Abstract
Purpose
To establish and validate a regional lymph node (LN) metastasis prediction model of colorectal cancer (CRC) based on 18F-FDG PET/CT and radiomic features using machine-learning methods.
Methods
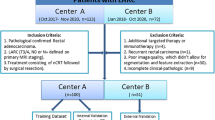
A total of 199 colorectal cancer patients underwent pre-therapy diagnostic 18F-FDG PET/CT scans and CRC radical surgery. The Chang-Gung Image Texture Analysis toolbox (CGITA) was used to extract 70 PET radiomic features reflecting 18F-FDG uptake heterogeneity of tumors. The least absolute shrinkage and selection operator (LASSO) algorithm was used to select radiomic features and develop a radiomic signature score (Rad-score). The training set was used to establish five machine-learning prediction models and the test set was used to test the efficacy of the models. The effectiveness of the models was compared by ROC analysis.
Results
The CRC patients were divided into a training set (n = 144) and a test set (n = 55). Two radiomic features were selected to build the Rad-score. Five machine-learning algorithms including logistic regression, support vector machine (SVM), random forest, neural network and eXtreme gradient boosting (XGBoost) were used to established models. Among the five machine-learning models, logistic regression (AUC 0.866, 95% CI 0.808–0.925) and XGBoost (AUC 0.903, 95% CI 0.855–0.951) models performed the best. In the training set, the AUC of these two models were significantly higher than that of the LN metastasis status reported by 18F-FDG PET/CT for differentiating positive and negative regional LN metastases in CRC (all p < 0.05). Good efficacy of the above two models was also achieved in the test set. We created a nomogram based on the logistic regression model that visualized the results and provided an easy-to-use method for predicting regional LN metastasis in patients with CRC.
Conclusion
In this study, five machine-learning models for preoperative prediction of regional LN metastasis of CRC based on 18F-FDG PET/CT and PET-based radiomic features were successfully developed and validated. Among them, the logistic regression and XGBoost models performed the best, with higher efficacy than 18F-FDG PET/CT in both the training and test sets.







Similar content being viewed by others
Code availability
The code in this study is written in R (https://www.r-project.org/).
Abbreviations
- AUC:
-
Area under the curve
- CEA:
-
Carcinoembryonic antigen
- CGITA:
-
Chang-Gung Image Texture Analysis toolbox
- CI:
-
Confidence interval
- CRC:
-
Colorectal cancer
- DICOM:
-
Digital imaging and communications in medicine
- 18F-FDG:
-
18F-Fluorodeoxy Glucose
- LASSO:
-
Least absolute shrinkage and selection operator
- LN:
-
Lymph node
- NCCN:
-
National Comprehensive Cancer Network
- NPV:
-
Negative predictive value
- MBq:
-
Megabecquerel
- MC:
-
Mean convergence
- PET/CT:
-
Positron Emission/Computed Tomography
- PPV:
-
Positive predictive value
- ROC:
-
Receiver-operating characteristic
- ROI:
-
Region of interest
- SD:
-
Standard deviation
- SE:
-
Standard error
- SUV:
-
Standardized uptake value
- SVM:
-
Support vector machine
- TFC:
-
Texture feature coding
- XGBoost:
-
eXtreme gradient boosting
References
Haggar FA, Boushey RP. Colorectal cancer epidemiology: incidence, mortality, survival, and risk factors. Clin Colon Rectal Surg. 2009;22(4):191–7. https://doi.org/10.1055/s-0029-1242458.
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2007. CA Cancer J Clin. 2007;57(1):43–66. https://doi.org/10.3322/canjclin.57.1.43.
Chang GJ, Rodriguez-Bigas MA, Skibber JM, et al. Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst. 2007;99(6):433–41. https://doi.org/10.1093/jnci/djk092.
Benson AB, Venook AP, Al-Hawary MM, et al. NCCN guidelines insights: colon cancer, version 2.2018. J Natl Compr Cancer Netw. 2018;16(4):359–69. https://doi.org/10.6004/jnccn.2018.0021.
Wright FC, Law CH, Berry S, et al. Clinically important aspects of lymph node assessment in colon cancer. J Surg Oncol. 2009;99(4):248–55. https://doi.org/10.1002/jso.21226.
Smith AJ, Driman DK, Spithoff K, Hunter A, et al. Guideline for optimization of colorectal cancer surgery and pathology. J Surg Oncol. 2010;101(1):5–12. https://doi.org/10.1002/jso.21395.
Tsunoda Y, Ito M, Fujii H, et al. Preoperative diagnosis of lymph node metastases of colorectal cancer by FDG-PET/CT. Jpn J Clin Oncol. 2008;38(5):347–53. https://doi.org/10.1093/jjco/hyn032 (Epub 2008 Apr 19).
Basu S, Kwee TC, Gatenby R, et al. Evolving role of molecular imaging with PET in detecting and characterizing heterogeneity of cancer tissue at the primary and metastatic sites, a plausible explanation for failed attempts to cure malignant disorders. Eur J Nucl Med Mol Imaging. 2011;38(6):987–91. https://doi.org/10.1007/s00259-011-1787-z.
Visvikis D, Hatt M, Tixier F, et al. The age of reason for FDG PET image-derived indices. Eur J Nucl Med Mol Imaging. 2012;39(11):1670–2. https://doi.org/10.1007/s00259-012-2239-0.
O’Connor JP, Rose CJ, Waterton JC, et al. Imaging intratumor heterogeneity: role in therapy response, resistance, and clinical outcome. Clin Cancer Res. 2015;21(2):249–57. https://doi.org/10.1158/1078-0432.CCR-14-0990.
Weber WA, Schwaiger M, Avril N. Quantitative assessment of tumor metabolism using FDG-PET imaging. Nucl Med Biol. 2000;27(7):683–7. https://doi.org/10.1016/s0969-8051(00)00141-4.
Tixier F, Hatt M, Le Rest CC, et al. Signaling pathways alteration involved in head and neck cancer can be identified through textural features analysis in 18F-FDG PET images: a prospective study. J Nucl Med. 2015;56:3–3.
Erickson BJ, Korfiatis P, Akkus Z, et al. Machine learning for medical imaging. Radiographics. 2017;37(2):505–15. https://doi.org/10.1148/rg.2017160130.
Tixier F, Le Rest CC, Hatt M, et al. Intratumor heterogeneity characterized by textural features on baseline 18F-FDG PET images predicts response to concomitant radiochemotherapy in esophageal cancer. J Nucl Med. 2011;52(3):369–78. https://doi.org/10.2967/jnumed.110.082404.
Cheng NM, Fang YH, Lee LY, et al. Zone-size nonuniformity of 18F-FDG PET regional textural features predicts survival in patients with oropharyngeal cancer. Eur J Nucl Med Mol Imaging. 2015;42(3):419–28. https://doi.org/10.1007/s00259-014-2933-1.
Hyun SH, Kim HS, Choi SH, et al. Intratumoral heterogeneity of (18)F-FDG uptake predicts survival in patients with pancreatic ductal adenocarcinoma. Eur J Nucl Med Mol Imaging. 2016;43(8):1461–8. https://doi.org/10.1007/s00259-016-3316-6.
Yip SS, Kim J, Coroller TP, et al. Associations between somatic mutations and metabolic imaging phenotypes in non-small cell lung cancer. J Nucl Med. 2017;58(4):569–76. https://doi.org/10.2967/jnumed.116.181826.
Cook GJR, Lovat E, Siddique M, et al. Characterisation of malignant peripheral nerve sheath tumours in neurofibromatosis-1 using heterogeneity analysis of 18F-FDG PET. Eur J Nucl Med Mol Imaging. 2017;44(11):1845–52. https://doi.org/10.1007/s00259-017-3733-1.
Lovinfosse P, Koopmansch B, Lambert F, et al. (18)F-FDG PET/CT imaging in rectal cancer: relationship with the RAS mutational status. Br J Radiol. 2016;89(1063):20160212. https://doi.org/10.1259/bjr.20160212.
Bundschuh RA, Dinges J, Neumann L, et al. Textural parameters of tumor heterogeneity in 18F-FDG PET/CT for therapy response assessment and prognosis in patients with locally advanced rectal cancer. J Nucl Med. 2014;55(6):891–7. https://doi.org/10.2967/jnumed.113.127340.
Nakajo M, Kajiya Y, Tani A, et al. A pilot study for texture analysis of 18F-FDG and 18F-FLT-PET/CT to predict tumor recurrence of patients with colorectal cancer who received surgery. Eur J Nucl Med Mol Imaging. 2017;44(13):2158–68. https://doi.org/10.1007/s00259-017-3787-0.
Brooks FJ, Grigsby PW. The effect of small tumor volumes on studies of intratumoral heterogeneity of tracer uptake. J Nucl Med. 2014;55(1):37–42. https://doi.org/10.2967/jnumed.112.116715.
Hohenberger W, Weber K, Matzel K, et al. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation–technical notes and outcome. Colorectal Dis. 2009;11(4):354–64. https://doi.org/10.1111/j.1463-1318.2008.01735.x (discussion 364-5).
Dighe S, Swift I, Brown G. CT staging of colon cancer. Clin Radiol. 2008;63(12):1372–9. https://doi.org/10.1016/j.crad.2008.04.021.
Lu YY, Chen JH, Ding HJ, Chien CR, Lin WY, Kao CH. A systematic review and meta-analysis of pretherapeutic lymph node staging of colorectal cancer by 18F-FDG PET or PET/CT. Nucl Med Commun. 2012;33(11):1127–33. https://doi.org/10.1097/MNM.0b013e328357b2d9.
Fang YH, Lin CY, Shih MJ, et al. Development and evaluation of an open-source software package “CGITA” for quantifying tumor heterogeneity with molecular images. Biomed Res Int. 2014;2014:248505. https://doi.org/10.1155/2014/248505.
Wada H, Shiozawa M, Katayama K, et al. Systematic review and meta-analysis of histopathological predictive factors for lymph node metastasis in T1 colorectal cancer. J Gastroenterol. 2015;50(7):727–34. https://doi.org/10.1007/s00535-015-1057-0.
Han EC, Kwon YH, Park KJ, et al. Significance of lymph node metastasis in the survival of stage IV colorectal cancer by hematogenous metastasis. Ann Surg Treat Res. 2018;95(4):201–12. https://doi.org/10.4174/astr.2018.95.4.201.
Lu YY, Chen JH, Ding HJ, et al. A systematic review and meta-analysis of pretherapeutic lymph node staging of colorectal cancer by 18F-FDG PET or PET/CT. Nucl Med Commun. 2012;33(11):1127–33. https://doi.org/10.1097/MNM.0b013e328357b2d9.
Sasaki K, Kawasaki H, Sato M, et al. Impact of fluorine-18 2-fluoro-2-deoxy-D-glucose uptake on preoperative positron emission tomography/computed tomography in the lymph nodes of patients with primary colorectal cancer. Dig Surg. 2017;34(1):60–7. https://doi.org/10.1159/000448222.
Horng MH, Sun YN, Lin XZ. Texture feature coding method for classification of liver sonography. Comput Med Imaging Graph. 2002;26(1):33–42. https://doi.org/10.1016/s0895-6111(01)00029-5.
Chen SW, Shen WC, Chen WT, et al. Metabolic imaging phenotype using radiomics of [18F]FDG PET/CT associated with genetic alterations of colorectal cancer. Mol Imaging Biol. 2019;21(1):183–90. https://doi.org/10.1007/s11307-018-1225-8.
Burton S, Brown G, Bees N, et al. Accuracy of CT prediction of poor prognostic features in colonic cancer. Br J Radiol. 2008;81(961):10–9. https://doi.org/10.1259/bjr/19492531.
Huang YQ, Liang CH, He L, et al. Development and validation of a radiomics nomogram for preoperative prediction of lymph node metastasis in colorectal cancer. J Clin Oncol. 2016;34(18):2157–64. https://doi.org/10.1200/JCO.2015.65.9128.
Toiyama Y, Inoue Y, Shimura T, et al. Serum angiopoietin-like protein 2 improves preoperative detection of lymph node metastasis in colorectal cancer. Anticancer Res. 2015;35(5):2849–56.
Smith AJ, Driman DK, Spithoff K, et al. Guideline for optimization of colorectal cancer surgery and pathology. J Surg Oncol. 2010;101(1):5–12. https://doi.org/10.1002/jso.21395.
Acknowledgements
The authors appreciate all the staff of the PET Center of Nanfang Hospital of Southern Medical University for their important help in this study.
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All the procedures performed in the study and involving human participants were carried out in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate
Yes.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
He, J., Wang, Q., Zhang, Y. et al. Preoperative prediction of regional lymph node metastasis of colorectal cancer based on 18F-FDG PET/CT and machine learning. Ann Nucl Med 35, 617–627 (2021). https://doi.org/10.1007/s12149-021-01605-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-021-01605-8