Abstract
Aim
To assess the diagnostic and therapeutic impact of PET/CT with 18F-DCFPyL with respect to 18F-Fluorocholine in initial staging of intermediate-/high-risk prostate cancer (PCa).
Material and methods
Patients with recent diagnosis of intermediate-/high-risk PCa without androgen deprivation therapy and previous 18F-Fluorocholine-PET/CT (negative for extraprostatic disease or with oligometastatic disease) were referred to 18F-DCFPyL-PET/CT. Patients’ disease characteristic as grade group, D’Amico risk category (intermediate/high), prostate-specific antigen (PSA) closest to PET/CTs and its kinetics were obtained. The overall detection rate (DR) and molecular imaging TNM (miTNM) stage according to the prostate cancer molecular imaging standardized evaluation (PROMISE) criteria were assessed for both radiotracers, and their concordance (Kappa coefficient) was analyzed. The diagnostic and therapeutic impact of 18F-DCFPyL with respect to 18F-Fluorocholine was evaluated.
Results
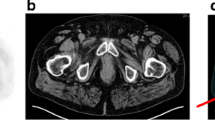
Fifty-eight patients were analyzed (84.5% high-risk). 18F-Fluorocholine showed a higher DR than 18F-DCFPyL of prostate gland involvement (100% versus 93.1%) and pelvic node disease (37.9% versus 31%; k = 0.436, p = 0.001). On the other hand, 18F-DCFPyL-PET/CT showed a higher DR of metastatic disease than 18F-Fluorocholine-PET/CT, 9/58 patients (15.5%): 3 M1a, 5 M1b and 1 M1c) versus 5/58 (8.6%) patients: 1 M1a and 4 M1b), k = 0.426; p = 0.001. No significant association was found between clinical characteristics (grade group, risk category, PSA level and kinetic) and 18F-Fluorocholine or 18F-DCFPyL results. The results of 18F-DCFPyL-PET/CT modified the previously planned treatment compared to 18F-Fluorocholine-PET/CT in 13 patients (22.4%).
Conclusions
18F-Fluorocholine and 18F-DCFPyL PET/CT showed a similar DR of prostate gland and lymph node involvement, although with moderate concordance for the latter. 18F-DCFPyL was superior to 18F-Fluorocholine in detecting regional and distant metastasis with a therapeutic impact in one of every five patients.






Similar content being viewed by others
References
Klingenberg S, Jochumsen MR, Ulhøi BP, Fredsøe J, Sørensen KD, Borre M, et al. 68Ga-PSMA PET/CT for primary lymph node and distant metastasis NM staging of high-risk prostate cancer. J Nucl Med. 2021;62(2):214–20.
Mottet N, Cornford P, Van den Bergh RCN, Briers E, De Santis M, Gillessen S, et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG Guidelines on Prostate cancer. Eur Urol [internet] 2022. https://uroweb.org/guidelines/prostate-cancer/chapter/citation-information.
Evangelista L, Cimitan M, Zattoni F, Gutilla A, Zattoni F, Saladini G. Comparison between conventional imaging (abdominal-pelvic computed tomography and bone scan) and [(18)F] Choline positron emission tomography/computed tomography imaging for the initial staging of patients with intermediate- to high-risk prostate cancer: a retrospective analysis. Scand J Urol. 2015;49(5):345–53.
Poulsen MH, Petersen H, Hoilund-Carlsen PF, Jakobsen JS, Gerke O, Karstoft J, et al. Spine metastases in prostate cancer: comparison of technetium-99m-MDP whole-body bone scintigraphy, [18F] Choline positron emission tomography (PET)/computed tomography (CT) and [18F]NaF PET/CT. BJU Int. 2014;114(6):818–23.
Pinaquy JB, De Clermont-Galleran H, Pasticier G, Rigou G, Alberti N, Hindie E, et al. Comparative effectiveness of 18F-fluorocholine PET-CT and pelvic MRI with diffusion-weighted imaging for staging in patients with high-risk prostate cancer. Prostate. 2015;75(3):323–31.
Vag T, Heck MM, Beer AJ, Souvatzoglou M, Weirich G, Holzapfel K, et al. Preoperative lymph node staging in patients with primary prostate cancer: comparison and correlation of quantitative imaging parameters in diffusion-weighted imaging and 11C-choline PET/CT. Eur Radiol. 2014;24(8):1821–6.
Lisney AR, Leitsmann C, Strauß A, Meller B, Bucerius JA, Sahlmann CO. The role of PSMA PET/CT in the primary diagnosis and follow-up of prostate cancer-a practical clinical review. Cancers (Basel). 2022;14(15):36–8.
Petersen LJ, Zacho HD. PSMA PET for primary lymph node staging of intermediate and high-risk prostate cancer: an expedited systematic review. Cancer Imaging. 2020;20(1):10.
Kim SJ, Lee SW, Ha HK. Diagnostic performance of radiolabeled prostate-specific membrane antigen positron emission tomography/computed tomography for primary lymph node staging in newly diagnosed intermediate to high-risk prostate cancer patients: a systematic review and meta-analysis. Urol Int. 2019;102(1):27–36.
Jansen BHE, Bodar YJL, Zwezerijnen GJC, Meijer D, van der Voorn JP, Nieuwenhuijzen JA, et al. Pelvic lymph-node staging with 18F-DCFPyL PET/CT prior to extended pelvic lymph-node dissection in primary prostate cancer—the SALT trial. Eur J Nucl Med Mol Imaging. 2021;48(2):509–20.
Pienta KJ, Gorin MA, Rowe SP, Carroll PR, Pouliot F, Probst S, et al. A phase 2/3 prospective multicenter study of the diagnostic accuracy of prostate specific membrane antigen PET/CT with 18F-DCFPyL in prostate cancer patients (OSPREY). J Urol. 2021;206(1):52–61.
Anttinen M, Ettala O, Malaspina S, Jambor I, Sandell M, Kajander S, et al. A prospective comparison of 18F-prostate-specific membrane antigen-1007 positron emission tomography computed tomography, whole-body 1.5 T magnetic resonance imaging with diffusion-weighted imaging, and single-photon emission computed tomography/computed tomography with traditional imaging in primary distant metastasis staging of prostate cancer (PROSTAGE). Eur Urol Oncol. 2021;4(4):635–44.
Donswijk ML, van Leeuwen PJ, Vegt E, Cheung Z, Heijmink SWTPJ, van der Poel HG, et al. Clinical impact of PSMA PET/CT in primary prostate cancer compared to conventional nodal and distant staging: a retrospective single center study. BMC Cancer. 2020;20(1):723.
Eastham JA, Auffenberg GB, Barocas DA, Chou R, Crispino T, Davis JW, et al. Clinically localized prostate cancer: AUA/ASTRO guideline part I: introduction, risk assessment, staging and risk-based management. J Urol. 2022;208(1):10–8.
Schwenck J, Rempp H, Reischl G, Kruck S, Stenzl A, Nikolaou K, et al. Comparison of 68Ga-labelled PSMA-11 and 11C-choline in the detection of prostate cancer metastases by PET/CT. Eur J Nucl Med Mol Imaging. 2017;44(1):92–101.
Umbehr MH, Muntener M, Hany T, Sulser T, Bachmann LM. The role of 11C-choline and 18F-fluorocholine positron emission tomography (PET) and PET/CT in prostate cancer: a systematic review and meta-analysis. Eur Urol. 2013;64(1):106–17.
Rowe S, Gorin M, Pienta K, Siegel B, Carroll P, Pouliot F, et al. Results from the OSPREY trial: a prospective phase 2/3 multi-center study of 18F-DCFPyL PET/CT imaging in patients with prostate cancer—examination of diagnostic accuracy. J Nucl Med. 2019;60(Sup 1):586.
D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA, et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA. 1998;280(11):969–74.
Sheikhbahaei S, Werner AW, Solnes LB, Pienta KJ, Pomper MG, Gorin MA, et al. Prostate-specific membrane antigen (PSMA)-targeted PET imaging of prostate cancer: an update on important pitfalls. Semin Nucl Med. 2019;49(4):255–70.
Eiber M, Herrmann K, Calais J, Hadaschik B, Giesel FL, Hartenbach M, et al. Prostate cancer molecular imaging standardized evaluation (PROMISE): proposed miTNM classification for the interpretation of PSMA-Ligand PET/CT. J Nucl Med. 2018;59(3):469–78.
Joniau S, Van den Bergh L, Lerut E, Deroose CM, Haustermans K, Oyen R, et al. Mapping of pelvic lymph node metastases in prostate cancer. Eur Urol. 2013;63(3):450–8.
Paño B, Sebastià C, Buñesch L, Mestres J, Salvador R, Macías NG, et al. Pathways of lymphatic spread in male urogenital pelvic malignancies. Radiographics. 2011;31(1):135–60.
Evangelista L, Guttilla A, Zattoni F, Muzzio PC, Zattoni F. Utility of choline positron emission tomography/computed tomography for lymph node involvement identification in intermediate- to high-risk prostate cancer: a systematic literature review and meta-analysis. Eur Urol. 2013;63(6):1040–8.
Puterman C, Bjöersdorff M, Amidi J, Anand A, Soller W, Jiborn T, et al. A retrospective study assessing the accuracy of [18F]-fluorocholine PET/CT for primary staging of lymph node metastases in intermediate and high-risk prostate cancer patients undergoing robotic-assisted laparoscopic prostatectomy with extended lymph node dissection. Scand J Urol. 2021;55(4):293–7.
Gorin MA, Rowe SP, Patel HD, Vidal I, Mana-Ay M, Javadi MS, et al. Prostate specific membrane antigen targeted 18F-DCFPyL positron emission tomography/computerized tomography for the preoperative staging of high-risk prostate cancer: results of a prospective, phase II, single center study. J Urol. 2018;199(1):126–32.
Hofman MS, Lawrentschuk N, Francis RJ, Tang C, Vela I, Thomas P, et al. Prostate specific membrane antigen PET-CT in patients with high risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicenter study. Lancet. 2020;395(10231):1208–16.
Stabile A, Pellegrino A, Mazzone E, Cannoletta D, de Angelis M, Barletta F, et al. Can negative prostate-specific membrane antigen positron emission tomography/computed tomography avoid the need for pelvic lymph node dissection in newly diagnosed prostate cancer patients? A systematic review and meta-analysis with backup histology as reference standard. Eur Urol Oncol. 2022;5(1):1–17.
Martorana G, Schiavina R, Corti B, Farsad M, Salizzoni E, Brunocilla E, et al. 11C-choline positron emission tomography/computerized tomography for tumor localization of primary prostate cancer in comparison with 12-core biopsy. J Urol. 2006;176(3):954–60.
Rinnab L, Blumstein NM, Mottaghy FM, Hautmann RE, Küfer R, Hohl K, et al. 11C-choline positron-emission tomography/computed tomography and transrectal ultrasonography for staging localized prostate cancer. BJU Int. 2007;99(6):1421–6.
De Bari B, Alongi F, Lestrade L, Giammarile F. Choline-PET in prostate cancer management: the point of view of the radiation oncologist. Crit Rev Oncol Hematol. 2014;91(3):234–47.
Hartenbach M, Hartenbach S, Bechtloff W, Danz B, Kraft K, Klemenz B, et al. Combined PET/MRI improves diagnostic accuracy in patients with prostate cancer: a prospective diagnostic trial. Clin Cancer Res. 2014;20(12):3244–53.
Farsad M, Schiavina R, Castellucci P, Nanni C, Corti B, Martorana G, et al. Detection and localization of prostate cancer: correlation of (11) C-choline PET/CT with histopathologic step-section analysis. J Nucl Med. 2005;46(10):1642–9.
Van Waarde A, Jager PL, Ishiwata K, Dierckx RA, Elsinga PH. Comparison of sigma-ligands and metabolic PET tracers for differentiating tumor from inflammation. J Nucl Med. 2006;47(1):150–4.
Mannweiler S, Amersdorfer P, Trajanoski S, Terrett JA, King D, Mehes G. Heterogeneity of prostate-specific membrane antigen (PSMA) expression in prostate carcinoma with distant metastasis. Pathol Oncol Res. 2009;15(2):167–72.
Huang Y-T, Tseng N-C, Chen Y-K, Huang K-H, Lin H-Y, Huang Y-Y, et al. The detection performance of 18F–prostate-specific membrane antigen-1007 PET/CT in primary prostate cancer. A systemic review and meta-analysis. Clin Nucl Med. 2022;47(9):755–62.
De Feria Cardet RE, Hofman MS, Segard T, Yim J, Williams S, Francis RJ, et al. Is prostate-specific membrane antigen positron emission tomography/computed tomography imaging cost-effective in prostate cancer: an analysis informed by the proPSMA trial. Eur Urol. 2021;79(3):413–8.
Wondergem M, van der Zant FM, Broos WAM, Roeleveld TA, Donker R, Ten Oever D, et al. 18F-DCFPyL PET/CT for primary staging in 160 high-risk prostate cancer patients; metastasis detection rate, influence on clinical management and preliminary results of treatment efficacy. Eur J Nucl Med Mol Imaging. 2021;48(2):521–31.
Funding
The authors declare that there is no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Study was approved by a reference Ethical Committee (internal code of 2022-53).
Consent for participating and for publication
Patients signed an informed consent to participate and to use their anonymous data for analysis and publication of results.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lucas Lucas, C., García Zoghby, L., Amo-Salas, M. et al. Diagnostic and therapeutic impact of PET/CT with 18F-DCFPyL versus 18F-Fluorocholine in initial staging of intermediate-/high-risk prostate cancer: a pilot study. Ann Nucl Med 37, 551–560 (2023). https://doi.org/10.1007/s12149-023-01859-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-023-01859-4