Abstract
Objective
The aim of the study was to retrospectively evaluate the recurrence detection rate of Gallium-68-prostate-specific membrane antigen [(68Ga)PSMA] imaging and therapy (I&T) positron emission tomography/computed tomography (PET/CT) at different PSA levels, which enables early detection of patients with radical prostatectomy. We also aimed to compare Gleason scores, used drugs (LHRH analogs and antiandrogens), PSA levels with SUVmax values, and detection rates.
Method
This retrospective study included 107 patients who underwent radical prostatectomy and who underwent (68Ga)PSMA I&T PET/CT imaging between January 2015 and December 2018 for the early detection of recurrence. The PSA values, Gleason scores, treatments, lesions detected on (68Ga)PSMA I&T, and SUVmax values were recorded for all patients.
Results
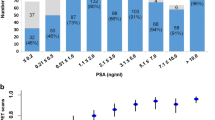
Patients with a median PSA level of 1.22 ng/mL were divided into seven groups according to the PSA values. The lowest lesion detection rate was found to be 7/16 patients (43.8%) when the PSA was < 0.2 ng/ml, and the highest lesion detection rate was found to be 33/33 patients (100%) when the PSA was > 3.5 ng/ml. There was a positive correlation between PSA level and ppSUVmax (per patient SUVmax) value of the patients with lesions (p < 0.001 and r = 0.49). As the Gleason score increased, the lesion detection rates also increased and there was a significant correlation between these values (p < 0.001 and r = 0.360). A positive correlation was determined between the Gleason scores and ppSUVmax values in patients with lesions (p = 0.007 and r = 0.302). A statistically significant correlation was found between bicalutamide use and lesion detection on (68Ga)PSMA I&T (p < 0.001). A similar relationship was also determined in patients undergoing maximal androgen blockade (MAB) (p = 0.003). Patients determined with lesions on (68Ga)PSMA I&T and who were administered luteinizing hormone-releasing hormone (LHRH) agonists were found to have statistically significantly higher ppSUVmax values than those who were not administered LHRH agonists (p < 0.001). In binary logistic regression test, when PSA levels and Gleason scores were selected as continuous variables, both PSA levels and Gleason scores were demonstrated as significant covariates (p = 0.006 and p = 0.022) for lesion detection; by contrast, bicalutamide and MAB were not found as significant factors.
Conclusion
In the present study, (68Ga)PSMA I&T was found to be quite successful in determining lesions in the biochemical recurrence, which is consistent with the findings of other I&T studies and studies conducted with different PSMA ligands. Thus, it can be considered that the use of (68Ga)PSMA I&T will become increasingly common.



Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86.
Tourinho-Barbosa R, Srougi V, Nunes-Silva I, Baghdadi M, Rembeyo G, Eiffel SS, et al. Biochemical recurrence after radical prostatectomy: what does it mean? Int Braz J Urol. 2018;44:14–21.
Freedland SJ, Humphreys EB, Mangold LA, Eisenberger M, Dorey FJ, Walsh PC, et al. Risk of prostate cancer-specific mortality following biochemical recurrence after radical prostatectomy. JAMA. 2005;294:433–9.
Kupelian PA, Mahadevan A, Reddy CA, Reuther AM, Klein EA. Use of different definitions of biochemical failure after external beam radiotherapy changes conclusions about relative treatment efficacy for localized prostate cancer. Urology. 2006;68:593–8.
Roehl KA, Han M, Ramos CG, Antenor JA, Catalona WJ. Cancer progression and survival rates following anatomical radical retropubic prostatectomy in 3478 consecutive patients: long-term results. J Urol. 2004;172:910–4.
Darwish OM, Raj GV. Management of biochemical recurrence after primary localized therapy for prostate cancer. Front Oncol. 2012;2:48.
Paller CJ, Antonarakis ES. Management of biochemically recurrent prostate cancer after local therapy: evolving standards of care and new directions. Clin Adv Hematol Oncol. 2013;11:14–23.
Krause BJ, Souvatzoglou M, Tuncel M, Herrmann K, Buck AK, Praus C, et al. The detection rate of [11C]choline-PET/CT depends on the serum PSA value in patients with biochemical recurrence of prostate cancer. Eur J Nuclear Med Mol Imaging. 2008;35:18–23.
Castellucci P, Picchio M. 11C-choline PET/CT and PSA kinetics. Eur J Nuclear Med Mol Imaging. 2013;40(Suppl 1):S36–40.
Graute V, Jansen N, Ubleis C, Seitz M, Hartenbach M, Scherr MK, et al. Relationship between PSA kinetics and [18F]fluorocholine PET/CT detection rates of recurrence in patients with prostate cancer after total prostatectomy. Eur J Nuclear Med Mol Imaging. 2012;39:271–82.
Wright GL Jr, Haley C, Beckett ML, Schellhammer PF. Expression of prostate-specific membrane antigen in normal, benign, and malignant prostate tissues. Urol Oncol. 1995;1:18–28.
Eder M, Schafer M, Bauder-Wust U, Hull WE, Wangler C, Mier W, et al. 68 Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem. 2012;23:688–97.
Afshar-Oromieh A, Malcher A, Eder M, Eisenhut M, Linhart HG, Hadaschik BA, et al. PET imaging with a [68 Ga]gallium-labelled PSMA ligand for the diagnosis of prostate cancer: biodistribution in humans and first evaluation of tumour lesions. Eur J Nuclear Med Mol Imaging. 2013;40:486–95.
Perera M, Papa N, Christidis D, Wetherell D, Hofman MS, Murphy DG, et al. Sensitivity, specificity, and predictors of positive 68Ga-prostate-specific membrane antigen positron emission tomography in advanced prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016;70:926–37.
von Eyben FE, Picchio M, von Eyben R, Rhee H, Bauman G. 68Ga-labeled prostate-specific membrane antigen ligand positron emission tomography/computed tomography for prostate cancer: a systematic review and meta-analysis. Eur Urol Focus. 2018;4:686–93.
Corfield J, Perera M, Bolton D, Lawrentschuk N. (68)Ga-prostate specific membrane antigen (PSMA) positron emission tomography (PET) for primary staging of high-risk prostate cancer: a systematic review. World J Urol. 2018;36:519–27.
Zacho HD, Nielsen JB, Haberkorn U, Stenholt L, Petersen LJ. (68)Ga-PSMA PET/CT for the detection of bone metastases in prostate cancer: a systematic review of the published literature. Clin Physiol Funct Imaging. 2017. https://doi.org/10.1111/cpf.12480.
Lavalaye J, Kaldeway P, van Melick HH. Diffuse bone metastases on (68)Ga-PSMA PET-CT in a patient with prostate cancer and normal bone scan. Eur J Nuclear Med Mol Imaging. 2016;43:1563–4.
Pyka T, Okamoto S, Dahlbender M, Tauber R, Retz M, Heck M, et al. Comparison of bone scintigraphy and (68)Ga-PSMA PET for skeletal staging in prostate cancer. Eur J Nuclear Med Mol Imaging. 2016;43:2114–21.
Afshar-Oromieh A, Hetzheim H, Kratochwil C, Benesova M, Eder M, Neels OC, et al. The theranostic PSMA ligand PSMA-617 in the diagnosis of prostate cancer by PET/CT: biodistribution in humans, radiation dosimetry, and first evaluation of tumor lesions. J Nucl Med. 2015;56:1697–705.
Weineisen M, Schottelius M, Simecek J, Baum RP, Yildiz A, Beykan S, et al. 68 Ga- and 177Lu-labeled PSMA I&T: optimization of a PSMA-targeted theranostic concept and first proof-of-concept human studies. J Nucl Med. 2015;56:1169–76.
Baum RP, Kulkarni HR, Schuchardt C, Singh A, Wirtz M, Wiessalla S, et al. 177Lu-labeled prostate-specific membrane antigen radioligand therapy of metastatic castration-resistant prostate cancer: safety and efficacy. J Nuclear Med. 2016;57:1006–13.
McCarthy M, Langton T, Kumar D, Campbell A. Comparison of PSMA-HBED and PSMA-I&T as diagnostic agents in prostate carcinoma. Eur J Nuclear Med Mol Imaging. 2017;44:1455–62.
Berliner C, Tienken M, Frenzel T, Kobayashi Y, Helberg A, Kirchner U, et al. Detection rate of PET/CT in patients with biochemical relapse of prostate cancer using [(68)Ga]PSMA I&T and comparison with published data of [(68)Ga]PSMA HBED-CC. Eur J Nuclear Med Mol Imaging. 2017;44:670–7.
Bluemel C, Krebs M, Polat B, Linke F, Eiber M, Samnick S, et al. 68 Ga-PSMA-PET/CT in patients with biochemical prostate cancer recurrence and negative 18F-Choline-PET/CT. Clin Nucl Med. 2016;41:515–21.
Weineisen M, Simecek J, Schottelius M, et al. Synthesis and preclinical evaluation of DOTAGA-conjugated PSMA ligands for functional imaging and endoradiotherapy of prostate cancer. EJNMMI Res. 2014;4:63.
Martin R, Juttler S, Muller M, Wester HJ. Cationic eluate pretreatment for automated synthesis of [(6)(8)Ga]CPCR4.2. Nucl Med Biol. 2014;41:84–9.
Hijazi S, Meller B, Leitsmann C, Strauss A, Ritter C, Lotz J, et al. See the unseen: mesorectal lymph node metastases in prostate cancer. Prostate. 2016;76:776–80.
Hijazi S, Meller B, Leitsmann C, Strauss A, Meller J, Ritter CO, et al. Pelvic lymph node dissection for nodal oligometastatic prostate cancer detected by 68 Ga-PSMA-positron emission tomography/computerized tomography. Prostate. 2015;75:1934–40.
Henkenberens C, Vonk CA, Ross TL, Bengel FM, Wester HJ, Katja H, et al. (68)Ga-PSMA ligand PET/CT-based radiotherapy for lymph node relapse of prostate cancer after primary therapy delays initiation of systemic therapy. Anticancer Res. 2017;37:1273–9.
Schmuck S, Nordlohne S, von Klot CA, Henkenberens C, Sohns JM, Christiansen H, et al. Comparison of standard and delayed imaging to improve the detection rate of [(68)Ga]PSMA I&T PET/CT in patients with biochemical recurrence or prostate-specific antigen persistence after primary therapy for prostate cancer. Eur J Nuclear Med Mol Imaging. 2017;44:960–8.
Afshar-Oromieh A, Holland-Letz T, Giesel FL, Kratochwil C, Mier W, Haufe S, et al. Diagnostic performance of (68)Ga-PSMA-11 (HBED-CC) PET/CT in patients with recurrent prostate cancer: evaluation in 1007 patients. Eur J Nuclear Med Mol Imaging. 2017;44:1258–68.
Afshar-Oromieh A, Zechmann CM, Malcher A, Eder M, Eisenhut M, Linhart HG, et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur J Nuclear Med Mol Imaging. 2014;41:11–20.
Uprimny C, Kroiss AS, Decristoforo C, Fritz J, von Guggenberg E, Kendler D, et al. (68)Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nuclear Med Mol Imaging. 2017;44:941–9.
Eiber M, Maurer T, Souvatzoglou M, Beer AJ, Ruffani A, Haller B, et al. Evaluation of hybrid (6)(8)Ga-PSMA ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56:668–74.
Afshar-Oromieh A, Avtzi E, Giesel FL, Holland-Letz T, Linhart HG, Eder M, et al. The diagnostic value of PET/CT imaging with the (68)Ga-labelled PSMA ligand HBED-CC in the diagnosis of recurrent prostate cancer. Eur J Nuclear Med Mol Imaging. 2015;42:197–209.
Wright GL Jr, Grob BM, Haley C, Grossman K, Newhall K, Petrylak D, et al. Upregulation of prostate-specific membrane antigen after androgen-deprivation therapy. Urology. 1996;48:326–34.
Evans MJ, Smith-Jones PM, Wongvipat J, Navarro V, Kim S, Bander NH, et al. Noninvasive measurement of androgen receptor signaling with a positron-emitting radiopharmaceutical that targets prostate-specific membrane antigen. Proc Natl Acad Sci USA. 2011;108:9578–82.
Liu T, Wu LY, Fulton MD, Johnson JM, Berkman CE. Prolonged androgen deprivation leads to downregulation of androgen receptor and prostate-specific membrane antigen in prostate cancer cells. Int J Oncol. 2012;41:2087–92.
Hope TA, Truillet C, Ehman EC, Afshar-Oromieh A, Aggarwal R, Ryan CJ, et al. 68 Ga-PSMA-11 PET imaging of response to androgen receptor inhibition: first human experience. J Nucl Med. 2017;58:81–4.
Marchal C, Redondo M, Padilla M, Caballero J, Rodrigo I, Garcia J, et al. Expression of prostate specific membrane antigen (PSMA) in prostatic adenocarcinoma and prostatic intraepithelial neoplasia. Histol Histopathol. 2004;19:715–8.
Valicenti RK, Thompson I Jr, Albertsen P, Davis BJ, Goldenberg SL, Wolf JS, et al. Adjuvant and salvage radiation therapy after prostatectomy: American Society for Radiation oncology/American urological association guidelines. Int J Radiat Oncol Biol Phys. 2013;86:822–8.
Acknowledgements
The research was not financially supported by grants or any other kind of funding from any pharmaceutical company or other possible sources. All the authors had an active involvement in data collection and analysis, as well as writing, preparing and reviewing the manuscript.
Funding
There is no funding resource supporting the work submitted.
Author information
Authors and Affiliations
Contributions
UY, KH, CC, and AS designed the study, analysed the data and contributed to the writing of the paper. UY and KH statistically analysed the data. All authors contributed to the writing of the paper. All the authors accept responsibility for the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yilmaz, U., Komek, H., Can, C. et al. The role of (68Ga)PSMA I&T in biochemical recurrence after radical prostatectomy: detection rate and the correlation between the level of PSA, Gleason score, and the SUVmax. Ann Nucl Med 33, 545–553 (2019). https://doi.org/10.1007/s12149-019-01360-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-019-01360-x