Abstract
Objectives
The aims of our study were to find the textural features on 18F-FDG PET/CT which reflect the different histological architectures between cervical cancer subtypes and to make a visual assessment of the association between 18F-FDG PET textural features in cervical cancer.
Methods
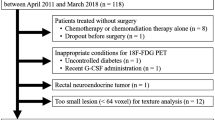
Eighty-three cervical cancer patients [62 squamous cell carcinomas (SCCs) and 21 non-SCCs (NSCCs)] who had undergone pretreatment 18F-FDG PET/CT were enrolled. A texture analysis was performed on PET/CT images, from which 18 PET radiomics features were extracted including first-order features such as standardized uptake value (SUV), metabolic tumor volume (MTV) and total lesion glycolysis (TLG), second- and high-order textural features using SUV histogram, normalized gray-level co-occurrence matrix (NGLCM), and neighborhood gray-tone difference matrix, respectively. These features were compared between SCC and NSCC using a Bonferroni adjusted P value threshold of 0.0028 (0.05/18). To assess the association between PET features, a heat map analysis with hierarchical clustering, one of the radiomics approaches, was performed.
Results
Among 18 PET features, correlation, a second-order textural feature derived from NGLCM, was a stable parameter and it was the only feature which showed a robust trend toward significant difference between SCC and NSCC. Cervical SCC showed a higher correlation (0.70 ± 0.07) than NSCC (0.64 ± 0.07, P = 0.0030). The other PET features did not show any significant differences between SCC and NSCC. A higher correlation in SCC might reflect higher structural integrity and stronger spatial/linear relationship of cancer cells compared with NSCC. A heat map with a PET feature dendrogram clearly showed 5 distinct clusters, where correlation belonged to a cluster including MTV and TLG. However, the association between correlation and MTV/TLG was not strong. Correlation was a relatively independent PET feature in cervical cancer.
Conclusions
18F-FDG PET textural features might reflect the differences in histological architecture between cervical cancer subtypes. PET radiomics approaches reveal the association between PET features and will be useful for finding a single feature or a combination of features leading to precise diagnoses, potential prognostic models, and effective therapeutic strategies.




Similar content being viewed by others
References
Galic V, Herzog TJ, Lewin SN, Neugut AI, Burke WM, Lu YS, et al. Prognostic significance of adenocarcinoma histology in women with cervical cancer. Gynecol Oncol. 2012;125:287–91.
Fujiwara K, Monk B, Devouassoux-Shisheboran M. Adenocarcinoma of the uterine cervix: why is it different? Curr Oncol Rep. 2014;16:416.
Sugawara Y, Eisbruch A, Kosuda S, Recker BE, Kison PV, Wahl RL. Evaluation of FDG PET in patients with cervical cancer. J Nucl Med. 1999;40:1125–31.
Wong TZ, Jones EL, Coleman RE. Positron emission tomography with 2-deoxy-2-[18F]fluoro-d-glucose for evaluating local and distant disease in patients with cervical cancer. Mol Imaging Biol. 2004;6:55–62.
Kidd EA, El Naqa I, Siegel BA, Dehdashti F, Grigsby PW. FDG-PET-based prognostic nomograms for locally advanced cervical cancer. Gynecol Oncol. 2012;127:136–40.
Lee YY, Choi CH, Kim CJ, Kang H, Kim TJ, Lee JW, et al. The prognostic significance of the SUVmax (maximum standardized uptake value for F-18 fluorodeoxyglucose) of the cervical tumor in PET imaging for early cervical cancer: preliminary results. GynecolOncol. 2009;115:65–8.
Chung HH, Nam BH, Kim JW, Kang KW, Park NH, Song YS, et al. Preoperative [18F]FDG PET/CT maximum standardized uptake value predicts recurrence of uterine cervical cancer. Eur J Nucl Med Mol Imaging. 2010;37:1467–73.
Chung HH, Kim JW, Han KH, Eo JS, Kang KW, Park NH, et al. Prognostic value of metabolic tumor volume measured by FDG-PET/CT in patients with cervical cancer. Gynecol Oncol. 2011;120:270–4.
Chou HH, Chang HP, Lai CH, Ng KK, Hsueh S, Wu TI, et al. 18F-FDG PET in stage IB/IIB cervical adenocarcinoma/adenosquamous carcinoma. Eur J Nucl Med Mol Imaging. 2010;37:728–35.
Yoo J, Choi JY, Moon SH, Bae DS, Park SB, Choe YS, et al. Prognostic significance of volume-based metabolic parameters in uterine cervical cancer determined using 18F-fluorodeoxyglucose positron emission tomography. Int J Gynecol Cancer. 2012;22:1226–33.
Rahman T, Tsujikawa T, Yamamoto M, Chino Y, Shinagawa A, Kurokawa T, et al. Different prognostic implications of 18F-FDG PET between histological subtypes in patients with cervical cancer. Medicine (Baltimore). 2016;95:e3017.
El Naqa I, Grigsby P, Apte A, Kidd E, Donnelly E, Khullar D, et al. Exploring feature-based approaches in PET images for predicting cancer treatment outcomes. Pattern Recogn. 2009;42:1162–71.
Chicklore S, Goh V, Siddique M, Roy A, Marsden PK, Cook GJ. Quantifying tumour heterogeneity in 18F-FDG PET/CT imaging by texture analysis. Eur J Nucl Med Mol Imaging. 2013;40:133–40.
Aerts HJ, Velazquez ER, Leijenaar RT, Parmar C, Grossmann P, Carvalho S, et al. Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun. 2014;5:4006.
Gillies RJ, Kinahan PE, Hricak H. Radiomics: images are more than pictures, they are data. Radiology. 2016;278:563–77.
Orlhac F, Soussan M, Chouahnia K, Martinod E, Buvat I. 18F-FDG PET-derived textural indices reflect tissue-specific uptake pattern in non-small cell lung cancer. PLoS One. 2015;10:e0145063.
Orlhac F, Thézé B, Soussan M, Boisgard R, Buvat I. Multiscale texture analysis: from 18F-FDG PET images to histologic images. J Nucl Med. 2016;57:1823–8.
Orlhac F, Nioche C, Soussan M, Buvat I. Understanding changes in tumor texture indices in PET: a comparison between visual assessment and index values in simulated and patient data. J Nucl Med. 2017;58:387–92.
Clausi DA. An analysis of co-occurrence texture statistics as a function of grey level quantization. Can J Rem Sens. 2002;28:45–62.
Haralick RM, Shanmugam K, Dinstein I. Textural features for image classification. IEEE Trans Syst Man Cybern. 1973;3:610–21.
Amadasun MR, King R. Textural features corresponding to textural properties. IEEE Trans Syst Man Cybern. 1989;19:1264–74.
Cheng NM, Fang YH, Chang JT, Huang CG, Tsan DL, Ng SH, et al. Textural features of pretreatment 18F-FDG PET/CT images: prognostic significance in patients with advanced T-stage oropharyngeal squamous cell carcinoma. J Nucl Med. 2013;54:1703–9.
Fang YH, Lin CY, Shih MJ, Wang HM, Ho TY, Liao CT, et al. Development and evaluation of an open-source software package “CGITA” for quantifying tumor heterogeneity with molecular images. Biomed Res Int. 2014;2014:248505.
Rahim MK, Kim SE, So H, Kim HJ, Cheon GJ, Lee ES, et al. Recent trends in PET image interpretations using volumetric and texture- based quantification methods in nuclear oncology. Nucl Med Mol Imaging. 2014;48:1–15.
Davnall F, Yip CS, Ljungqvist G, Selmi M, Ng F, Sanghera B, et al. Assessment of tumor heterogeneity: an emerging imaging tool for clinical practice? Insights Imaging. 2012;3:573–89.
Hatt M, Majdoub M, Vallières M, Tixier F, Le Rest CC, Groheux D, et al. 18F-FDG PET uptake characterization through texture analysis: investigating the complementary nature of heterogeneity and functional tumor volume in a multi-cancer site patient cohort. J Nucl Med. 2015;56:38–44.
Hatt M, Tixier F, Pierce L, Kinahan PE, Le Rest CC, Visvikis D. Characterization of PET/CT images using texture analysis: the past, the presenta… any future? Eur J Nucl Med Mol I. 2017;44:151–65.
Acknowledgements
The authors thank Dr. Yu-Hua Dean Fang, the original developer of the Chang-Gung Image Texture Analysis toolbox, and the staff of the Department of Radiology and Biological Imaging Research Center, University of Fukui, for their clinical and technical supports.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was partly funded by Grants-in-Aid for scientific research from the Japan Society for the Promotion of Science (15H04981, 16K10345, and 16K20181) and Takeda Science Foundation.
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
This study was approved by the Ethics Committee of the University of Fukui, Faculty of Medical Sciences.
Consent to participate
Formal consent was not required for this type of retrospective study.
Rights and permissions
About this article
Cite this article
Tsujikawa, T., Rahman, T., Yamamoto, M. et al. 18F-FDG PET radiomics approaches: comparing and clustering features in cervical cancer. Ann Nucl Med 31, 678–685 (2017). https://doi.org/10.1007/s12149-017-1199-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-017-1199-7