Abstract
Objective
Radioiodide is commonly used to diagnose and treat hyperthyroidism and thyroid carcinoma. However, we could not find any experimental data that strictly compared the biodistribution and thyroid uptake of radioactive iodide between the oral and intravenous (iv) routes with time. This prompted us to compare 123I biodistribution and thyroid uptake to clarify the differences between oral and iv bolus administration in rats.
Methods
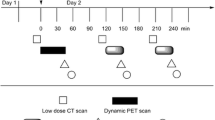
The rats were divided into two groups, A and B (n = 5, each). In the first imaging experiment, Na123I solution (35 MBq/200 μL) was administered as a bolus to the rats orally in group A and intravenously in group B. Two weeks later, the second imaging experiment was performed as a crossover experiment. 123I biodistribution was evaluated visually and quantitatively with a gamma camera at 10 min, 3, 6, 12, 24, and 48 h after 123I administration. Thyroid uptake was compared between oral and iv groups. Correlation of 123I thyroid uptake and whole-body excretion was evaluated. The area under the curve (AUC) of thyroid uptake was also calculated.
Results
123I biodistribution differed visually during 6 h between the two groups. 123I thyroid uptake was significantly higher in the iv group at 10 min (P < 0.05) and in the oral group at 6 or more hour time points (P < 0.005–P < 0.0001) and peaked at 12 h in both groups (oral: 24.4 ± 2.8 %ID, iv: 15.2 ± 2.8 %ID). 123I thyroid uptake showed significant inverse correlations with whole-body excretion from 6 h (r = −0.799, P < 0.0001), and thereafter [12 h (r = −0.957, P < 0.0001), 24 h (r = −0.905, P < 0.0001) and 48 h (r = −0.893, P < 0.0001)], respectively. 123I whole-body excretion was significantly higher in the iv group at each time point (P < 0.0001). The AUC of 123I thyroid uptake was 1.6 times higher in the oral group than the iv group.
Conclusions
These results suggest that radioiodide accumulates in the rat thyroid more effectively by oral than iv administration probably due to slower and lower 123I clearance from the body in the oral administration when administered in a bolus fashion.






Similar content being viewed by others
References
Silberstein EB. Radioiodine: the classic theranostic agent. Semin Nucl Med. 2012;42:164–70.
Watts RWE, Durbin PW. The distribution and acute effect of large dose of Iodine-131 in the rat. UCRL. 1956;3376:2–16.
Durbin PW, Barr GD, Williams MH, Jeung N, Johnston ME. Estimation of radiation dosage delivered by a single massive injection of 131I in the rat. Radiat Res. 1961;14:149–66.
Maloof F, Dobyns BM, Vickery AL. The effect of various dose of radioactive iodine on the function and structure of the thyroid of the rat. Endocrinology. 1952;50:612–38.
Brown J. Extra-thyroidal iodine metabolism in the rat. Endocrinology. 1956;58:68–78.
Perry WF, Hughes JFS. The urinary excretion and thyroid uptake of iodine in renal disease. J Clin Invest. 1952;31:457–63.
Théon AP, Van Vechten MK, Feldman E. Prospective randomized comparison of intravenous versus subcutaneous administration of radioiodine for treatment of hyperthyroidism in cats. Am J Vet Res. 1994;55(12):1734–8.
Schneider P, Biko J, Hänscheid H, Hilliger S, Koutsampelas C, Kranzfelder M, et al. The route of administration (oral vs intravenous) does not influence dose or outcome in Graves’ disease and unifocal autonomy. Eur J Nucl Med Mol Imaging. 2005;32:788–93.
Chung JK. Sodium iodide symporter: its role in nuclear medicine. J Nucl Med. 2002;43:1188–200.
Baril P, Martin-Duque P, Vassaux G. Visualization of gene expression in the live subject using the Na/I symporter as a reporter gene: applications in biotherapy. Br J Pharmacol. 2010;159:761–71.
Ahn BC. Sodium iodide symporter for nuclear molecular imaging and gene therapy: from bedside to bench and back. Theranostics. 2012;2:392–402.
Josefsson M, Grunditz T, Ohlsson T, Bkblad E. Sodium/iodide-symporter: distribution in different mammals and role in entero-thyroid circulation of iodide. Acta Physiol Scand. 2002;175:129–37.
Nicola JP, Basquin C, Portulano C, Reyna-Neyra A, Paroder M, Carrasco N. The Na+/I− symporter mediates active iodide uptake in the intestine. Am J Physiol Cell Physiol. 2009;296:C654–62.
Brown-Grant K. Extrathyroidal iodide concentrating mechanisms. Physiol Rev. 1961;41:189–213.
Nakamoto Y, Saga T, Misaki T, Kobayashi H, Sato N, Ishimori T, et al. Establishment and characterization of a breast cancer cell line expressing Na+/I− symporters for radioiodide concentrator gene therapy. J Nucl Med. 2000;41:1898–904.
Huang M, Batra RK, Kogai T, Lin YQ, Hershman JM, Lichtenstein A, et al. Ectopic expression of the thyroperoxidase gene augments radioiodide uptake and retention mediated by the sodium iodide symporter in non-small cell lung cancer. Cancer Gene Ther. 2001;8:612–8.
Parthasarathy KL, Crawford ES. Treatment of thyroid carcinoma: emphasis on high-dose 131I outpatient therapy. J Nucl Med Technol. 2002;30:165–71.
Conflict of interest
Hiroyuki Kurosawa, Kazuhisa Sakurai, Hideaki Hasegawa, Keisuke Uchida, Hiroyuki Kasahara, Takao Minamizawa are employees of FUJIFILM RI Pharma Co., Ltd. The remaining authors have no conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurosawa, H., Sakurai, K., Hasegawa, H. et al. Comparison of radioactive iodide uptake in the rat thyroid between oral and intravenous bolus administration. Ann Nucl Med 28, 986–993 (2014). https://doi.org/10.1007/s12149-014-0909-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-014-0909-7