by day 21 in the 5 % PHAE treatment, whereas the 5 % ADE still contained approximately 40 mg/L of nitrogen, primarily in the form of ammonia (Fig. 4). This is consistent with lipid data found by Converti whereas C. vulgaris cultures cultivated in media with a quarter of the control nitrogen resulted in a threefold increase in cellular lipids [29].
Total dissolved nitrogen (TDN) concentrations in solution over the 21-day time course of the algal growth experiments in ADE (top) and PHAE (bottom) for 5, 10, and 20 % dilutions. Treatment levels that share a letter are not significantly different from one another (p > 0.05). Mean values (n = 3), ± one standard deviation
C. vulgaris cultivated in PHAE was higher in carbohydrate content (31.3 % in PHAE vs. 26.7 % in ADE). As with lipid accumulation, carbohydrate accumulation is induced in C. vulgaris under N-limitation [31, 32]. Algal protein content was similar for both medias, ADE (37.3 %) and PHAE (39.5 %). These differences could influence the value of the algal biomass and its ultimate application. For example, biomass with a higher lipid content is more appropriate for as feedstock for a biofuel produced via hydrothermal liquefaction [33].
Nutrient Sequestration
The dissolved phosphorus and nitrogen concentrations in the ADE and PHAE were tracked throughout the growth experiments as a means to measure nutrient removal for estimating the utility of algae to provide wastewater nutrient remediation. We can assume that, in addition to multiple physicochemical factors inherent in the wastewater, the form of N present greatly influences the sequestration potential.
Nitrogen
The N species were notably different between the two wastewaters, ammonia was the primary species in ADE at 39.6–83.6 % of the total N, and nitrate was the primary species in PHAE at 40.3–60.5 % of the total N (Table 2). A one-way ANOVA between sampling times over weekly time increments (0, 7, 14, 21 days) for each wastewater and for each concentration showed a significant decrease in total dissolved nitrogen (TDN) for all of the PHAE dilutions, whereas no significant change was found for any of the ADE treatments (Table 2, Fig. 4). This may be a result of the higher nitrate/nitrogen ratio in the PHAE compared to ADE, as nitrate is a more bio-available form of N for C. vulgaris than NH3 [34]. In addition, decomposition of particulate and/or dissolved organic matter by heterotrophic microbial activity may be occurring in the ADE, releasing organically bound nitrogen into solution. This organic N consequently may have added to the dissolved N pool and artificially reduced the observed TDN removal rates. Interestingly, both nitrate and ammonia were removed from the PHAE wastewater whereas neither species was significantly removed from the ADE wastewater (Fig. 5).
The mean overall TDN rate of change in the 5, 10, and 20 % PHAE media was −0.85, −3.44, and −2.13 mg∙L−1 day−1, respectively, with most of the TDN being removed within the first 7 days of incubation, encompassing the period of exponential phase growth. The mean overall TDN rate of change in the 5, 10, and 20 % ADE media was −0.52, −0.95, and +1.57 mg∙L−1 day−1. Notably, the TDN removal rates in the ADE wastewater were not statistically significant over the sampling times (0, 7, 14, 21 days).
In both wastewater medias, the 10 % dilutions had the highest mean dissolved N removal efficiency, at 68 and 20 % for PHAE and ADE, respectively, over the incubation period. This compares to research by Wang et al. who found that TDN was removed in 21 day batch cultures of Chlorella sp. at 75.7–82.5 % efficiency in AD dairy manure that was previously filtered prior to use (i.e., particulate organic material removed), with TDN loading ranging from 81 to 178 mg∙L−1 [13]. In our case, the wastewaters were not filtered, thus allowing for organically bound N to replenish the dissolved N pool.
Phosphorus
The total dissolved phosphorus (TDP) loading was two- to fourfold higher in the ADE wastewaters compared to PHAE for equal dilutions (Table 2). The mean overall TDP rate of change in the 5, 10, and 20 % PHAE media was +0.05, −0.04, and +0.11 mg∙L−1 day−1, respectively. The mean overall TDP rate of change in the 5, 10, and 20 % ADE media was +0.14, −0.67, and. −0.33 mg∙L−1 day−1. The removal of TDP was not as significant as expected; a large concentration of dissolved P was still present at the end of the 21-day incubation time for both wastewaters (Fig. 3). Although the highest P removal rates were for the 10 % wastewaters in both cases, these reductions were only temporary as TDP eventually recovered to day zero levels. Wang et al. found that after a 21-day incubation, Chlorella sp. can remove 62.5–74.7 % of the TDP in ADE that has been filtered through a 1.5-μm glass filter (i.e., when the particulate organic matter is removed prior to the effluent being used as an algal cultivation medium) [13].
Similar to the patterns observed for TDN, the low removal rate of TDP in both ADE and PHAE systems was most likely a result of decomposition of organic matter resulting in a slow release of particulate P into soluble forms, even as algal uptake occurred. In addition to dissolved organic matter (DOM), both wastewaters had a large amount of visible particulate organic matter associated with them; this particulate material accounted for 54–65 % of the available phosphorus in the system overall. Francko et al. found that phosphorus is labile in an aqueous system; particulate organic matter in the wastewaters can act to sorb labile phosphorus resulting in cycling between dissolved and sorbed forms in response to changing conditions (pH, temperature, etc.) [35]. Organically bound P generally exists as an iron complex (Fe 3+), with release induced by aerobic conditions and lower redox potentials associated with elevated microbial activity [36]. Importantly, in our system, particulate organic matter was not filtered out of the wastewaters prior to use; thus, the presence of this matter likely reduced the observed nitrogen and phosphorus removal efficiencies.
Potential Inhibitory Components Influencing C. Vulgaris Growth
Yields for algae biomass grown in agricultural wastewaters can vary widely, from 6 to 700 mg·L−1·day−1 depending on wastewater source and physicochemical properties [37]. While C. vulgaris cultures exhibited sustained positive growth in 5 and 10 % PHAE dilutions, all concentrations of ADE and the 20 % PHAE demonstrated reduced and/or negative biphasic growth kinetics following the exponential phase. Biphasic growth has previously been documented in algal cultures grown on diluted sewage treatment effluent, although no explanation for this phenomena was reached in this case [38]. Characterization of the ADE and PHAE dairy wastewaters shows a dramatic difference in their chemical, biological, and optical properties (Table 1). We present the following observations and controlled experiments that suggest the causes for the biphasic growth response of C. vulgaris in these wastewaters is multifaceted due to the complex makeup of the wastewaters.
Organic Acids
The ADE dairy wastewater contained organic acids, the most abundant being acetate (456 mg∙L−1) and propionate (156 mg∙L−1), whereas organic acids were absent or below detection in the PHAE dairy wastewater (Table 1). Organic acids at low concentrations may be helpful in some algal cultures, as some algae can grow mixotrophically on carbon substrates such as glycerol, acetate, and glucose during times of low or no light to achieve higher biomass concentrations than would otherwise be obtained under photoautotrophic conditions [39]. Even though organic acids can be assimilated into algal biomass, elevated levels of organic carbon are known to be inhibitory to algal growth. C. vulgaris cultures exposed to increasing doses of glycerol from 1 to 10 % (v/v) reduced biomass productivity from 102 to 7 mg·L−1·day−1, and exposure to increasing doses of glucose from 1 to 10 % (w/v) reduced biomass productivity from 254 to 62 mg·L−1·day−1 [40].
Batch experiments were employed with the addition of organic acids to standard mineral media (Modified Chu 13) to determine the effects of acetate and propionate, at the levels present in the ADE, on the growth of C. vulgaris. The addition of acetate at concentrations simulating 20 % ADE (111.9 mg·L−1 CH3COO−) resulted in 39 % less chlorophyll per volume of culture, mean chlorophyll values (± std. dev.) on day 8 for the acetate and control cultures were 1.86 (0.28) mg∙L−1 and 3.07 (1.25) mg∙L−1, respectively (Fig. 6). The algal culture absorbance was also reduced by 48 %, mean A 680 values (±std. dev.) on day 8 for acetate and control cultures were 0.15 (0.03) and 0.29 (0.17), respectively.
Similarly, the addition of propionate at concentrations simulating 20 % ADE (44.7 mg·L−1 C2H5COO−) resulted in 65 % less chlorophyll per volume of culture, mean chlorophyll values on day 8 for the propionate, and control cultures were 2.25 (0.47) mg·L−1 and 6.45 (1.11) mg·L−1, respectively (Fig. 6). The algal culture absorbance was also reduced by 65 %, mean A 680 values (±std. dev.) on day 8 for propionate and control cultures were 0.25 (0.05) and 0.70 (0.16), respectively.
The chlorophyll content of C. vulgaris can be expected to decrease in the presence of organic carbon substrates if the algae employ heterotrophic instead of phototrophic metabolism. In addition, we would expect to see the culture absorbance (A 680) increase as cell density increases during a growth phase. However, both the chlorophyll and absorbance values followed the same patterns for both organic carbon substances tested; therefore, no indication of heterotrophic growth using acetate or propionate was seen. This suggests that both acetate and propionate in the ADE dairy wastewater had an inhibitory effect on algae growth.
Nitrogen
Wastewaters often contain sufficient concentrations of nutrients for algal growth, although not always in the most favorable form; a large pool of N and P can exist in organically bound material that is not accessible to algae [34, 41]. Additionally, nitrogen speciation varies by wastewater, with ammonia being the primary species in ADE and nitrate being the primary species in PHAE. Elevated ammonia levels can inhibit phototrophic growth. For example, the green alga Nephroselmis pyriformis displays toxicity to ammonia at a concentration of 32.8 μg∙L−1 NH3-N [42]. Similarly, sewage oxidation ponds used as a means to treat wastewater with algae are adversely affected by ammonia, particularly when present in the un-ionized form (NH3) (common at high pH > 8) as opposed to ionized form (NH4 +) [43]. Typical dairy ADE ammonia concentration can vary widely, ranging from 255 to 519 mg∙L−1, depending on the reactor type (one phase or two phase) [44]. In our case, the undiluted ADE was much higher even yet (761 mg·L−1 NH3-N) and was expected to negatively influence algal growth kinetics, even though the ammonia was primarily in the ionized form (NH4 +) since the pH was buffered between 6.5 and 7 through CO2 addition.
Batch experiments were performed using ammonia as the sole nitrogen source to standard mineral media (Modified Chu 13) and compared to nitrate as the sole nitrogen source. Ammonia at 33.7 mg·L−1 NH3-N (simulating 5 % ADE) inhibited C. vulgaris growth, resulting in a 62 % lower chlorophyll content. The mean values were statistically significant on day 12; chlorophyll content for algal cultures in ammonia media was 3.46 (1.51) mg·L−1 compared to 9.10 (0.79) mg·L−1 for controls (Fig. 6). Ammonia levels were below detection level in the PHAE dairy wastewater and therefore were not considered to be inhibitory to phototrophic growth in that case.
Nutrient Competition
The presence of an initial indigenous bacteria load (as measured by colony forming units) was starkly different for the two wastewaters, with total CFUs approximately 1000 times higher for ADE relative to PHAE (Table 1). The initial presence and subsequent proliferation of prokaryotic microorganisms in batch cultures can increase algal growth through heterotrophic CO2 production, decomposition of organic matter, assimilation of algal by-products, and ammonia oxidation [38]. However, the same microorganisms can reduce algal growth through competition for nutrients [38].
Algae and bacteria compete for dissolved nutrients in wastewater and bacteria have a higher affinity for P than algae due to the existence of two high-affinity regulated transport systems [45]. Further, Currie et al. explained that growth of bacteria is typically not P limiting, rather growth tends to be restricted by energy supply (e. g. dissolved reduced carbon for heterotrophic metabolism) [46–48]. In relation to our wastewaters, the ADE had an abundance of dissolved reduced carbon (i.e., organic acids) that was absent in the PHAE. Because of this, we would expect that algae grown in ADE wastewater (high bacterial loads and dissolved reduced carbon) might experience algae P limitation due to competition with endogenous bacteria, causing slower overall growth rates and reducing yield.
Optical Properties
Photosynthetic activity in dairy wastewater can be limited by light attenuation (absorption, scattering, and fluorescence) caused by the presence of DOM and suspended solids [49]. Chromophoric dissolved organic matter (CDOM) is the optically active fraction of DOM that absorbs light in the short wavelengths (blue to UV). CDOM effects phototrophic growth to varying degrees dependent on algal species and in some cases can stimulate growth by reducing photoinhibition [50]. Suspended solids contribute to turbidity, resulting in light attenuation and diminished phototrophic activity. For example, Wang et al. found that specific growth rate of Chlorella sp. is inversely proportional to initial turbidity for algae grown in digested dairy manure [13].
An absorbance measurement at 680 nm shows that light attenuation is over three times higher in ADE compared to PHAE (Table 1). Similarly, UV–Vis absorbance spectra (200–800 nm) for the three dilutions of both wastewaters revealed that light absorbance over this wide range was 2–3 times higher in ADE compared to PHAE (Fig. 7). All of the ADE concentrations and the 20 % PHAE had higher absorptions than the 5 and 10 % concentrations of PHAE, including strong absorption characteristics in the blue and red regions. More specifically, the two absorption maxima for C. vulgaris (430 and 675 nm) that are typical of algae light harvesting pigments [51] were more strongly absorbed by the ADE in the absence of C. vulgaris. The percent transmittance of light (%T) through the wastewaters at 430 nm was 86, 73, and 52 % for increasing concentrations of PHAE and 75, 61, and 34 % for increasing concentrations of ADE. The corresponding %T at 675 nm was 99, 98, and 96 % for PHAE and 96, 94, and 87 % for ADE. Based on the reduced light transmittance levels, we can deduce that less photo-reactive light is available for phototrophic metabolism, particularly in the ADE-based treatments and the high concentration (20 %) PHAE treatment. This low light transmittance likely affected growth rates due to limitation in light availability between the two types of wastewater-based growth medias and between the concentration levels.
Conclusion
Anaerobic digestion and polyhydroxyalkanoate reactor deployment are two logical solutions for dairy manure management and generation of renewable energy or other bio-products; however, complete nutrient removal associated with wastewater discharge is a challenge for both processes [17, 52]. To realize broader deployment and acceptance, an integrated process should be constructed such that all environmental emissions are maximally mitigated concurrently [17]. To accomplish this, a coupling of reactor effluents with algal biomass production provides an elevated carbon and nutrient recovery along with generation of value added products. This combination can inherently minimize costs while reducing the carbon footprint and environmental contamination traditionally associated with dairy manure.
The growth kinetics and yield of C. vulgaris was dramatically affected by the wastewater source. The ADE wastewater contained more chemical inhibitory components (organic acids and ammonia) as well as higher bacteria loads and increased light attenuation, creating a harsher environment for algae cultures to grow and negatively affecting growth rates and biomass yield.
PHAE proved to be a better medium for algal cultivation in batch experiments compared to the same dilution of ADE, based on higher cell counts as a result of longer growth period. Algal cultures in all of the ADE wastewater dilutions and the 20 % PHAE demonstrated a growth phase marked by a biphasic pattern, ultimately resulting in decreased cellular yields. Individual inhibitory components or a combination of these components, such as organic acids, ammonia, light attenuation, and herbivory and/or nutrient competition by indigenous microorganisms were possible causes for the distinctly different growth patterns.
Nutrient sequestration is a potential added value of an integrated AD/PHA—algae dairy waste treatment system. The utility of using algae for wastewater remediation was most successful in the PHAE, as TDN removal occurred over all dilutions with 3.44 mg·L−1·day−1 being the highest rate observed. Conversely, there was no significant nitrogen removal from the ADE over the 21-day experimental duration. Total dissolved phosphorous (TDP) removal was not significant for either wastewater media. If nutrient sequestration is to be an achievable goal for a wastewater/algae system, separation of particular organic matter from the wastewater may improve nutrient removal rates.
The algal biomass from a waste-to-algae process is of value for a variety of applications; feedstock for a thermochemical conversion to bio-crude, extraction of lipids for conversion to biodiesel, use as a high protein animal feed, slow release fertilizer, or as a supplemental feedstock to the AD system [30, 53–55]. The carbohydrate and lipid compositions of the dry biomass varied depending on the type of wastewater employed, which would ultimately determine the appropriate use for the biomass. This work demonstrates the utility of dairy PHA reactor effluent as a suitable alternative to AD effluent as a nutrient source for algal cultivation. Although ADE has been demonstrated as a potentially viable nutrient source for algal cultivation, here, algae cultivated in PHAE resulted in biomass with a higher lipid content and thus has greater potential as biofuel feedstock.
Abbreviations
- AD:
-
Anaerobic digester
- ADE:
-
Anaerobic digester effluent
- CFU:
-
Colony forming unit
- CH4 :
-
Methane
- COD:
-
Chemical oxygen demand
- CO2e:
-
Carbon dioxide equivalent
- DOM:
-
Dissolved organic matter
- EROI:
-
Energy return on investment
- GHG:
-
Greenhouse gas
- N:
-
Nitrogen
- NH3 :
-
Ammonia
- NO3 – :
-
Nitrate
- PHA:
-
Polyhydroxyalkanoate
- PHAE:
-
Polyhydroxyalkanoate reactor effluent
- P:
-
Phosphorus
- RFS:
-
Renewable fuel standard
- SD:
-
Standard deviation
- TDP:
-
Total dissolved phosphorus
- TDN:
-
Total dissolved nitrogen
- TS:
-
Total solids
- VFA:
-
Volatile fatty acid
References
National Research Council (U.S.). Committee on the Sustainable Development of Algal Biofuels., National Research Council (U.S.). Board on Agriculture and Natural Resources., National Research Council (U.S.). Board on Energy and Environmental Systems., National Academies Press (U.S.), Sustainable development of algal biofuels in the United States, National Academies Press, Washington, D.C. 2012
Liebrand CB, Ling KC (2009) Research Report 217
United States, SCS (2012) Agricultural waste management field handbook
Pate R, Klise G, Wu B (2011) Resource demand implications for US algae biofuels production scale-up. Appl Energy 88:3377–3388
Craggs R, Heubeck S, Lundquist T, Benemann J (2011) Algal biofuels from wastewater treatment high rate algal ponds. Water Sci Technol 63(4):660–665
Massingill M, Carlbert J, Schwartz G, Van Olst J, et al (2008) Algae Biomass Summit, Seattle, WA
U.S. Energy Information Administration, U.S (2013) Department of Energy
Innovation Center for U.S (2011) Dairy
Coats ER, Loge FJ, Wolcott MP, Englund K, McDonald AG (2007) Synthesis of polyhydroxyalkanoates in municipal wastewater treatment. Water Environ Res 79:2396–2403
Liu HY, Hall PV, Darby JL, Coats ER et al (2008) Production of polyhydroxyalkanoate during treatment of tomato cannery wastewater. Water Environ Res 80:367–372
Wei L, Guho NM, Coats ER, McDonald AG (2014) Characterization of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) biosynthesized by mixed microbial consortia fed fermented dairy manure. J Appl Polymer Sci. 131, n/a-n/a
Coats ER, Searcy E, Feris K, Shrestha D et al (2013) An integrated two-stage anaerobic digestion and biofuel production process to reduce life cycle GHG emissions from US dairies. Biofuels Bioprod Biorefin 7:459–473
Wang L, Li Y, Chen P, Min M et al (2010) Anaerobic digested dairy manure as a nutrient supplement for cultivation of oil-rich green microalgae Chlorella sp. Bioresour Technol 101:2623–2628
Bosco F, Chiampo F (2010) Production of polyhydroxyalcanoates (PHAs) using milk whey and dairy wastewater activated sludge: production of bioplastics using dairy residues. J Biosci Bioeng 109:418–421
Coats ER, Gregg M, Crawford RL (2011) Effect of organic loading and retention time on dairy manure fermentation. Bioresour Technol 102:2572–2577
Christenson L, Sims R (2011) Production and harvesting of microalgae for wastewater treatment, biofuels, and bioproducts. Biotechnol Adv 29:686–702
Coats ER, Searcy E, Feris K, Shrestha D, et al (2013) An integrated two-stage anaerobic digestion and biofuel production process to reduce life cycle GHG emissions from US dairies. Biofuels Bioprod Biorefin
Sooknah RD, Wilkie AC (2004) Nutrient removal by floating aquatic macrophytes cultured in anaerobically digested flushed dairy manure wastewater. Ecol Eng 22:27–42
Kebede-Westhead E, Pizarro C, Mulbry WW (2003) Production and nutrient removal by periphyton growth under different loading rates of anaerobically digested flushed dairy manure. J Phycol 39:1275–1282
University of Texas Culture Collection (2013) University of Texas
Hipkins M (1986) Photosynthesis energy transduction a practical approach. IRL press, Oxford
Osman NB, McDonald AG, Laborie M-PG (2012) Analysis of DCM extractable components from hot-pressed hybrid poplar. Holzforschung 68:927–934
Chakraborty M, McDonald AG, Nindo C, Chen S (2013) An α-glucan isolated as a co-product of biofuel by hydrothermal liquefaction of Chlorella sorokiniana biomass. Algal Res 2:230–236
Slocombe SP, Ross M, Thomas N, McNeill S, Stanley MS (2013) A rapid and general method for measurement of protein in micro-algal biomass. Bioresour Technol 129:51–57
Reardon J, Foreman JA, Searcy RL (1966) New reactants for the colorimetric determination of ammonia. Clin Chim Acta Int J Clin Chem 14:203
Largeau C, Casadevall E, Berkaloff C, Dhamelincourt P (1980) Sites of accumulation and composition of hydrocarbons in Botryococcus braunii. Phytochemistry 19:1043–1051
Levine RB, Costanza-Robinson MS, Spatafora GA (2011) Neochloris oleoabundans grown on anaerobically digested dairy manure for concomitant nutrient removal and biodiesel feedstock production. Biomass Bioenergy 35:40–49
Chia MA, Lombardi AT, Melano MdGG (2011) World Aquaculture Society
Converti A, Casazza AA, Ortiz EY, Perego P, Del Borghi M (2009) Effect of temperature and nitrogen concentration on the growth and lipid content of < i > Nannochloropsis oculata</i > and < i > Chlorella vulgaris</i > for biodiesel production. Chem Eng Process Process Intensif 48:1146–1151
Biller P, Ross A (2011) Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour Technol 102:215–225
Behrens PW, Bingham SE, Hoeksema SD, Cohoon DL, Cox JC (1989) J Appl Phycol, pp. 123–130
Dragone G, Fernandes BD, Abreu AP, Vicente AA, Teixeira JA (2011) Nutrient limitation as a strategy for increasing starch accumulation in microalgae. Appl Energy 88:3331–3335
Ross AB, Biller P, Kubacki ML, Li H et al (2010) Hydrothermal processing of microalgae using alkali and organic acids. Fuel 89:2234–2243
Pehlivanoglu E, Sedlak DL (2004) Bioavailability of wastewater-derived organic nitrogen to the alga Selenastrum Capricornutum. Water Res 38:3189–3196
Francko DA, Heath RT (1982) UV-sensitive complex phosphorus: association with dissolved humic material and iron in a bog lake. Limnol Oceanogr 27:564–569
Jensen HS, Andersen FO (1992) Importance of temperature, nitrate, and pH for phosphate release from aerobic sediments of four shallow, eutrophic lakes. Limnol Oceanogr 37:577–589
Pittman JK, Dean AP, Osundeko O (2011) The potential of sustainable algal biofuel production using wastewater resources. Bioresour Technol 102:17–25
Bolier G, Donze M (1989) On the accuracy and interpretation of growth curves of planktonic algae. Hydrobiologia 188:175–179
Liu X, Duan S, Li A, Xu N et al (2009) Effects of organic carbon sources on growth, photosynthesis, and respiration of Phaeodactylum tricornutum. J Appl Physiol 21:239–246
Liang Y, Sarkany N, Cui Y (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol Lett 31:1043–1049
Sonzogni WC, Chapra SC, Armstrong DE, Logan TJ (1982) Bioavailability of phosphorus inputs to lakes. J Environ Qual 11:555–563
Källqvist T, Svenson A (2003) Assessment of ammonia toxicity in tests with the microalga, Nephroselmis pyriformis, Chlorophyta. Water Res 37:477–484
Abeliovich A, Azov Y (1976) Toxicity of ammonia to algae in sewage oxidation ponds. Appl Environ Microbiol 31:801–806
Uludag-Demirer S, Demirer GN, Chen S (2005) Ammonia removal from anaerobically digested dairy manure by struvite precipitation. Process Biochem 40:3667–3674
Jansson M (1988) Phosphate uptake and utilization by bacteria and algae, Phosphorus in Freshwater Ecosystems, Springer, pp. 177–189
Currie DJ, Kalff J (1984) A comparison of the abilities of freshwater algae and bacteria to acquire and retain phosphorus. Limnol Oceanogr 29:298–310
Currie DJ, Kalff J (1984) Can bacteria outcompete phytoplankton for phosphorus? A chemostat test. Microb Ecol 10:205–216
Currie DJ, Kalff J (1984) The relative importance of bacterioplankton and phytoplankton in phosphorus uptake in freshwater. Limnol Oceanogr 29:311–321
Davies-Colley R, Craggs R, Park J, Nagels J (2005) Optical characteristics of waste stabilization ponds: recommendations for monitoring. Water Sci Technol 51:153–161
Bährs H, Steinberg CEW (2012) Impact of two different humic substances on selected coccal green algae and cyanobacteria—changes in growth and photosynthetic performance. Environ Sci Pollut Res 19:335–346
Yun YS, Park JM (2001) Attenuation of monochromatic and polychromatic lights in Chlorella vulgaris suspensions. Appl Microbiol Biotechnol 55:765–770
Engler CR, Jordan ER, McFarland MJ, Lacewell RD (2003) Economics and environmental impact of biogas production as a manure management strategy. Department of Agricultural Engineering, Texas A&M University
Wilkie AC, Mulbry WW (2002) Recovery of dairy manure nutrients by benthic freshwater algae. Bioresour Technol 84:81–91
Mulbry W, Westhead EK, Pizarro C, Sikora L (2005) Recycling of manure nutrients: use of algal biomass from dairy manure treatment as a slow release fertilizer. Bioresour Technol 96:451–458
Sialve B, Bernet N, Bernard O (2009) Anaerobic digestion of microalgae as a necessary step to make microalgal biodiesel sustainable. Biotechnol Adv 27:409–416
Acknowledgments
This research was funded in part by the Idaho National Laboratory (INL) and the Center for Advanced Energy Studies (CAES), 00041394 Task Order 33. Additional funding was provided by the Environmental Protection Agency (EPA), Science to Achieve Results (STAR) graduate fellowship, 2011–2013. FP-91736101, and the United States Department of Agriculture National Institute of Food and Agriculture (USDA-NIFA) award number 2012–68002–19952.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Passero, M., Cragin, B., Coats, E.R. et al. Dairy Wastewaters for Algae Cultivation, Polyhydroxyalkanote Reactor Effluent Versus Anaerobic Digester Effluent. Bioenerg. Res. 8, 1647–1660 (2015). https://doi.org/10.1007/s12155-015-9619-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12155-015-9619-9